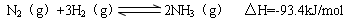��Ŀ����
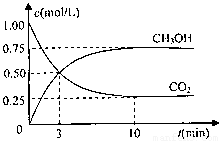
��1��Ϊ̽����CO2������ȼ�ϼ״��ķ�Ӧԭ�����ֽ�������ʵ�飺�����Ϊ1L���ܱ������У�����1mol
CO2��3molH2��һ�������·�����Ӧ��CO2(g)+3H2(g)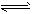 CH3OH(g)+H2O(g) ��H=-49.0kJ/mol ���CO2��
CH3OH(g)+H2O(g) ��H=-49.0kJ/mol ���CO2��
CH3OH��g����Ũ����ʱ��仯��ͼ����ش�
CO2��3molH2��һ�������·�����Ӧ��CO2(g)+3H2(g)
 CH3OH(g)+H2O(g) ��H=-49.0kJ/mol ���CO2��
CH3OH(g)+H2O(g) ��H=-49.0kJ/mol ���CO2��CH3OH��g����Ũ����ʱ��仯��ͼ����ش�
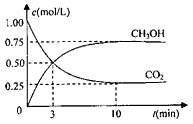
�� �ӷ�Ӧ��ʼ��ƽ�⣬������ƽ����Ӧ���ʣ�v(H2)=________mol/��L��min����
�������¶ȣ��÷�Ӧ��ƽ�ⳣ������ֵ��________�����������С�����䡱����
�����д�ʩ����ʹƽ��������n��CH3OH��/n��CO2���������________
A���������
B������He��g����ʹ��ϵѹǿ����
C����H2O��g������ϵ�з���
D���ٳ���3molH2
��2���ϳɰ����Ȼ�ѧ����ʽΪ��N2(g)+3H2(g)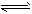 2NH3(g) ��H=-92 kJ��mol-1��
2NH3(g) ��H=-92 kJ��mol-1��
��ij�¶��£�����10mol N2��30mol H2�������Ϊ10L���ܱպϳ����ڣ���Ӧ�ﵽƽ��״̬ʱ�����ƽ���������а����������Ϊ1/5������¶��·�Ӧ��K=___________�����÷�����ʾ����
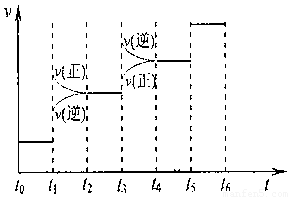
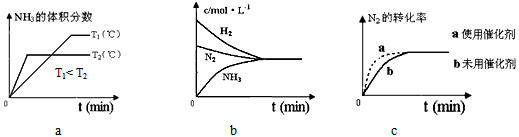
�ڶ��ںϳɰ���Ӧ���ԣ������й�ͼ��һ����ȷ���ǣ�����ţ�_______________
�������¶ȣ��÷�Ӧ��ƽ�ⳣ������ֵ��________�����������С�����䡱����
�����д�ʩ����ʹƽ��������n��CH3OH��/n��CO2���������________
A���������
B������He��g����ʹ��ϵѹǿ����
C����H2O��g������ϵ�з���
D���ٳ���3molH2
��2���ϳɰ����Ȼ�ѧ����ʽΪ��N2(g)+3H2(g)
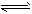 2NH3(g) ��H=-92 kJ��mol-1��
2NH3(g) ��H=-92 kJ��mol-1����ij�¶��£�����10mol N2��30mol H2�������Ϊ10L���ܱպϳ����ڣ���Ӧ�ﵽƽ��״̬ʱ�����ƽ���������а����������Ϊ1/5������¶��·�Ӧ��K=___________�����÷�����ʾ����
�ڶ��ںϳɰ���Ӧ���ԣ������й�ͼ��һ����ȷ���ǣ�����ţ�_______________
��1����0.225���ڼ�С����CD
��2����1/12 L2��mol-2����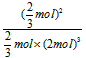 ������ac
������ac
��2����1/12 L2��mol-2����
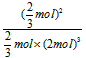 ������ac
������ac
��ϰ��ϵ�д�
�����Ŀ
 ��2013?�麣ģ�⣩�״���һ������ȼ�ϣ�������ȼ�ϵ�أ�
��2013?�麣ģ�⣩�״���һ������ȼ�ϣ�������ȼ�ϵ�أ�
 CH3OH(g)��H2O(g) ��H����49.0kJ/mol
CH3OH(g)��H2O(g) ��H����49.0kJ/mol
 CO2��g��+3H2��g�� CH3OH��g��+H2O��g����H=-49.0kJ/mol
CO2��g��+3H2��g�� CH3OH��g��+H2O��g����H=-49.0kJ/mol