��Ŀ����
13��������Ԫ��X��Y��Z��ɵĻ�����Y2X��ZX2��Y2X����ˮ�γɵ���Һ����ZX2��Ӧ����һ�ֻ�����Y2ZX3����֪����Ԫ��ԭ�ӵ���������Ϊ25����Z��Y��ԭ������֮�ͱ�X��ԭ������2������1��Zԭ�ӵ������������Ǵ�����������2�����Իش���1��X��Y��ZԪ�ص����Ƽ����ţ�X������O�� Y���ƣ�Na�� Z��̼��C��
��2��д��ZX2�Ļ�ѧʽ��CO2
��3��д��Y2X��Ӧˮ����ĵ���ʽ
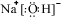 ����ָ�����д��ڵĻ�ѧ�������Ӽ����ۼ�
����ָ�����д��ڵĻ�ѧ�������Ӽ����ۼ���4��д��Y2X����ˮ����Һ��ZX2��Ӧ�Ļ�ѧ����ʽ��2NaOH+CO2=Na2CO3+H2O��
���� ��Zԭ�ӵ������������Ǵ�����������2������ZΪ�������Ӳ�ʱ���Ƴ�ZΪ̼Ԫ�أ�
��������Ԫ��ԭ�ӵĵ�������Ϊ25����Z��Y��ԭ������֮�ͱ�X��ԭ��������2������1������Xԭ�ӵ�������Ϊx��Yԭ�ӵ�������Ϊy���������⣬�ɵó����¹�ϵ��$\left\{\begin{array}{l}{6+x+y=25}\\{6+y=2x+1}\end{array}\right.$����ã�x=8��y=11����XΪ��Ԫ�أ�YΪ��Ԫ�أ�Ȼ�����õ��ʼ�����������������
��� �⣺��1�������Ϸ�����֪XΪ��Ԫ�أ�O����YΪ��Ԫ�أ�Na����ZΪ̼Ԫ�أ�C�����ʴ�Ϊ������O�����ƣ�Na����̼��C����
��2��XΪ��Ԫ�أ�O����ZΪ̼Ԫ�أ�C����ZX2�Ļ�ѧʽΪCO2���ʴ�Ϊ��CO2��
��3��������Y2X��Ӧ��ˮ����ΪNaOH�������ʽΪ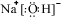 �����ڵĻ�ѧ�������Ӽ����ۼ���
�����ڵĻ�ѧ�������Ӽ����ۼ���
�ʴ�Ϊ��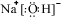 �����Ӽ����ۼ���
�����Ӽ����ۼ���
��4��NaOH��Һ�������̼��Ӧ����̼���ƣ���ѧ����ʽΪ2NaOH+CO2=Na2CO3+H2O���ʴ�Ϊ��2NaOH+CO2=Na2CO3+H2O��
���� ���⿼��λ�á��ṹ�����ʵĹ�ϵ��Ϊ�߿��������ͣ���һ���ۺ���ϰ�⣬ѧ����Ϥ�й�ԭ�ӵĺ�������Ų���ԭ���������������Ĺ�ϵ������ʽ����ѧ�������ʵ����ʵȷ����֪ʶ�ǽ����Ĺؼ���

| A�� | ��ϩ�Ľṹ��ʽ��CH2CH2 | B�� | ����Ľṹʽ��C2H6 | ||
| C�� | ���ķ���ʽ��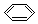 | D�� |  --���ĵ���ʽ�� --���ĵ���ʽ�� |
| A�� | MgO+H2SO4�TMgSO4+H2O | B�� | Na2O+H2O�T2NaOH | ||
| C�� | Zn+H2SO4�TZnSO4+H2�� | D�� | SO3+H2O�TH2SO4 |
| X | Y | ||
| Z | W | Q |
| A�� | X��W�������γ�Ħ��������ͬ�Ļ������������������Ϊ1��2 | |
| B�� | X��ZԪ���γɵĻ�����㷺Ӧ�������켯�ɵ�·��̫���ܵ�ذ�� | |
| C�� | �������Ӧ��ˮ�������ԣ�W��Q | |
| D�� | ԭ�Ӱ뾶��С��ϵ��Q��W��Z��Y |
| A�� | ��ʳ����ϴ��ˮƿ�е�ˮ������Ҫ�ɷ���CaCO3��Mg��OH��2�� | |
| B�� | ��ú����ȼ����Ȼ��Ϊ�����ṩ���� | |
| C�� | �ղ��ù��������������ó����ֺ���ɫ�� | |
| D�� | ţ�̾��ñ��ʸ��� |