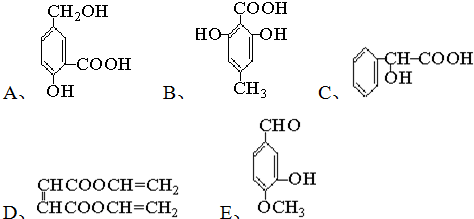
��Ŀ����
ʵ����������480mL 0.1mol/L Na2CO3��Һ����ݴ˻ش��������⣺
��1��ʵ��ʱ��Ӧ��������ƽ��ȡʮˮ̼���ƾ��� g��
��2��������Na2CO3��Һʱ���õIJ���������Ҫ����Ͳ���ձ����������� �� ��
��3����ʵ��������������������������������Һ��Ũ��ƫ�͵��� ��������ĸ��
A������ƿ�ڱڸ���ˮ���δ���ﴦ��
B�����ǽ�ϴ��Һ��������ƿ
C������ʱ���ӿ̶���
D��ҡ�Ⱥ���Һ����ڿ̶��ߣ���ˮ����Һ��ǡ����̶�������
��4��ȡ��õ�̼������Һ10��������ˮϡ�͵�50������ϡ�ͺ�̼������Һ�����ʵ���Ũ��Ϊ mol/L��
��1��ʵ��ʱ��Ӧ��������ƽ��ȡʮˮ̼���ƾ���
��2��������Na2CO3��Һʱ���õIJ���������Ҫ����Ͳ���ձ�����������
��3����ʵ��������������������������������Һ��Ũ��ƫ�͵���
A������ƿ�ڱڸ���ˮ���δ���ﴦ��
B�����ǽ�ϴ��Һ��������ƿ
C������ʱ���ӿ̶���
D��ҡ�Ⱥ���Һ����ڿ̶��ߣ���ˮ����Һ��ǡ����̶�������
��4��ȡ��õ�̼������Һ10��������ˮϡ�͵�50������ϡ�ͺ�̼������Һ�����ʵ���Ũ��Ϊ
���㣺����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺ʵ����
��������1������n=C��V��m=n��M��������ʮˮ̼���ƾ����������
��2������ʵ������IJ����Լ�ÿ��������Ҫ����ȷ����Ӧ����������
��3������c=
������������
��4������ϡ��ǰ�����ʵ����ʵ���������
��2������ʵ������IJ����Լ�ÿ��������Ҫ����ȷ����Ӧ����������
��3������c=
| n |
| V |
��4������ϡ��ǰ�����ʵ����ʵ���������
���
�⣺��1������480mL 0.1mol/L Na2CO3��Һ��Ӧѡ��500ml����ƿ����̼���ƾ�������ʵ���n=C��V=0.5L��0.1mol/L=0.05mol����Ҫ��ȡʮˮ̼���ƾ��������m=n��M=0.05mol��286g/mol=14.3g��
�ʴ�Ϊ��14.3��
��2������һ�����ʵ���Ũ����Һ���������У�������ƽ����Ͳ���ձ�����������500mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ����ͷ�ιܣ�500ml����ƿ��
��3����c=
��֪��
A������ƿ�ڱڸ���ˮ���δ���ﴦ������������Һ����������ʵ����ʵ�����û��Ӱ�죬��A����
B�����ǽ�ϴ��Һ��������ƿ�����ʵ��������٣�n��С��Ũ��ƫ�ͣ���B��ȷ��
C������ʱ���ӿ̶��ߣ�������Һ�����ƫС��Ũ��ƫ�ߣ���C����
D��ҡ�Ⱥ���Һ����ڿ̶��ߣ���ˮ����Һ��ǡ����̶������У�������Һ�����ƫ��Ũ��ƫС����D��ȷ��
��ѡ��BD��
��4��ϡ��ǰ�����ʵ����ʵ�������ϡ�ͺ���Һ��Ũ��ΪC����0.1mol/L��10ml=C��50ml�����C=0.02mol/L���ʴ�Ϊ��0.02��
�ʴ�Ϊ��14.3��
��2������һ�����ʵ���Ũ����Һ���������У�������ƽ����Ͳ���ձ�����������500mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ����ͷ�ιܣ�500ml����ƿ��
��3����c=
| n |
| V |
A������ƿ�ڱڸ���ˮ���δ���ﴦ������������Һ����������ʵ����ʵ�����û��Ӱ�죬��A����
B�����ǽ�ϴ��Һ��������ƿ�����ʵ��������٣�n��С��Ũ��ƫ�ͣ���B��ȷ��
C������ʱ���ӿ̶��ߣ�������Һ�����ƫС��Ũ��ƫ�ߣ���C����
D��ҡ�Ⱥ���Һ����ڿ̶��ߣ���ˮ����Һ��ǡ����̶������У�������Һ�����ƫ��Ũ��ƫС����D��ȷ��
��ѡ��BD��
��4��ϡ��ǰ�����ʵ����ʵ�������ϡ�ͺ���Һ��Ũ��ΪC����0.1mol/L��10ml=C��50ml�����C=0.02mol/L���ʴ�Ϊ��0.02��
���������⿼��������һ�����ʵ���Ũ�ȵ���Һ�ķ�������ȷԭ������Ϥ���ò����������ʹ���ǽ���ؼ���

��ϰ��ϵ�д�
 53���ò�ϵ�д�
53���ò�ϵ�д�
�����Ŀ
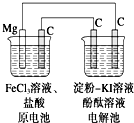 ij�о���ѧϰС��Ŀ���Ϊ��MgCԭ��ص�����KI��Һ���о��������о�װ����ͼ��ʾ�������й�˵������ȷ���ǣ�������
ij�о���ѧϰС��Ŀ���Ϊ��MgCԭ��ص�����KI��Һ���о��������о�װ����ͼ��ʾ�������й�˵������ȷ���ǣ�������| A��þΪ������������ԭ��Ӧ |
| B��ԭ�����̼����ӦʽΪFe3++e-�TFe2+ |
| C������������������ |
| D����������������� |
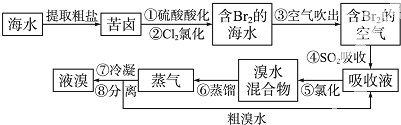
���������������Ҫ�����ǣ�1���������ֱ����С��2����������ʵ����Ķ��٣�3��������Ӽ��ƽ�����루4��������ӵ���Է���������������
| A����1����2�� |
| B����1����3�� |
| C����2����3�� |
| D����2����4�� |
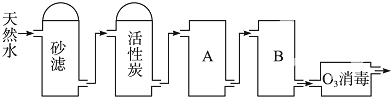
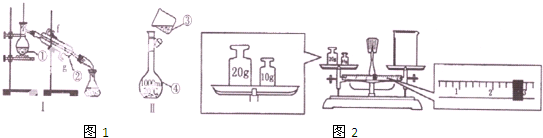
 ��1��������������������ͷ�չ����Ҫ���ʻ�����
��1��������������������ͷ�չ����Ҫ���ʻ�����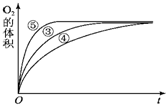 ijʵ��С����H2O2�ֽ�Ϊ�����о�Ũ�ȡ���������Һ����ԶԷ�Ӧ���ʵ�Ӱ�죮�ڳ����°������·������ʵ�飮
ijʵ��С����H2O2�ֽ�Ϊ�����о�Ũ�ȡ���������Һ����ԶԷ�Ӧ���ʵ�Ӱ�죮�ڳ����°������·������ʵ�飮