��Ŀ����
1�� ��A��һ����Ҫ�Ļ���ԭ�ϣ���֪A�ڱ�״���µ��ܶ�Ϊ1.16g•L-1��B�ɷ���������Ӧ������֮���ת����ϵ��ͼ����ش�
��A��һ����Ҫ�Ļ���ԭ�ϣ���֪A�ڱ�״���µ��ܶ�Ϊ1.16g•L-1��B�ɷ���������Ӧ������֮���ת����ϵ��ͼ����ش���1���л���A�к��еĹ�����������̼̼������
��2����Ӧ�ܵ�ԭ�������ʴ�100%���÷�Ӧ�Ļ�ѧ����ʽ��2CH3CHO$\stackrel{����}{��}$CH3COOCH2CH3��
��3�������������л���D��Ӧ���õIJ�������ˮ�ñ�����Һ���μ�2�η�̪��Һ�����Ⱥ�ˮ��Һ�Ժ�ɫ����ԭ����CH3COO-+H2O��CH3COOH+OH-�������ӷ���ʽ��ʾ��
��4������˵����ȷ����BD
A���л���C��һ�������ºϳ���������Ҳ��ʵ��ԭ��������100%
B����Ӧ�ڡ��ۡ��ݵ���������ͬ
C����������������ˮ��Ϊ��ɫҺ�壬���÷�Һ©�����������������ˮ�Ļ����
D����ͬ��������������������������Һ��ˮ�����ϡ�����и���ȫ��
���� A�ڱ�״���µ��ܶ�Ϊ1.16g•L-1����A��Ħ������Ϊ1.16g/L��22.4L/mol=26g/mol��B�ɷ���������Ӧ����BΪȩ��������ͼ��֪��A��ˮ��Ӧ����B��B��������D��B���ԭ����C��C��D������Ӧ����������������AΪCH��CH��BΪCH3CHO��CΪCH3CH2OH��DΪCH3COOH���Դ������
��� �⣺��1��������������֪��AΪCH��CH������������Ϊ̼̼�������ʴ�Ϊ��̼̼������
��2��������������֪��BΪCH3CHO����Ӧ�ܵ�ԭ�������ʴ�100%����CH3CHO�������Ӽ�ӳɷ�Ӧ����CH3COOCH2CH3���÷�Ӧ�Ļ�ѧ����ʽ��2CH3CHO$\stackrel{����}{��}$CH3COOCH2CH3���ʴ�Ϊ��2CH3CHO$\stackrel{����}{��}$CH3COOCH2CH3��
��3��������������֪��DΪCH3COOH�������������л���D��CH3COOH��Ӧ����CH3COONa��CH3COONa����ˮ��CH3COO-����ˮ�ⷴӦ��������������������Եμ�2�η�̪��Һ��ˮ��Һ�Ժ�ɫ����Ӧ���ӷ���ʽΪCH3COO-+H2O��CH3COOH+OH-���ʴ�Ϊ��CH3COO-+H2O��CH3COOH+OH-��
��4��A��CΪCH3CH2OH�����ᷢ��������Ӧ��������������ˮ��������һ�������ºϳ�������������ʵ��ԭ��������100%����A����
B����Ӧ��Ϊ��ԭ��Ӧ����Ϊ������Ӧ����Ϊ������Ӧ��������������ͬ����B��ȷ��
C������������ˮ�����ܣ����Կ����÷�Һ©���������������ˮ�Ļ�����C����
D����������������������Һ��ˮ�����������ƺ��Ҵ��������棬����ϡ�����и���ȫ����D��ȷ��
�ʴ�Ϊ��BD��
���� ���⿼���л�����ƶϣ�Ϊ��Ƶ���㣬������Ϣ�������ŵı仯Ϊ���Ĺؼ������ط������ƶ������Ŀ��飬ע��AΪ��Ȳ����Ŀ�ѶȲ���

| A�� | ���³�ѹ�£�8gCH4�к��е���ԭ����Ϊ2NA | |
| B�� | ����NA����ԭ�ӵĺ����ڱ�״���µ����ԼΪ22.4L | |
| C�� | ���³�ѹ�£�48�����ͳ�����O3���������������ԭ������Ϊ3NA | |
| D�� | ���ʵ���Ũ��Ϊ0.5 mol•L-1��MgCl2��Һ�У�����Cl-����Ϊ1NA |
| A�� | C+H2O$\frac{\underline{\;\;��\;\;}}{\;}$CO+H2 | B�� | H2+CuO$\frac{\underline{\;\;��\;\;}}{\;}$Cu+H2O | ||
| C�� | Cl2+H2O?HCl+HC1O | D�� | 2F2+2H2O=4HF+O2 |
| A�� | һ�������£����������������ǡ������ʶ�����ˮ����ˮ�ⷴӦ | |
| B�� | CCl4���ӵı���ģ��Ϊ  ��ռ乹��Ϊ�������� ��ռ乹��Ϊ�������� | |
| C�� | ȫ��������  ���ɻ������� ���ɻ�������  ��CO2�����Ƶ� ��CO2�����Ƶ� | |
| D�� | һ�������£���ȫȼ��14g������������Ϊa����ϩ����ȩ������壬����ˮ������Ϊ18��1-a��g |
| A�� | NaHCO3 | B�� | AgBr | C�� | AgCl | D�� | HC1O |
| A�� | SO2��SiO2��P2O5 | B�� | PCl3��CO2��H2SO4 | C�� | SiC��H2O��NH3 | D�� | HF��CO2��Si |
| A�� | 1 L 0.2 mol•L-1 CH3COONa��Һ�к���0.2NA��CH3COO- | |
| B�� | ��״���£�22.4 L CO2����NA��̼��˫�� | |
| C�� | һ���¶��£���2 mol SO2��1 mol O2����װ�д����������з�����Ӧ��ת��4NA������ | |
| D�� | 1 mol�ǻ���-OH���к�9NA������ |
| A�� | CH2=CH-CH=CH2 | B�� | HOCH2-CH2OH | C�� | 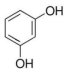 | D�� | HCOOH |