��Ŀ����
��FeCl3��AlCl3�Ļ����Һ100ml�������м��������NaOH��Һ����ַ�Ӧ���ˣ���������ɳ���10.7g��
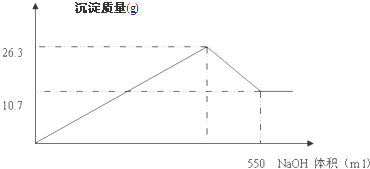
��1��д�������Һ�м������NaOH��Һ�����ӷ���ʽ �� ��
��2����10.7g�����������գ����õ����� �ˣ�
��3��FeCl3�����ʵ�����Ũ���� mol/L��
��4���ڼ���NaOH��Һ�Ĺ����У����ɳ��������������NaOH��Һ���������ͼ��ʾ������AlCl3�����ʵ�����Ũ�� mol/L��
��5������NaOH��Һ�����ʵ�����Ũ�� mol/L��

��1��д�������Һ�м������NaOH��Һ�����ӷ���ʽ
��2����10.7g�����������գ����õ�����
��3��FeCl3�����ʵ�����Ũ����
��4���ڼ���NaOH��Һ�Ĺ����У����ɳ��������������NaOH��Һ���������ͼ��ʾ������AlCl3�����ʵ�����Ũ��
��5������NaOH��Һ�����ʵ�����Ũ��
���㣺���ӷ���ʽ���йؼ���,�йػ���ﷴӦ�ļ���
ר�⣺
��������1���Ȼ������������Ʒ�Ӧ�������������������Ȼ��ƣ��Ȼ��������������������Һ��Ӧ����ƫ��������ˮ��
��2�����ճ���10.7gΪFe��OH��3�����ȷֽ�����Fe2O3������n=
���������������ʵ���������FeԪ���غ����n��Fe2O3�����ٸ���m=nM����m��Fe2O3����
��3������FeԪ���غ����n��FeCl3��=n[Fe��OH��3]���ٸ���c=
����c��FeCl3����
��4���������ʱΪ26.3g��Ϊ������������������������֮�ͣ�������������������������n=
�����������������ʵ���������AlԪ���غ����n��AlCl3�����ٸ���c=
����c��AlCl3����
��5������550mL NaOH��Һ��ʱ����Һ������ΪNaCl��NaAlO2�������������غ�n��NaCl��=3n��FeCl3��+3n��AlCl3��������AlԪ���غ�n��NaAlO2��=n��AlCl3���������������غ�n��NaOH��=n��NaCl��+n��NaAlO2�����ٸ���c=
����c��NaOH����
��2�����ճ���10.7gΪFe��OH��3�����ȷֽ�����Fe2O3������n=
| m |
| M |
��3������FeԪ���غ����n��FeCl3��=n[Fe��OH��3]���ٸ���c=
| n |
| V |
��4���������ʱΪ26.3g��Ϊ������������������������֮�ͣ�������������������������n=
| m |
| M |
| n |
| V |
��5������550mL NaOH��Һ��ʱ����Һ������ΪNaCl��NaAlO2�������������غ�n��NaCl��=3n��FeCl3��+3n��AlCl3��������AlԪ���غ�n��NaAlO2��=n��AlCl3���������������غ�n��NaOH��=n��NaCl��+n��NaAlO2�����ٸ���c=
| n |
| V |
���
�⣺��1���Ȼ������������Ʒ�Ӧ�������������������Ȼ��ƣ���Ӧ���ӷ���ʽΪ��Fe3++3OH-=Fe��OH��3�����Ȼ��������������������Һ��Ӧ����ƫ��������ˮ����Ӧ���ӷ���ʽΪ��Al3++4OH-=AlO2-+2H2O��
�ʴ�Ϊ��Fe3++3OH-=Fe��OH��3����Al3++4OH-=AlO2-+2H2O��
��2�����ճ���10.7gΪFe��OH��3�����ȷֽ�����Fe2O3��n[Fe��OH��3]=
=0.1mol������FeԪ���غ�n��Fe2O3��=
=0.05mol����m��Fe2O3��=0.05mol��160g/mol=8.0g��
�ʴ�Ϊ��8.0��
��3������FeԪ���غ�n��FeCl3��=n[Fe��OH��3]=0.1mol����c��FeCl3��=
=1mol/L��
�ʴ�Ϊ��1��
��4���������ʱΪ26.3g��Ϊ������������������������֮�ͣ���������������Ϊ26.3g-10.7g=15.6g���������������ʵ���Ϊ
=0.2mol������AlԪ���غ�n��AlCl3��=0.2mol��
��c��AlCl3��=
=2mol/L��
�ʴ�Ϊ��2��
��5������550mL NaOH��Һ��ʱ����Һ������ΪNaCl��NaAlO2�������������غ�n��NaCl��=3n��FeCl3��+3n��AlCl3��=3��0.1mol+3��0.2mol=0.9mol������AlԪ���غ�n��NaAlO2��=n��AlCl3��=0.2mol�������������غ�n��NaOH��=n��NaCl��+n��NaAlO2��=0.9mol+0.2mol=1.1mol����c��NaOH��=
=2mol/L��
�ʴ�Ϊ��2��
�ʴ�Ϊ��Fe3++3OH-=Fe��OH��3����Al3++4OH-=AlO2-+2H2O��
��2�����ճ���10.7gΪFe��OH��3�����ȷֽ�����Fe2O3��n[Fe��OH��3]=
| 10.7g |
| 107g/mol |
| 0.1mol |
| 2 |
�ʴ�Ϊ��8.0��
��3������FeԪ���غ�n��FeCl3��=n[Fe��OH��3]=0.1mol����c��FeCl3��=
| 0.1mol |
| 0.1L |
�ʴ�Ϊ��1��
��4���������ʱΪ26.3g��Ϊ������������������������֮�ͣ���������������Ϊ26.3g-10.7g=15.6g���������������ʵ���Ϊ
| 15.6g |
| 78g/mol |
��c��AlCl3��=
| 0.2mol |
| 0.1L |
�ʴ�Ϊ��2��
��5������550mL NaOH��Һ��ʱ����Һ������ΪNaCl��NaAlO2�������������غ�n��NaCl��=3n��FeCl3��+3n��AlCl3��=3��0.1mol+3��0.2mol=0.9mol������AlԪ���غ�n��NaAlO2��=n��AlCl3��=0.2mol�������������غ�n��NaOH��=n��NaCl��+n��NaAlO2��=0.9mol+0.2mol=1.1mol����c��NaOH��=
| 1.1mol |
| 0.55L |
�ʴ�Ϊ��2��
�����������Ի�ѧ��Ӧͼ����ʽ���������ӷ�Ӧ�ļ��㣬��Ŀ�Ѷ��еȣ���ȷ���η����ķ�ӦΪ���ؼ���ע�������غ㷨�ڻ�ѧ�����е�Ӧ�÷���������֪ʶ��϶ࡢ�ۺ��Խ�ǿ����ֿ�����ѧ���ķ�����������ѧ����������

��ϰ��ϵ�д�
 ����νӽ̲���ĩ���Ԥϰ�人������ϵ�д�
����νӽ̲���ĩ���Ԥϰ�人������ϵ�д� ������ҵ��ٳɳ����½������������ϵ�д�
������ҵ��ٳɳ����½������������ϵ�д�
�����Ŀ
���¶ȡ��ݻ���ͬ��3���ܱ������У����±�Ͷ�뷴Ӧ�������Ӧ��H2��g��+I2��g���T2HI��g����H=14.9kJ?mol-1���ں��¡����������£���÷�Ӧ�ﵽƽ��ʱ���й��������£�
����˵����ȷ���ǣ�������
| ���� | �� | �� | �� |
| ��Ӧ��Ͷ���� | 1mol H2 1mol I2 | 2mol HI | 4mol HI |
| HI��Ũ�� | c1 | c2 | c3 |
| ��Ӧ�������仯 | �ų�akg | ����bkg | ����ckg |
| ��Ӧ��ת���� | ��1 | ��2 | ��3 |
| A��2��1=��3 |
| B����1+��2=1 |
| C��c1=c2=c3 |
| D��a+b=14.9 |
��������Һ�У�ͨ������CO2���壬�����ܲ�����ɫ�������ǣ�������
| A������Na2CO3��Һ |
| B������ʯ��ˮ |
| C��CaCl2��Һ |
| D������ |
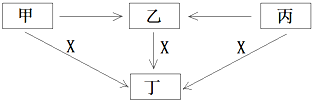

 ��R��R�������
��R��R���Ϊ����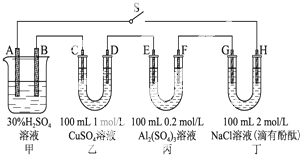 ��ͼʾ���ĸ������зֱ�ʢ�в�ͬ����Һ����A��B�⣬����缫��Ϊʯī�缫����ΪǦ���أ��乤��ԭ��Ϊ��Pb+PbO2+2H2SO4
��ͼʾ���ĸ������зֱ�ʢ�в�ͬ����Һ����A��B�⣬����缫��Ϊʯī�缫����ΪǦ���أ��乤��ԭ��Ϊ��Pb+PbO2+2H2SO4