��Ŀ����
������һ���ϳɷ�Ӧ������ͼ��A��G��Ϊ�л����Ӧ�в����������ʡ�ԣ���һ����Ӧ�е��Լ��������е���ע�����е�δע��������ͼ��գ� 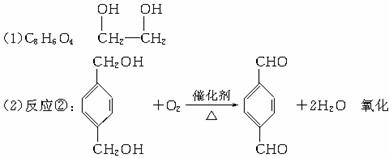
��1��д��D�ķ���ʽ____________________��G�Ľṹ��ʽ________________��
��2��д�����л�ѧ��Ӧ����ʽ��ע����Ӧ����
��Ӧ��___________________________��____________��
��Ӧ��___________________________��____________��
��Ӧ��___________________________��____________��
��3��д�����ַ���������������D��ͬ���칹��Ľṹ��ʽ��������������ȡ�������ڶ�λ���������ࣩ��____________________________��_____________________________��
��4���Լ���������У�ÿ����1.00�ָ߷��ӻ��������������NaOH����_______�֡�
��5����ɫ��ѧ��������ԭ����ԭ�ӣ�ʵ�����ŷŻ����ŷš��������д�������NaOH��̸һ̸��Ľ�������룺___________________________________________________________��

��Ӧ�ݣ�BrCH2CH2Br+2NaOH![]() HOCH2CH2OH+2NaBr ȡ����ˮ�⣩
HOCH2CH2OH+2NaBr ȡ����ˮ�⣩
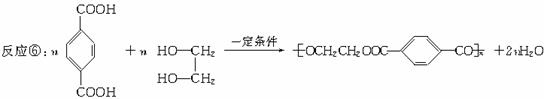 �ۺϣ����ۣ�
�ۺϣ����ۣ�
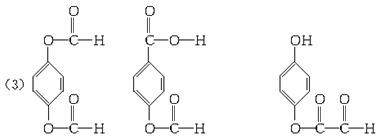
��4��0.833
��5���ɸ����ȼ�գ�������NaCl��Һ��⣺
2NaCl+2H2O![]() 2NaOH+H2��+Cl2
2NaOH+H2��+Cl2
����NaOHѭ�������л��ϳɣ�����Cl2���û���Br2��
Cl2+2NaBr====2NaCl+Br2
ѭ�������л��ϳɣ�H2�ռ�������Ʒ���ۡ������������ɵ÷֣�
������ͼ�У���B��������������ΪD��DΪ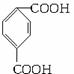 ���ɸ߷��ӻ�����Ľṹ��ʽ֪GΪHOCH2CH2OH��AΪ
���ɸ߷��ӻ�����Ľṹ��ʽ֪GΪHOCH2CH2OH��AΪ![]() ��BΪ
��BΪ![]() ��FΪBrCH2CH2Br��
��FΪBrCH2CH2Br��
��Ӧ��Ϊ![]() ��Ӧ��ΪBrCH2CH2Br+2NaOH
��Ӧ��ΪBrCH2CH2Br+2NaOH![]() HOCH2CH2OH+2NaBr���ɷ�Ӧ�٢ݢ�
HOCH2CH2OH+2NaBr���ɷ�Ӧ�٢ݢ�
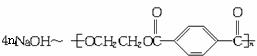
160 192
![]() 1 t
1 t
���ÿ����1.00�ָ߷��ӻ��������������NaOH����![]() =0.833 t��
=0.833 t��

��ÿһС��2�ֹ�12�֣�������һ���ϳɷ�Ӧ������ͼ��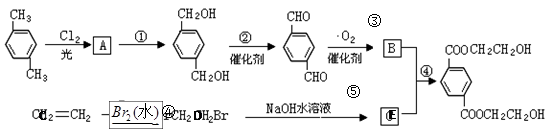
��1����д���������ʵĽṹ��ʽ
| A���� | B���� |
| C���� | D�� . |
�� ���ݡ� ��
 ת���ɾ���ϩ������һ�������ù����ļ״�������������Ӧ��ʵ�ֵġ�
ת���ɾ���ϩ������һ�������ù����ļ״�������������Ӧ��ʵ�ֵġ�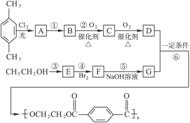
 ת���ɾ���ϩ������һ�������ù����ļ״�������������Ӧ��ʵ�ֵġ�
ת���ɾ���ϩ������һ�������ù����ļ״�������������Ӧ��ʵ�ֵġ�