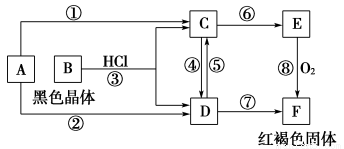
��Ŀ����
����ȷ��ʾ���з�Ӧ�����ӷ���ʽ��
A��NaNO2��Һ�м�������KMnO4��Һ��2MnO4��+5NO2��+ 6H+ �� 2Mn2+ + 5NO3�� + 3H2O
B��̼�������Һ�м�����������������Һ: NH4++HCO3��+2OH�� �� CO32��+ NH3?H2O +H2O
C��Fe(NO3)3��Һ�м��������HI��Һ��2Fe3+ + 2I�� �� 2Fe 2+ + I2
D���ö��Ե缫��������Ȼ��ƣ�2Cl��+2H2O �� Cl2��+H2��+2OH��
A
��������
���������A��MnO4?�����������°�NO2-����ΪNO3?�����ӷ���ʽΪ��2MnO4��+5NO2��+ 6H+ �� 2Mn2+ + 5NO3�� + 3H2O����A��ȷ��B��̼�������Һ�м�����������������Һ������BaCO3��������B����C��Fe(NO3)3��Һ�м��������HI��Һ�������ķ�ӦΪNO3?��H+����I?����C����D���ö��Ե缫��������Ȼ��ƣ����ӷ���ʽΪ��2Cl?+2Na+ Cl2��+2Na����D����
Cl2��+2Na����D����
���㣺���⿼�����ӷ���ʽ��

25 ��ʱ������ƽ�ⳣ����
��ѧʽ | CH3COOH | H2CO3 | HClO |
����ƽ�ⳣ�� | 1.8��10��5 | K1 4.3��10��7 K2 5.6��10��11 | 3.0��10��8 |
�ش��������⣺
��1�����ʵ���Ũ��Ϊ0.1 mol/L�������������ʣ�a.Na2CO3��b.NaClO��c.CH3COONa��d.NaHCO3��pH�ɴ�С��˳����________________��(����)
��2��������0.1 mol/L��CH3COOH��Һ��ˮϡ���̣����б���ʽ������һ����С����_____��
A��c(H��) B��c(H��)/c(CH3COOH) C��c(H��)��c(OH��) D��c(OH��)/c(H��)
��3�����Ϊ10 mL pH��2�Ĵ�����Һ��һԪ��HX�ֱ��ˮϡ����1 000 mL��ϡ����pH�仯��ͼ����HX�ĵ���ƽ�ⳣ��________(����ڡ��������ڡ���С�ڡ�)�����ƽ�ⳣ����������__________________��

ϡ�ͺ�HX��Һ��ˮ���������c(H��)________������Һ��ˮ���������c(H��)(����ڡ��������ڡ���С�ڡ�)�������ǣ�__________________��
��4��25 ��ʱ��CH3COOH��CH3COONa�Ļ����Һ������û��ҺpH��6������Һ��c(CH3COO��)��c(Na��)��________��(��ȷ��ֵ)