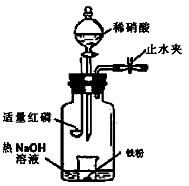
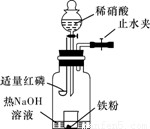
��Ŀ����
Ϊ̽������ϡ���ᷴӦ����Һ����Ԫ�ش�����ʽ��ij��ѧʵ����ȤС�����ʵ�����£�������裺
����1����Һ��ֻ��Fe2+��
����2��
����3��
���ʵ�鷽������֤���裺����������б���
| ʵ����� | Ԥ����������� |
| 1�� |
����Һ����죬�����1��ȷ�� ����Һ��죬�����2�����3��ȷ |
| 2����ȡ������Ӧ����Һ���Թ��У��μ�1-2��0.01mol/L KMnO4��Һ���� |
��1������֪����ϡ���ᷴӦ���ɱ�״����NO����2.24L����������Ӧ�вμӷ�Ӧ���������ʵ���Ϊ
��2��������1��ȷ����д������ϡ���ᷢ���÷�Ӧ�����ӷ���ʽ��
��3��ijͬѧ��Ӧ����Һ�еμ�����NaOH��Һ��δ���ְ�ɫ�����������ɴ˶϶���Һ��û��Fe2+��������˵��������������ɣ�
������������Ԫ�صļ�̬Ϊ+2��+3���ж���Ԫ�ش�����ʽΪ�����Ӻ��������ӣ��ݴ��ƶϿ��ܴ��ڵ������������ʹ�����軯����Һ���������ӣ�Ȼ�������������ӵĻ�ԭ��ʹ�ø��������Һ�����������ӣ�
��1�����ݷ�Ӧ����ʽ3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O��Fe+4H++NO3-�TFe3++NO��+2H2O��֪��������������ʵ���Ϊһ��������4����
��2������1����Һ��ֻ��Fe2+������ϡ���ᷴӦ��������������һ��������ˮ���ݴ�д����Ӧ�����ӷ���ʽ��
��3����������Ϊ���ɫ����������������Ϊ��ɫ���������ɫ�ܹ��ڸǰ�ɫ��
��1�����ݷ�Ӧ����ʽ3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O��Fe+4H++NO3-�TFe3++NO��+2H2O��֪��������������ʵ���Ϊһ��������4����
��2������1����Һ��ֻ��Fe2+������ϡ���ᷴӦ��������������һ��������ˮ���ݴ�д����Ӧ�����ӷ���ʽ��
��3����������Ϊ���ɫ����������������Ϊ��ɫ���������ɫ�ܹ��ڸǰ�ɫ��
����⣺����ϡ���ᷴӦ����Һ����Ԫ�صĴ�����ʽ����������������ӡ��������ӣ�������Һ�д������ּ��裺Һ��ֻ��Fe2+����Һ��ֻ��Fe3+����Һ�м���Fe2+������Fe3+��
������跽��Ϊ��ȡ������Ӧ����Һ���Թ��У��μ�1-2��KSCN��Һ��������Һ����죬�����1��ȷ������Һ��죬�����2�����3��ȷ��
��ȡ������Ӧ����Һ���Թ��У��μ�1-2��0.01mol/L KMnO4��Һ������KMnO4��Һ����ɫ�������2��ȷ����KMnO4��Һ��ɫ�������1�����3��ȷ��
�ʴ�Ϊ����Һ��ֻ��Fe3+����Һ�м���Fe2+������Fe3+��
�����������
��1����״����NO����2.24L�����ʵ���Ϊ0.1mol���ɷ���ʽ3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O��Fe+4H++NO3-�TFe3++NO��+2H2O��֪��������������ʵ���Ϊһ��������4�������������������ʵ���Ϊ0.4mol��
�ʴ�Ϊ��0.4mol��
��2������1��ֻ�������������ɣ���������ϡ���ᷴӦ�����ӷ���ʽΪ��3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O��
�ʴ�Ϊ��3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O��
��3�������������������ӷ�Ӧ���ɺ��ɫ������������������������������������Ӧ���ɵ��ǰ�ɫ���������������ں��ɫ�����������ܹ�����ɫ�ڸǣ���������˵������
�ʴ�Ϊ�����ԣ���ͬʱ��Fe3+����Fe��OH��3��ɫ�Ὣ��ɫ�ڸǣ�
������跽��Ϊ��ȡ������Ӧ����Һ���Թ��У��μ�1-2��KSCN��Һ��������Һ����죬�����1��ȷ������Һ��죬�����2�����3��ȷ��
��ȡ������Ӧ����Һ���Թ��У��μ�1-2��0.01mol/L KMnO4��Һ������KMnO4��Һ����ɫ�������2��ȷ����KMnO4��Һ��ɫ�������1�����3��ȷ��
�ʴ�Ϊ����Һ��ֻ��Fe3+����Һ�м���Fe2+������Fe3+��
| ʵ����� | Ԥ����������� |
| 1��ȡ������Ӧ����Һ���Թ��У��μ�1-2��KSCN��Һ���� | |
| ��KMnO4��Һ����ɫ�������2��ȷ�� ��KMnO4��Һ��ɫ�������1�����3��ȷ |
��1����״����NO����2.24L�����ʵ���Ϊ0.1mol���ɷ���ʽ3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O��Fe+4H++NO3-�TFe3++NO��+2H2O��֪��������������ʵ���Ϊһ��������4�������������������ʵ���Ϊ0.4mol��
�ʴ�Ϊ��0.4mol��
��2������1��ֻ�������������ɣ���������ϡ���ᷴӦ�����ӷ���ʽΪ��3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O��
�ʴ�Ϊ��3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O��
��3�������������������ӷ�Ӧ���ɺ��ɫ������������������������������������Ӧ���ɵ��ǰ�ɫ���������������ں��ɫ�����������ܹ�����ɫ�ڸǣ���������˵������
�ʴ�Ϊ�����ԣ���ͬʱ��Fe3+����Fe��OH��3��ɫ�Ὣ��ɫ�ڸǣ�
���������⿼���������ӡ��������ӵļ��飬��Ŀ�Ѷ��еȣ�ע����ȷ�������ӡ������Ӿ��еĻ�ѧ���ʼ����鷽��������������ѧ�����Ӧ����ѧ֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
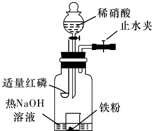 ijͬѧ��ͼʾװ�ý�������ϡ���ᷴӦ��ʵ�鲢������ص�ʵ��̽����
ijͬѧ��ͼʾװ�ý�������ϡ���ᷴӦ��ʵ�鲢������ص�ʵ��̽����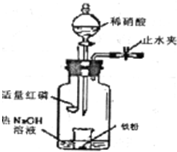 ijͬѧ����ͼװ�ý�������ϡ���ᷴӦ��ʵ��̽����ʵ�鲽��Ϊ��
ijͬѧ����ͼװ�ý�������ϡ���ᷴӦ��ʵ��̽����ʵ�鲽��Ϊ��