��Ŀ����
8����NA��������ӵ�������ֵ������˵����ȷ���ǣ���������1L pH=1��������Һ�к��е�H+��Ϊ0.2NA
�ڱ�״���£�2.24L��ϩ�ͱ�ϩ�Ļ�������к��е�̼ԭ������0.2NA
��7.2g CaO2�����������Ӻ�����������Ϊ0.3NA
�ܷ�Ӧ3H2��g��+N2��g��?2NH3��g����H=-92kJ/mol�ų�����9.2kJʱ��ת�Ƶ�����0.6NA
�ݳ��³�ѹ�£�1mol�������еĺ��������Ϊ4NA
���³�ѹ�£�1mol����-14CH3����������������Ϊ8NA��
| A�� | �٢� | B�� | �ܢ� | C�� | �ܢ� | D�� | �ۢ� |
���� ��pH=1�����ᣬ������Ũ��Ϊ0.1mol/L��
����ϩ�������ϩ���Ӻ���̼ԭ������ͬ��
��CaO2������������ΪO22-��
�ܷ�Ӧ3H2��g��+N2��g��?2NH3��g����H=-92 kJ/molת��6mol���ӣ�
�ݺ���Ϊ��ԭ�ӷ��ӣ�
��1��������8�����ӣ�
��� �⣺��pH=1�����ᣬ������Ũ��Ϊ0.1mol/L��1L pH=1��������Һ�к��е�H+��Ϊ0.1NA���ʴ���
����ϩ�������ϩ���Ӻ���̼ԭ������ͬ��֪��������������ֻ�����������ʵ�������ϩ���ϩ�������ʵ���δ֪������������̼ԭ�������ʴ���
��7.2g CaO2�������ʵ���Ϊ$\frac{7.2g}{72g/mol}$=0.1mol������0.1mol�����Ӻ�0.1mol���������ӣ���������������Ϊ0.2NA���ʴ���
�ܷ�Ӧ3H2��g��+N2��g��?2NH3��g����H=-92 kJ/molת��6mol���ӣ��ų�����9.2 kJʱ��ת�Ƶ���0.6 NA������ȷ��
�ݳ��³�ѹ�£�1mol�������еĺ��������ΪNA���ʴ���
��1��������8�����ӣ����³�ѹ�£�1mol����-14CH3����������������Ϊ8NA������ȷ��
��ѡ��C��
���� ���⿼���˰���٤���������йؼ��㣬���չ�ʽ�����á����ʵ�״̬�����ʵĽṹ�ǹؼ����ѶȲ���

| A�� | 2 g��������ԭ����ΪNA | |
| B�� | ���³�ѹ�£�1.06gNa2CO3���е�Na+������Ϊ0.02NA | |
| C�� | �ڳ��³�ѹ�£�11.2LN2����ԭ����ΪNA | |
| D�� | 2.7g���������������ᷴӦ��ʧȥ�ĵ�����ĿΪ0.2NA |
 ��˵������ȷ���ǣ�������
��˵������ȷ���ǣ�������| A�� | ������ˮ�����ӳɷ�Ӧ | B�� | ���������е�̼ԭ����ͬһƽ���� | ||
| C�� | ����̼�����Ʒ�����Ӧ | D�� | �ܷ���ȡ����������Ӧ |
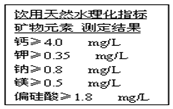
| A�� | ԭ�� | B�� | ���� | C�� | ���� | D�� | Ԫ�� |
| A�� | �����ĵ���ʽ��Cl��Cl | |
| B�� | ��ϩ�Ľṹ��ʽCH2CH2 | |
| C�� | ��ԭ�ӵĽṹʾ��ͼ��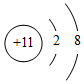 | |
| D�� | ���������ĵ��뷽��ʽFeSO4�TFe2++SO42- |
| A�� | ̼������Һ��ϡ���� | B�� | ������Һ������������Һ | ||
| C�� | ̼��������Һ��ϡ���� | D�� | ƫ��������Һ��ϡ���� |
| A�� | ������ֳ��������Ժ����� | B�� | ��ԭ����������֮��Ϊ1��4 | ||
| C�� | ÿ��������ӵõ�2������ | D�� | ����ͭ�ǻ�ԭ���� |
| A�� | 2Na+2H2O=2NaOH+H2������Ӧ | |
| B�� | ClO-+H2O?HClO+OH-����Ӧ | |
| C�� | ����ǿ��˳��OH-��ClO-��CH3COO- | |
| D�� | HCO3-���������ǼNH3�Ȳ������ֲ��Ǽ� |