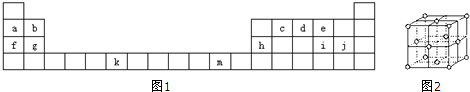
��Ŀ����
A��B��C��D��E�����ڱ���ǰ�����ڵ�Ԫ�أ����й����ʻ�ṹ��Ϣ�����
��1��Ԫ��Y��C��һ����ͬ����Ԫ�أ��Ƚ�B��YԪ�صĵ�һ������I1��B�� I1��Y��0
��2��EԪ��ԭ�ӵĺ�������Ų�ʽΪ ��
��3����X��ˮ��Һ�� ������ԡ��������ԡ������ԡ�����BԪ�����{�ۺ�����һ����DԪ����ۺ���������� ���ǿ������������
��4��C���ʷ����ЦҼ��ͦм��ĸ�����Ϊ ��C���⻯����ͬ��Ԫ�ص��⻯���зе���ַ�������ԭ���� ��
��5���ø�����������Һ̬H2Aʱ��һ��H2A�������ͷų�һ�����ӣ�ͬʱ����һ�־��н�ǿ�����Ե������ӣ���д���������ӵĵ���ʽ�� ��д����������������⻯���ˮ��Һ��Ӧ�����ӷ���ʽ�� ��
| Ԫ�� | �й����ʻ�ṹ��Ϣ |
| A | �����۵�AԪ�ص��⻯����ͨ��״������һ��Һ�壬����A����������Ϊ88.9% |
| B | Bԭ�ӵõ�һ�����Ӻ�3p���ȫ���� |
| C | Cԭ�ӵ�p����������������̬�⻯������������������ˮ���ﷴӦ����һ�ֳ�������X |
| D | DԪ�ص�����ϼ�����ͻ��ϼ۵Ĵ�����Ϊ�㣬�����������Ϊ���Ӿ��� |
| E | EԪ�صĺ˵��������Aԭ�Ӻ�BԪ���⻯��ĺ˵����֮�� |
��2��EԪ��ԭ�ӵĺ�������Ų�ʽΪ
��3����X��ˮ��Һ��
��4��C���ʷ����ЦҼ��ͦм��ĸ�����Ϊ
��5���ø�����������Һ̬H2Aʱ��һ��H2A�������ͷų�һ�����ӣ�ͬʱ����һ�־��н�ǿ�����Ե������ӣ���д���������ӵĵ���ʽ��
���㣺λ�ýṹ���ʵ����ϵӦ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
�����������۵�AԪ�ص��⻯����ͨ��״������һ��Һ�壬����A����������Ϊ88.9%����AΪ��Ԫ�أ�Bԭ�ӵõ�һ�����Ӻ�3p���ȫ��������BΪ��Ԫ�أ�Cԭ�ӵ�p����������������̬�⻯������������������ˮ���ﷴӦ����һ�ֳ�������X����CΪ��Ԫ�أ�XΪ����泥�DԪ�ص�����ϼ�����ͻ��ϼ۵Ĵ�����Ϊ�㣬�����������Ϊ���Ӿ��壬��Ϊǰ������Ԫ�أ�����DΪ̼Ԫ�أ�EԪ�صĺ˵��������Aԭ�Ӻˡ�BԪ���⻯��ĺ˵����֮�ͣ���EΪ��Ԫ�أ���A��E�ֱ�ΪO��Cl��N��C��Fe���ݴ˴��⣻
���
�⣺�����۵�AԪ�ص��⻯����ͨ��״������һ��Һ�壬����A����������Ϊ88.9%����AΪ��Ԫ�أ�Bԭ�ӵõ�һ�����Ӻ�3p���ȫ��������BΪ��Ԫ�أ�Cԭ�ӵ�p����������������̬�⻯������������������ˮ���ﷴӦ����һ�ֳ�������X����CΪ��Ԫ�أ�XΪ����泥�DԪ�ص�����ϼ�����ͻ��ϼ۵Ĵ�����Ϊ�㣬�����������Ϊ���Ӿ��壬��Ϊǰ������Ԫ�أ�����DΪ̼Ԫ�أ�EԪ�صĺ˵��������Aԭ�Ӻˡ�BԪ���⻯��ĺ˵����֮�ͣ���EΪ��Ԫ�أ���A��E�ֱ�ΪO��Cl��N��C��Fe��
��1��Ԫ��Y��C��һ����ͬ����Ԫ�أ���YΪPԪ�أ���BΪClԪ�أ�����Ԫ�������ɣ�P�ĵ�һ������С��Cl�ĵ�һ�����ܣ��ʴ�Ϊ������
��2��FeΪ26��Ԫ�أ����������Ų�ʽΪ1s22s22p63s23p63d 64s2���ʴ�Ϊ��1s22s22p63s23p63d 64s2��
��3����XΪNH4NO3����ǿ�������Σ���ˮ��������ԣ�����������ǿ��̼�ᣬ�ʴ�Ϊ�����ԣ� ǿ��
��4��C����ΪN2��N2���Ӻ������������ļ��ͦм��ĸ�����Ϊ1��2�����ڰ����Ӽ�����������ʹ��е���ַ������ʴ�Ϊ��1��2�� NH3���Ӽ���������
��5��H2AΪH2O��H2O�ͷų�һ����������H2O+������Ԫ��Ϊ-1�ۣ������ʽΪ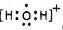 ������ǿ�����ԣ���������H2S�������ӷ���ʽΪ 2H2O++H2S=S��+2H2O+2H+���ʴ�Ϊ��
������ǿ�����ԣ���������H2S�������ӷ���ʽΪ 2H2O++H2S=S��+2H2O+2H+���ʴ�Ϊ��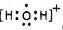 ��2H2O++H2S=S��+2H2O+2H+��
��2H2O++H2S=S��+2H2O+2H+��
��1��Ԫ��Y��C��һ����ͬ����Ԫ�أ���YΪPԪ�أ���BΪClԪ�أ�����Ԫ�������ɣ�P�ĵ�һ������С��Cl�ĵ�һ�����ܣ��ʴ�Ϊ������
��2��FeΪ26��Ԫ�أ����������Ų�ʽΪ1s22s22p63s23p63d 64s2���ʴ�Ϊ��1s22s22p63s23p63d 64s2��
��3����XΪNH4NO3����ǿ�������Σ���ˮ��������ԣ�����������ǿ��̼�ᣬ�ʴ�Ϊ�����ԣ� ǿ��
��4��C����ΪN2��N2���Ӻ������������ļ��ͦм��ĸ�����Ϊ1��2�����ڰ����Ӽ�����������ʹ��е���ַ������ʴ�Ϊ��1��2�� NH3���Ӽ���������
��5��H2AΪH2O��H2O�ͷų�һ����������H2O+������Ԫ��Ϊ-1�ۣ������ʽΪ
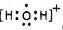 ������ǿ�����ԣ���������H2S�������ӷ���ʽΪ 2H2O++H2S=S��+2H2O+2H+���ʴ�Ϊ��
������ǿ�����ԣ���������H2S�������ӷ���ʽΪ 2H2O++H2S=S��+2H2O+2H+���ʴ�Ϊ��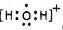 ��2H2O++H2S=S��+2H2O+2H+��
��2H2O++H2S=S��+2H2O+2H+��
������������Ҫ�����˵�һ�����ܡ������Ų�ʽ����ѧ��������ʽ�����ӷ�Ӧ��֪ʶ�㣬�е��Ѷȣ�����ؼ�Ҫ����������е���Ϣ��

��ϰ��ϵ�д�
�����Ŀ
һ���¶��£����ڷ�ӦN2+O2?2NO���ܱ������н��У����д�ʩ�ܼӿ췴Ӧ���ʵ��ǣ�������
| A���ı��¶� |
| B�����ݣ�����N2 |
| C�����ݣ�����He�� |
| D����ѹ������He�� |
pH=1������һԪ��HX��HY��Һ���ֱ�ȡ50mL����������þ�ۣ���ַ�Ӧ���ռ���H2����ֱ�ΪV��HX����V��HY������V��HX����V��HY����������˵����ȷ���ǣ�������
| A��HX������ǿ�� |
| B��HX�����Ա�HY�������� |
| C�������Ũ�ȴ�С[HX]��[HY] |
| D��pH=1������һԪ���ϡ��100����ϡ�ͺ�����Һ��pH��Ϊ3 |
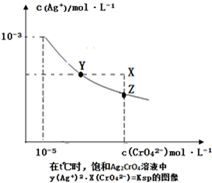 ��֪t��ʱAgCl��Ksp=4��10-l0����t��ʱ��Ag2CrO4��ˮ�еij����ܽ�ƽ��������ͼ��ʾ������˵��������ǣ�������
��֪t��ʱAgCl��Ksp=4��10-l0����t��ʱ��Ag2CrO4��ˮ�еij����ܽ�ƽ��������ͼ��ʾ������˵��������ǣ�������| A����t��ʱ��Ag2CrO4��KspΪ1��10-11 |
| B���ڱ�����Һ�м���K2CrO4��ʹ��Һ��Y�㵽Z�� |
| C����t��ʱ��Ag2CrO4��s��+2Cl-��aq��?2AgCl��s��+CrO42-��aq��ƽ�ⳣ��K=6.25��107 |
| D����t��ʱ����0.001mol/L AgNO3��Һ�ζ�20mL 0.001mol/L KCl��0.001mol/L��K2CrO4�Ļ����Һ��CrO42-�ȳ��� |
��1L����0.4molCuSO4��0.1molNaCl��ˮ��Һ�ö��Ե缫��⣬���һ��ʱ�����һ���缫�ϵõ�0.25molCu����һ���缫�ϲ������壨�����������ǣ�������
| A��4.48L |
| B��3.36L |
| C��6.72L |
| D��13.44L |
���и��������У�ֻ��һ���Լ����ܼ�����ǣ�������
| A��������ϩ������ |
| B����ϩ����Ȳ������ |
| C����ϩ���������Ȼ�̼ |
| D�����顢��ϩ������ |