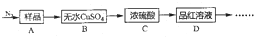
��Ŀ����
10����һ��1L���ܱ������У�����2mol A��1mol B���������з�Ӧ��2A��g��+B��g��?3C��g��+D��s�����ﵽƽ��ʱ��C��Ũ��Ϊ1.2mol•L-1����1��ά���������¶Ȳ��䣬����С�������������ƽ�ⲻ�ƶ����������Ӧ�������淴Ӧ����������
��2��ά��������������¶Ȳ��䣬���������������Ϊ��ʼ���ʣ��ﵽƽ���C��Ũ������1.2mol•L-1����BC��
A��4mol A+2mol B
B��3mol C+1mol D
C��2mol A+1mol B+1mol D��
���� ��1���÷�ӦΪ�����������Ŀ��淴Ӧ��ѹǿ��Ӱ�컯ѧƽ�⣻
��2��ά������������¶Ȳ��䣬�ﵽƽ��ʱC��Ũ����Ϊ1.2mol/L��Ϊ��Чƽ�⣬��Ϊ��ȫ��Чƽ�⣬����ѧ������ת�����������2molA��1molB��
��� �⣺��1����С����������൱������ѹǿ�����ڸ÷�ӦΪ�����������ķ�Ӧ����������ѹǿ����ѧƽ�ⲻ�ƶ���
�ʴ�Ϊ������
��2�����ݵ�Чƽ�⣬�Ѹ����ʶ�����ɷ���ʽ��ߵ����ʣ����ʼ�����2molA��1molB��ȼ��ɣ�
A.4mol A+2mol B ���ʼ2mol A��1molB��ͬ�������ڵ�Чƽ�⣬��A����
B.3molC+l mol D�����A��B����A��BŨ�ȷֱ�Ϊ2mol��1mol�����ʼŨ����ͬ����B��ȷ��
C.2mol A+1mol B+1mol D������DΪ��̬����Ӱ�컯ѧƽ�⣬����ԭ��Ӧ��2mol A��1molB��Ϊ��Чƽ�⣬�ﵽƽ��ʱC��Ũ����ȻΪ1.2mol/L����C��ȷ��
�ʴ�Ϊ��BC��
���� ���⿼���˵�Чƽ���Ӱ�졢��Чƽ����жϼ�Ӧ�ã���Ŀ�Ѷ��еȣ�ע�����յ�Чƽ������ͼ��жϷ�������ȷ��ѧƽ�⼰��Ӱ�����أ���������ע��D��״̬Ϊ���壬Ϊ�״��㣮

��ϰ��ϵ�д�
�����Ŀ
8�� 25��ʱ����֪��
25��ʱ����֪��
H2CO3?H++HCO3- K1=4.3��10-7
HCO3-?H++CO32- K2=5.6��10-11
H2O?H++OH- KW=1.0��10-14
��ȡ10.6g Na2CO3 �����������õ�һ�����Ϊ1L ����Һ����Һ�в�������pH �Ĺ�ϵ��ͼ��ʾ����25��ʱ�������й���Һ������Ũ�ȹ�ϵ������ȷ���ǣ�������
 25��ʱ����֪��
25��ʱ����֪��H2CO3?H++HCO3- K1=4.3��10-7
HCO3-?H++CO32- K2=5.6��10-11
H2O?H++OH- KW=1.0��10-14
��ȡ10.6g Na2CO3 �����������õ�һ�����Ϊ1L ����Һ����Һ�в�������pH �Ĺ�ϵ��ͼ��ʾ����25��ʱ�������й���Һ������Ũ�ȹ�ϵ������ȷ���ǣ�������
| A�� | W ����ʾ����Һ�У�c��Na+��+c��H+��=2c��CO32-��+c��OH-��+c��HCO3-�� | |
| B�� | pH=4 ����Һ�У�c��H2CO3��+c��HCO3-��+c��CO32-��=0.1 mol•L-1 | |
| C�� | pH=8 ����Һ�У�c��H+��+c��H2CO3��+c��HCO3-��=c��OH-��+c��Cl-�� | |
| D�� | pH=11 ����Һ�У�CO32-+H2O HCO3-+OH- K��1.8��l0-4 |
9����֪������������������Һ�з������з�Ӧ�٣�����������Һ�����ܸ�ˮ�������з�Ӧ�ڣ�
��CH3COOH+Na0H=CH3COONa+H20
��CH3COONa+H20?CH3COOH+Na0H
������ѧ����ʽ�ٺ͢ڿɺϲ�дΪ
��CH3COOH+Na0H=CH3COONa+H20
����������ȷ���ǣ�������
��CH3COOH+Na0H=CH3COONa+H20
��CH3COONa+H20?CH3COOH+Na0H
������ѧ����ʽ�ٺ͢ڿɺϲ�дΪ
��CH3COOH+Na0H=CH3COONa+H20
����������ȷ���ǣ�������
| A�� | ��Ӧ�ٺ͢ڶ������ӷ�Ӧ | B�� | ��Ӧ�ٺ͢ڶ��������� | ||
| C�� | ��Ӧ�ٺ͢ڶ��ų����� | D�� | ��Ӧ�ۼȲ���������Ҳ���ų����� |
6���й����ӹ����˵������ط�����ȷ���ǣ�������
| A�� | ������Al3+����Һ�У�K+��Na+��NO3-��CO32-���ܴ������棬����Al2��CO3��3�������� | |
| B�� | ������Fe3+����Һ�У�K+Mg2+��I-��Br2-���ܴ������棬��2Fe3++2I-=2Fe2++I2 | |
| C�� | ��ˮ�����c��H+��=1��10-14mol/L����Һ�У�Ca2+��NO3-��HCO3-��Cl-���ܴ������棬����Һ�����ԣ���HCO3-��Ӧ����CO2���� | |
| D�� | ��NH4Fe��SO4��2��Һ�У�Na+��K+��S2-��SCN-���ܴ������棬ֻ����Fe3+��S2-��Ӧ��������ɫ��S���� |
15��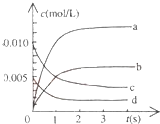 ��2L�ܱ������ڣ�800��ʱ��Ӧ��2NO��g��+O2��g��?2NO2��g����ϵ�У�n��NO����ʱ��ı仯�����
��2L�ܱ������ڣ�800��ʱ��Ӧ��2NO��g��+O2��g��?2NO2��g����ϵ�У�n��NO����ʱ��ı仯�����
��1��д���÷�Ӧ��ƽ�ⳣ������ʽ��K=$\frac{{c}^{2}��N{O}_{2}��}{{c}^{2}��NO��•c��{O}_{2}��}$��
��2����ͼ�б�ʾNO2�ı仯��������b����O2��ʾ��0��2s�ڸ÷�Ӧ��ƽ������v=0.0015mol/��L•s��
��3����˵���÷�Ӧ�Ѵﵽƽ��״̬����bc��
a��v��NO2��=2v��O2�� b��������ѹǿ���ֲ���
c��v����NO��=2v����O2�� d���������ܶȱ��ֲ���
��4��ijͬѧ�������ϵ�֪���÷�ӦK300����K350����25��ʱ������1molNO2�������仯Ϊ56.4kJ�����Ǹ�ͬѧ�������������ע����+������-����������Ŀ��Ϣ���÷�Ӧ���Ȼ�ѧ����ʽΪ2NO��g��+O2��g��?2NO2��g����H=-112.8kJ/mol��
��5��Ϊʹ�÷�Ӧ�ķ�Ӧ����������ƽ��������Ӧ�����ƶ�����c��
a����ʱ�����NO2���� b���ʵ������¶�
c������O2��Ũ�� d��ѡ���Ч������
 ��2L�ܱ������ڣ�800��ʱ��Ӧ��2NO��g��+O2��g��?2NO2��g����ϵ�У�n��NO����ʱ��ı仯�����
��2L�ܱ������ڣ�800��ʱ��Ӧ��2NO��g��+O2��g��?2NO2��g����ϵ�У�n��NO����ʱ��ı仯�����| ʱ�䣨s�� | 0 | 1 | 2 | 3 | 4 | 5 |
| n��NO����mol�� | 0.020 | 0.011 | 0.008 | 0.007 | 0.007 | 0.007 |
��2����ͼ�б�ʾNO2�ı仯��������b����O2��ʾ��0��2s�ڸ÷�Ӧ��ƽ������v=0.0015mol/��L•s��
��3����˵���÷�Ӧ�Ѵﵽƽ��״̬����bc��
a��v��NO2��=2v��O2�� b��������ѹǿ���ֲ���
c��v����NO��=2v����O2�� d���������ܶȱ��ֲ���
��4��ijͬѧ�������ϵ�֪���÷�ӦK300����K350����25��ʱ������1molNO2�������仯Ϊ56.4kJ�����Ǹ�ͬѧ�������������ע����+������-����������Ŀ��Ϣ���÷�Ӧ���Ȼ�ѧ����ʽΪ2NO��g��+O2��g��?2NO2��g����H=-112.8kJ/mol��
��5��Ϊʹ�÷�Ӧ�ķ�Ӧ����������ƽ��������Ӧ�����ƶ�����c��
a����ʱ�����NO2���� b���ʵ������¶�
c������O2��Ũ�� d��ѡ���Ч������
2������һ�����ʵ���Ũ�ȵ���Һʱ�����ڲ���������ʹ��Һ���Գ�������ƿ�Ŀ̶ȣ����ߣ�����ʱӦ��ȡ�Ĵ�ʩ�ǣ�������
| A�� | �ټӵ����ʵ�����ƿ��ȥ | B�� | Ӱ�첻���ٴ��� | ||
| C�� | �ý�ͷ�ι�ȷ���������ϵ���Һ | D�� | �������� |
19�������й�˵�����ʾ��ȷ���ǣ�������
| A�� | ���ۺ���ά�ض����ã�C6H10O5��n��ʾ������Ϊͬ���칹�� | |
| B�� | Mg5��Si4O10��2��OH��2•4H2O����������ʽ��5MgO•8SiO2•5H2O | |
| C�� | Fe-2e-=Fe2+�ɱ�ʾ�������ⸯʴ��������ʴ��������Ӧ | |
| D�� | �Ҵ����ӵı���ģ�ͣ�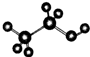 |
20������ʵ���������ȷ���ǣ�������
| A�� | ��ȡ����ʱ������ѡ��CCl4��ƾ���Ϊ��ȡ���ӵ�ˮ����ȡ�� | |
| B�� | ��������ʱ��Ӧʹ������е�ˮ����ȫ���ɺ���ֹͣ���� | |
| C�� | ��Һ����ʱ���²�Һ��ӷ�Һ©���¿ڷų����ϲ�Һ����¿ڷų�����һ���ձ��� | |
| D�� | �������ʱ���ռ�����ֺ���ֹͣ���ȣ����ָ����º���ֹͣͨ����ˮ |