��Ŀ����
��12�֣��������洦����Ƥ�����ơ�ӡȾ�ȶ�������ɸ���Ⱦ�����۸������۸����Ըߣ����ױ����������������������
��1����ҵ�ϴ������Ժ�Cr2O72����ˮ�ķ������£�
����Cr2O72�������Է�ˮ�м���FeSO4��Һ��ʹCr2O72��ȫ��ת��ΪCr3+��д���÷�Ӧ�����ӷ���ʽ�� ��
�ڵ�����Һ��pH��ʹCr3����ȫ������ʵ���Ҵ��Բⶨ��ҺpH�ķ���Ϊ
��25�棬��������Һ��pH��8������Һ�в���Cr3+�����ʵ���Ũ��Ϊ mol/L������֪25��ʱ��Ksp[Cr(OH)3]��6.3��10��31��
��2����Ԫ����Ũ�ȵIJⶨ��ȷ��ȡ25.00mL��Cr2O72����Cr3+�����Է�ˮ�������м���������(NH4)2S2O8��Һ��Cr3+������Cr2O72������г�ȥ������(NH4)2S2O8����������Һ�м��������KI��Һ����ַ�Ӧ���Ե���Ϊָʾ���������еμ�0.015mol/L��Na2S2O3����Һ���յ�ʱ����Na2S2O3��Һ20.00mL��
�����ˮ�и�Ԫ����Ũ�ȣ���λ��mg��L��1��д��������̣���
��֪�ⶨ�����з����ķ�Ӧ���£�
��2Cr3+ + 3S2O82�� + 7H2O Cr2O72�� + 6SO42�� + 14H+
Cr2O72�� + 6SO42�� + 14H+
��Cr2O72�� + 6I�� + 14H+ 2Cr3+ + 3I2 + 7H2O
2Cr3+ + 3I2 + 7H2O
��I2 + 2S2O32�� 2I�� + S4O62��
2I�� + S4O62��
��1����Cr2O72����6Fe2����14H����2Cr3����6Fe3����7H2O��2�֣�
�ڽ�pH��ֽ���ڽྻ�ı������ϣ��ò�����պȡ��Һ�� ����pH��ֽ�ϣ��������ɫ�����գ�2�֣�
6.3��10��13��2�֣�
��2���ɷ���ʽ��֪��Cr~3Na2S2O3
n(Na2S2O3)��20.00mL��0.015mol/L��3��10��4mol
n(Cr)��1��10��4mol
m(Cr)��1��10��4mol��52g��mol��1��5.2��10��3 g��5.2mg
��ˮ�и�Ԫ����Ũ�ȣ�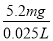 ��208 mg��L��1��6�֣�
��208 mg��L��1��6�֣�
��������
�����������1����Cr2O72�������������°�Fe2+����ΪFe3+����ƽ�ɵ����ӷ���ʽ��Cr2O72����6Fe2����14H����2Cr3����6Fe3����7H2O��
��ʵ������pH��ֽ���Բⶨ��ҺpH���ⶨ����Ϊ����pH��ֽ���ڽྻ�ı������ϣ��ò�����պȡ��Һ�� ����pH��ֽ�ϣ��������ɫ�����գ�����Ksp[Cr(OH)3]��6.3��10��31��c��Cr3+��?c��OH?��3=6.3��10��31��pH��8��c��OH?��=10-6mol?L?1������ɵ�c��Cr3+��=6.3��10��13mol?L?1��
��2������Ŀ������ѧ����ʽ��֪��Ӧ��ϵ��Cr~3Na2S2O3
n(Na2S2O3)��20.00mL��0.015mol/L��3��10��4mol
n(Cr)��1/3 n(Na2S2O3)=1��10��4mol
m(Cr)��1��10��4mol��52g��mol��1��5.2��10��3 g��5.2mg
��ˮ�и�Ԫ����Ũ�ȣ�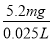 ��208 mg��L��1
��208 mg��L��1
���㣺���⿼�����ӷ���ʽ����д��pH�IJⶨ����ѧ���㡣

 �¿α�����Ķ�ѵ��ϵ�д�
�¿α�����Ķ�ѵ��ϵ�д� ������������Ӧ����ϵ�д�
������������Ӧ����ϵ�д�

 BaSO4��
BaSO4�� NiOOH + MH
NiOOH + MH
 Si + 2CO��
Si + 2CO��