��Ŀ����
10��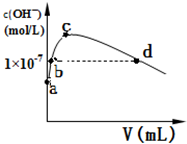 CO2��CH4��һ�������ºϳ����CO2+CH4$\stackrel{TiO_{2}/Cu_{2}Al_{2}O_{4}}{��}$CH3COOH�����������գ�
CO2��CH4��һ�������ºϳ����CO2+CH4$\stackrel{TiO_{2}/Cu_{2}Al_{2}O_{4}}{��}$CH3COOH�����������գ���1����д��̼ԭ��������Ӳ�Ĺ����ʾʽ
 ��
�����ѣ�Ti����22��Ԫ�أ�����bd��ѡ���ţ���
a������Ԫ�� b������Ԫ�� c��������Ԫ�� d��������Ԫ��
��2��CS2���ӵĵ���ʽΪ
 �����۷е��CO2�ߣ�ԭ���Ƕ��߶�Ϊ���Ӿ��壬��Է�������Խ���Ӽ�������Խ��
�����۷е��CO2�ߣ�ԭ���Ƕ��߶�Ϊ���Ӿ��壬��Է�������Խ���Ӽ�������Խ�� �����£���1L pH=10�� NaOH��Һ�г���ͨ��CO2��ͨ��CO2�������V������Һ��ˮ���������OH-����Ũ�ȣ�c���Ĺ�ϵ��ͼ��ʾ��
��3��c����Һ������Ũ���ɴ���С�Ĺ�ϵ�ǣ�c��Na+����c��CO32-����c��HCO3-����
��4��a����Һ����ˮ���������c��H+��=1��10-10mol/L��b����Һ��c��H+��С��1��10-7mol/L����д�����ڡ��������ڡ���С�ڡ�����
��5����ʹ0.1mol/L������Һ�ĵ���̶��Լ�pH���������ad����ѡ����ţ�
a����ˮϡ�� b���������������ƹ��� c��ͨ�Ȼ��� d���������������ƹ��壮
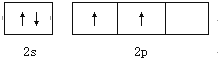
���� ��1����Cԭ�ӹ�2�����Ӳ㣬��������Ϊ2s��2p���ӣ�
���ѣ�Ti����22��Ԫ�أ�λ�ڵ������ڢ�B�壻
��2��CS2�ĵ���ʽ������CO2�����߶�Ϊ���Ӿ��壬��Է�������Խ���۵�Խ�ߣ�
��3����ˮ�������OH-����Ũ�����ʱ��˵����ʱ����Һ��̼������Һ����ˮ�ĵ�����ٽ����ã�
��4��ˮ������������������������ӵ�Ũ��ʼ����ȣ�a��û��ͨ������̼���������pH=10�� NaOH��Һ�����ˮ�����ӳ���������⣻
b����ǿ����Һ��ˮ�ĵ������������ã�������Һ�е�c��H+����1��10-7mol/L��
��5������̶�������ƽ��Ӧ�����ƶ�����pH���������Լ������Դ������
��� �⣺��1����Cԭ�ӹ�2�����Ӳ㣬��������Ϊ2s��2p���ӣ�������Ӳ�Ĺ����ʾʽΪ ��
��
�ʴ�Ϊ�� ��
��
���ѣ�Ti����22��Ԫ�أ�λ�ڵ������ڢ�B�壬Ϊ�����ڸ���Ԫ�أ��ʴ�Ϊ��bd��
��2��CS2�ĵ���ʽ������CO2������ʽΪ �����߶�Ϊ���Ӿ��壬��Է�������Խ���Ӽ�������Խ�����۵�Խ�ߣ�
�����߶�Ϊ���Ӿ��壬��Է�������Խ���Ӽ�������Խ�����۵�Խ�ߣ�
�ʴ�Ϊ�� �����߶�Ϊ���Ӿ��壬��Է�������Խ���Ӽ�������Խ��
�����߶�Ϊ���Ӿ��壬��Է�������Խ���Ӽ�������Խ��
��3����ˮ�������OH-����Ũ�����ʱ��˵����ʱ����Һ��̼������Һ����ˮ�ĵ�����ٽ����ã�����������ҺŨ�ȴ�СΪc��Na+����c��CO32-����c��HCO3-����
�ʴ�Ϊ��c��Na+����c��CO32-����c��HCO3-����
��4��ˮ������������������������ӵ�Ũ��ʼ����ȣ�a��û��ͨ������̼���������pH=10�� NaOH��Һ����c��H+��=1��10-10mol/L������������Դ��ˮ�ĵ��룬����ˮ�������c��H+��=1��10-10mol/L��b����ǿ����Һ��ˮ�ĵ������������ã�������Һ�е�c��H+����1��10-7mol/L��
�ʴ�Ϊ��1��10-10mol/L��С�ڣ�
��5��a����ˮϡ�ͣ��ٽ����룬��������Ũ�ȼ�С��pH����ѡ��
b���������������ƹ��壬���Ƶ��룬����̶ȼ�С���ʲ�ѡ��
c��ͨ�Ȼ��⣬������ǿ��pH��С����C��ѡ��
d���������������ƹ��壬�ٽ����룬��������Ũ�ȼ�С��pH����ѡ��
�ʴ�Ϊ��ad��
���� ���⿼����ۺϣ��漰����ϼ��㡢Ԫ�����ڱ���Ӧ�ü������Ų��ȣ�Ϊ��Ƶ���㣬���շ�Ӧԭ����ͼ�������Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬��Ŀ�ѶȲ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | 5min��ƽ����Ӧ����vA=0.15mol•L-1•min-1 | |
| B�� | ��ͬʱ��ԭƽ����ϵ����һ���¶Ⱥ�����һ��ѹǿʱ����ƽ���C�����ʵ�������Ϊ60%����÷�Ӧ������ӦΪ���ȷ�Ӧ | |
| C�� | ���������������䣬����ʼʱ�������м���0.5molA��0.5molB��1.5molC����Ӧ�����淴Ӧ������� | |
| D�� | t�棬��2L�ܱա������������Գ���1molA��1molB����Ӧ�ﵽƽ��״̬ʱ��C�����ʵ�������С��60% |
 ��������ȷ���ǣ�������
��������ȷ���ǣ�������| A�� | 2��3��3-�������� | B�� | 2-����-3-�һ����� | ||
| C�� | 2��2-����-3-�һ����� | D�� | 3��4��4-�������� |
| A�� | pH��7����Һһ��������Һ | |
| B�� | �����£���pH=7�Ĵ����ƺʹ�������Һ�У�c��CH3COO-����c��Naʮ�� | |
| C�� | ��pH=5���Ȼ��ƺ�ϡ����Ļ����Һ�У�c��Na+��=c��C1-�� | |
| D�� | 0.1 mol•L-1��������Һ�У�c��OH-��=c��H+��+c��HS-��+c��H2S�� |
| A�� | ���������麬�����еĵ�Ԫ�� | |
| B�� | �Ŵ����մɡ�ש�ߡ��ִ��IJ�����ˮ��ȣ����ǹ����β�Ʒ | |
| C�� | ���������ڼ��������µ�ˮ�ⷴӦ��Ϊ������Ӧ | |
| D�� | ����ᵼ��ijЩ��Ȫ�г�������ζ���������������ˮ�帻Ӫ���� |
| A�� | NA���������ӵ����ԼΪ11.2L | |
| B�� | 1.8g��NH4+�����к��еĵ�����Ϊ10NA | |
| C�� | ��״���£�2.24 L�������������������ɵĻ�����壬������ԭ����ԼΪ0.2NA | |
| D�� | 7.8g�������ƹ����к��е����������ӵ�����ԼΪ0.4 NA |
| A�� | �ܢۢ٢� | B�� | �ڢۢݢ� | C�� | �ܢݢۢ� | D�� | �٢ڢۢ� |
 ��ij�ܱ������г���H2S��O2��������Ӧ��2H2S��g��+O2��g��?S2��g��+2H2O��g����H��0���ﵽƽ��������ı������x��ֵ�����´ﵽƽ���������y��x�仯���Ʒ���ͼ����ǣ�������
��ij�ܱ������г���H2S��O2��������Ӧ��2H2S��g��+O2��g��?S2��g��+2H2O��g����H��0���ﵽƽ��������ı������x��ֵ�����´ﵽƽ���������y��x�仯���Ʒ���ͼ����ǣ�������| ѡ�� | x | y |
| A | �¶� | �������ƽ����Է������� |
| B | ѹǿ | S2����������� |
| C | ������� | H2SŨ�� |
| D | O2Ũ�� | ƽ�������淴Ӧ���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
 ��0.1mol��þ�������������100mL 2mol•L-1��H2SO4��Һ�У�Ȼ���ٵμ�1mol•L-1��NaOH��Һ����ش�
��0.1mol��þ�������������100mL 2mol•L-1��H2SO4��Һ�У�Ȼ���ٵμ�1mol•L-1��NaOH��Һ����ش�