��Ŀ����
��֪����25��ʱ H2O?H++OH-��KW=10-14 CH3COOH?H++CH3COO- Ka=1.8��10-5
��1��������ˮ���ƽ�ⳣ��Kh��KW��Ka�Ĺ�ϵ��Kh= ���������¶�ʱ��Kh�� ���������С�������䡱����
��2��0.9mol?L-1��������Һ�е�����������Ũ��Ϊ2.2��10-5mol?L-1����ij��Һ�к�Mg2+��Cd2+��Zn2+�������ӵ�Ũ�Ⱦ�Ϊ0.01mol?L-1�������м����������ƣ�ʹ��Ũ��Ϊ0.9mol?L-1���������ֽ��������� �����ɳ�����
��KSP[Mg��OH��2]=1.8��10-11��KSP[Zn��OH��2]=1.2��10-17��KSP[Cd��OH��2]=2.5��10-14��
��1��������ˮ���ƽ�ⳣ��Kh��KW��Ka�Ĺ�ϵ��Kh=
��2��0.9mol?L-1��������Һ�е�����������Ũ��Ϊ2.2��10-5mol?L-1����ij��Һ�к�Mg2+��Cd2+��Zn2+�������ӵ�Ũ�Ⱦ�Ϊ0.01mol?L-1�������м����������ƣ�ʹ��Ũ��Ϊ0.9mol?L-1���������ֽ���������
��KSP[Mg��OH��2]=1.8��10-11��KSP[Zn��OH��2]=1.2��10-17��KSP[Cd��OH��2]=2.5��10-14��
���㣺����ˮ���ԭ��,���ܵ���ʵ��ܽ�ƽ�⼰����ת���ı���
ר�⣺
��������1��CH3COONa��ˮ��ƽ�ⳣ��Kh=
�����KW��Ka�����Ƶ���ע�������ˮ��Ϊ���ȷ�Ӧ��
��2���������ӵ�Ũ�Ⱥ�����������Ũ�ȣ�������������ӵ����ӻ���Ȼ�����KSP[Mg��OH��2]=1.8��10-11��KSP[Zn��OH��2]=1.2��10-17��KSP[Cd��OH��2]=2.5��10-14�����ж��Ƿ����ɳ�����
| c(CH3COOH)?c(OH-) |
| c(CH3COO-) |
��2���������ӵ�Ũ�Ⱥ�����������Ũ�ȣ�������������ӵ����ӻ���Ȼ�����KSP[Mg��OH��2]=1.8��10-11��KSP[Zn��OH��2]=1.2��10-17��KSP[Cd��OH��2]=2.5��10-14�����ж��Ƿ����ɳ�����
���
�⣺��1��CH3COONa��ˮ��ƽ�ⳣ��Kh�T
=
=
�������ˮ��Ϊ���ȷ�Ӧ�������¶ȣ�Kh������
�ʴ�Ϊ��
������
��2����Һ������������Ũ���ǣ�[OH-]=2.2��10-5mol?L-1������[M2+][OH-]2=��2.2��10-5��2����0.01=4.36��10-12��mol?L-1��3�жϣ�
����4.36��10-12С��KSP[Mg��OH��2]=1.8��10-11��û��������þ�������ɣ�
����4.36��10-12����KSP[Zn��OH��2]=1.2��10-17����������п�������ɣ�
����4.36��10-12����KSP[Cd��OH��2]=2.5��10-14����Cd��OH��2�������ɣ�
�ʴ�Ϊ��Cd2+��Zn2+��
| c(CH3COOH)?c(OH-) |
| c(CH3COO-) |
| c(CH3COOH)?c(OH-)?c(H+) |
| c(CH3COO-)?c(H+) |
| Kw |
| Ka |
�ʴ�Ϊ��
| Kw |
| Ka |
��2����Һ������������Ũ���ǣ�[OH-]=2.2��10-5mol?L-1������[M2+][OH-]2=��2.2��10-5��2����0.01=4.36��10-12��mol?L-1��3�жϣ�
����4.36��10-12С��KSP[Mg��OH��2]=1.8��10-11��û��������þ�������ɣ�
����4.36��10-12����KSP[Zn��OH��2]=1.2��10-17����������п�������ɣ�
����4.36��10-12����KSP[Cd��OH��2]=2.5��10-14����Cd��OH��2�������ɣ�
�ʴ�Ϊ��Cd2+��Zn2+��
�����������ۺϿ���ѧ���ε�ˮ�⡢������ʵĵ���ļ���֪ʶ��Ϊ��Ƶ���㣬������ѧ���ķ��������������Ŀ��飬ע��֪ʶ�Ĺ��ɺ������ǽ���Ĺؼ����Ѷ��еȣ�

��ϰ��ϵ�д�
 �»����ܶ�Ա��ϵ�д�
�»����ܶ�Ա��ϵ�д� ����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
�����Ŀ
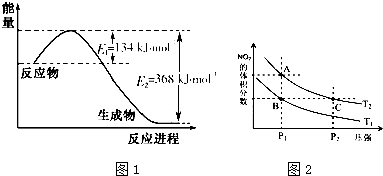
��2A+B?3C+4D��Ӧ�У���ʾ�÷�Ӧ���������ǣ�������
| A��v��A��=0.5 mol?L-1?min-1 |
| B��v��B��=0.01 mol?L-1?s-1 |
| C��v��C��=0.8 mol?L-1?min-1 |
| D��v��D��=1 mol?L-1?min-1 |
����������Ԫ��X��Y��Z��W��Ԫ�����ڱ��е����λ�������ʾ����֪X���������Y��������۴�����Ϊ�㣬����˵����ȷ���ǣ�������
| X | ||||
| Y | Z | W |
| A��ԭ�Ӱ뾶�Ĵ�С˳��r��Y����r��Z����r��W����r��X�� |
| B��Z������������ˮ�������Ա�W��ǿ |
| C��X�ļ���̬�⻯����ȶ��Ա�W���� |
| D��X�ֱ���Y��Z�γɵĻ������л�ѧ��������ͬ |
���й����л����������ȷ���ǣ�������
| A����ϩ�ͱ�����ʹ��ˮ��ɫ����ɫ��ԭ����ͬ |
| B������ʽΪC2H4O2������NaOH��Һ��Ӧ���л���һ�������� |
| C����֬����ά�ض����ܷ���ˮ�ⷴӦ�ĸ߷��ӻ����� |
| D�����顢�����Ҵ��������������Է���ȡ����Ӧ |
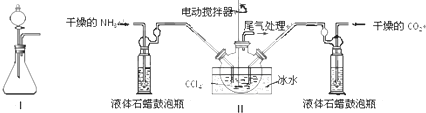
 ��ͼΪʵ����ijŨ�����Լ�ƿ��ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݣ��ش��������⣺
��ͼΪʵ����ijŨ�����Լ�ƿ��ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݣ��ش��������⣺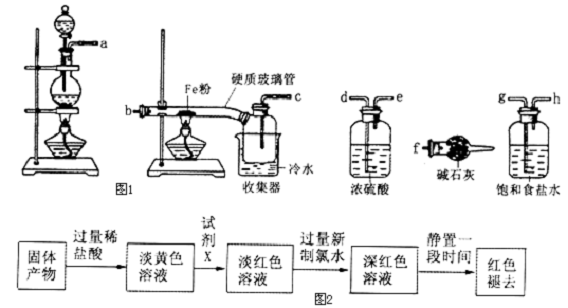
 ���ǵ����Ϻ����ḻ��һ��Ԫ�أ������仯������������������������Ҫ���ã�
���ǵ����Ϻ����ḻ��һ��Ԫ�أ������仯������������������������Ҫ���ã�