��Ŀ����
5�����ǵ����ϼ�Ϊ�ḻ��Ԫ�أ���1��Li3N�����е���N3-���ڣ���̬N3-�ĺ�����ӵĹ����ʾʽΪ
 ��
����2��N��N�ļ���Ϊ942kJ/mol��N-N�����ļ���Ϊ247kJ/mol������˵��N2�еĦм��ȦҼ��ȶ�����ҡ����С�����
��3�����ݱ����ݣ�д��������������Ӧ���ɰ������Ȼ�ѧ����ʽN2��g��+3H2��g��=2NH3��g����H=-92kJ•mol-1��
| ��ѧ�� | N-N | N=N | N��N | N-H | H-H |
| ����/kJ•mol-1 | 159 | 418 | 946 | 391 | 436 |
���� ��1��N3-����ԭ�Ӻ����10�����ӣ������������ԭ����д�������Ų�ʽ����������ԭ��������������Ų�ͼ��
��2��N��N�к���2���м���1���Ҽ�����֪N��N����Ϊ942kJ/mol��N-N��������Ϊ247kJ/mol�������1���м��ļ������жϣ�
��3����ѧ��Ӧ��ʵ���Ǿɼ��Ķ��Ѻ��¼����γɣ���Ӧ�ȵ��ڷ�Ӧ���ܼ��ܼ��������ܼ��ܣ�
��� �⣺��1��Nԭ��ԭ������Ϊ7��N3-����ԭ�Ӻ����10�����ӣ��������ﵽ�ȶ��ṹ�������������ԭ����д�������Ų�ʽ1s22s22p6��ÿ��������ֻ�������������ӣ��������෴��ԣ����ù����ʾ����������Ų�ͼΪ�� ��
��
�ʴ�Ϊ�� ��
��
��2��N��N�к���2���м���1���Ҽ�����֪N��N����Ϊ942kJ/mol��N-N��������Ϊ247kJ/mol����1���м��ļ���Ϊ$\frac{942-247}{2}$kJ/mol=347.5kJ/mol����N2�еĦм����ܴ��ڦҼ����ܣ����ȶ���
�ʴ�Ϊ���У��ң�
��3����Ӧ�Ⱦ��Ƕϼ����յ��������γɻ�ѧ�����ų��������IJ�ֵ�����ڷ�ӦN2��g��+3H2��g��=2NH3��g����
��Ӧ�ȡ�H=3��436 kJ/mol+946kJ/mol-2��3��391kJ/mol=-92kJ/mol�����Ȼ�ѧ����ʽ��N2��g��+3H2��g��=2NH3��g����H=-92kJ/mol��
�ʴ�Ϊ��N2��g��+3H2��g��=2NH3��g����H=-92kJ•mol-1��
���� ���⿼���˺�������Ų�ͼ����д����ѧ�����ȶ����жϡ��Ȼ�ѧ����ʽ����д���ѵ��ǻ�ѧ���ȶ��Ե��жϣ����ض�ѧ�����������������ڹ���ѧ���Ļ��������ѧ���Ĵ���������ѧϰЧ�ʣ�

 Сѧ��10���ӿ������100��ϵ�д�
Сѧ��10���ӿ������100��ϵ�д�| A�� | ij��Һ�еμ�AgNO3��Һ�а�ɫ������˵��ԭ��Һ��һ������Cl- | |
| B�� | ij��Һ�ȼ����������������ٵμ�BaCl2��Һ�а�ɫ��������ԭ��Һ��һ������SO42- | |
| C�� | ij��ɫ��Һ�����̪�Ժ�ɫ������Һ��һ�����д�����H+ | |
| D�� | ij��Һ�еμ�ϡ����������ɫ���壬˵��ԭ��Һ��һ������CO32- |
 ����Cu2O���ھ��������Ĵ����ܶ��ܵ���ע���±�Ϊ��ȡCu2O�����ַ�����
����Cu2O���ھ��������Ĵ����ܶ��ܵ���ע���±�Ϊ��ȡCu2O�����ַ�����| ������ | ��̿���ڸ��������»�ԭCuO |
| ������ | ��ⷨ����ӦΪ2Cu+H2O $\frac{\underline{\;���\;}}{\;}$ Cu2O+H2���� |
| ������ | ���£�N2H4����ԭ����Cu��OH��2 |
��2����֪��2Cu��s��+$\frac{1}{2}$O2��g��=Cu2O��s����H=-akJ•mol-1
C��s��+$\frac{1}{2}$O2��g��=CO��g����H=-bkJ•mol-1
Cu��s��+$\frac{1}{2}$O2��g��=CuO��s����H=-ckJ•mol-1
�������ķ�Ӧ��2CuO��s��+C��s��=Cu2O��s��+CO��g������H=-��b-2c-$\frac{a}{2}$��kJ•mol-1��
��3��������������ӽ���Ĥ���Ƶ��Һ��OH-��Ũ�ȶ��Ʊ�����Cu2O��װ����ͼ��ʾ���õ�ص���������Cu2O��ӦʽΪ2Cu-2e-+2OH-=Cu2O+H2O��
��4��������Ϊ������������Һ̬�£�N2H4����ԭ����Cu��OH��2���Ʊ�����Cu2O��ͬʱ�ų�N2�����Ʒ��Ļ�ѧ����ʽΪ4Cu��OH��2+N2H4$\frac{\underline{\;\;��\;\;}}{\;}$2Cu2O+N2��+6H2O��
��5������ͬ���ܱ������У����������ַ����Ƶõ�Cu2O�ֱ���д��ֽ�ˮ��ʵ�飺2H2O��g��$?_{Cu_{2}O}^{����}$2H2��g��+O2��g����H��0��ˮ������Ũ�ȣ�mol/L����ʱ��t��min���仯�����ʾ��
| ��� | �¶� | 0 | 10 | 20 | 30 | 40 | 50 |
| �� | T1 | 0.050 | 0.0492 | 0.0486 | 0.0482 | 0.0480 | 0.0480 |
| �� | T1 | 0.050 | 0.0488 | 0.0484 | 0.0480 | 0.0480 | 0.0480 |
| �� | T2 | 0.10 | 0.094 | 0.090 | 0.090 | 0.090 | 0.090 |
A��ʵ����¶ȣ�T2��T1
B��ʵ���ǰ20min��ƽ����Ӧ���� v��O2��=7��10-5 mol•L-1 min-1
C��ʵ��ڱ�ʵ������õĴ�����Ч�ʸߣ�
 ����β���dz��е���Ҫ������Ⱦ��о���������β����Ϊ������������Ҫ�����������ϰ�װ��Ч��ת��������ʹ����β���е���Ҫ��Ⱦ��������Ӧ�����������ʣ�����ʹ������ȼ�ϵ����������������������β����Ⱦ��
����β���dz��е���Ҫ������Ⱦ��о���������β����Ϊ������������Ҫ�����������ϰ�װ��Ч��ת��������ʹ����β���е���Ҫ��Ⱦ��������Ӧ�����������ʣ�����ʹ������ȼ�ϵ����������������������β����Ⱦ����1��H2��CO���Դ���ԭNO�Դﵽ������Ⱦ��Ŀ�ģ�
��֪��N2��g��+O2��g��=2NO��g����H=+180.5kJ•mol-1
2H2��g��+O2��g��=2H2O��l����H=-571.6kJ•mol-1
��H2��g����NO��g����Ӧ����N2��g����H2O��l�����Ȼ�ѧ����ʽ��2H2��g��+2NO��g��=N2��g��+2H2O��l����H=-752.1 kJ•mol-1��
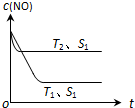
��2��������һ��ʱ�������������ı���������ѧ��Ӧ���ʣ�ͼ��ʾ��������������ʱ����Ӧ��2CO��g��+2NO��g��?2CO2��g��+N2��g�� ��NO��Ũ��[c��NO��]���¶ȣ�T���������������S����ʱ�䣨t���ı仯���ߣ�
�ٸ÷�Ӧ�ġ�H��0 ���������������
���������ı����S1��S2������ͼ�л���c ��NO�� ��T1��S2�����´ﵽƽ������еı仯���ߣ�
��3����ij�¶�ʱ������������̽��ij�ִ��������µķ�Ӧ���ʣ������崫������ò�ͬʱ���NO��COŨ�������

| ʱ��/s | 0 | 1 | 2 | 3 | 4 |
| c ��NO������10-3 mol•L-1�� | 1.00 | 0.50 | 0.20 | 0.10 | 0.10 |
| c ��CO������10-3 mol•L-1�� | 3.00 | 2.50 | 2.20 | 2.10 | 2.10 |
�ڼ���ǰ2s�ڵ�ƽ����Ӧ����v ��N2����
�ۼ���ﵽƽ��ʱNO��ת���ʣ�
| A�� | ����������Һ�������ЧӦ�� | B�� | ��ѹ��������Ӿ���� | ||
| C�� | �Ӻ����㴦�ɳ���ɳ�ޣ��۳��� | D�� | ��������Ӧ�ã������˶��� |
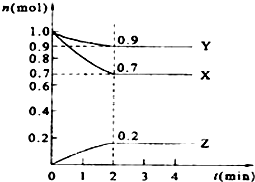 ij�¶�ʱ����2L���ܱ������У�X��Y��Z�������ʵ�����ʱ��ı仯������ͼ��ʾ��
ij�¶�ʱ����2L���ܱ������У�X��Y��Z�������ʵ�����ʱ��ı仯������ͼ��ʾ��
 ��������һ����Ҫ���л�����ԭ�ϣ���е�Ϊ38.4�森�Ʊ��������һ�ַ������Ҵ��������ᷴӦ���÷�Ӧ�Ļ�ѧ����ʽ��CH3CH2OH+HBr$��_{��}^{Ũ����}$CH3CH2Br+H2O��
��������һ����Ҫ���л�����ԭ�ϣ���е�Ϊ38.4�森�Ʊ��������һ�ַ������Ҵ��������ᷴӦ���÷�Ӧ�Ļ�ѧ����ʽ��CH3CH2OH+HBr$��_{��}^{Ũ����}$CH3CH2Br+H2O��