��Ŀ����
�ս�����еĺ�ѧͬѧ��ѧϰ�µĻ�ѧ֪ʶ�����ϳ��к��е�֪ʶ̽��CO2�����ʡ�������ͼ��װ��������������ʵ�飺
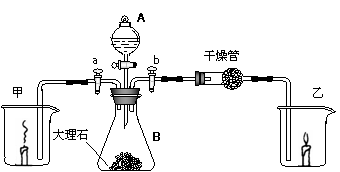
I���ر�b���ţ���a���ţ�����A�����Ļ����������е�Һ����B�еĴ���ʯ��Ӧ������B���д������ݲ�����ͬʱ���ձ���ȼ�յ�������Ϩ��
II���ر�a���ţ���b���ţ���B�в��������徭������ܲ������еĹ��巴Ӧ�����ɵ�����ͨ�����ձ����ɷ������ձ��е�����Խ��Խ����
�ʣ���1��A������������������ ����B�����������ǣ������������� ��
��2��A��װ��Һ������������ ��B�в�����������ӷ�Ӧ����ʽΪ���������������������������� ������������������������ ��
��3���ں�ѧͬѧ�ĵ�һ�������У�����Ϩ���ԭ�������� ������������������
��4���ڸ������ʢװ�������������������� ���ѧʽ����Ϊʲô���ձ��е������Ϩ�𣬶����ձ��е�����ȴԽ��Խ���� �����������������������������������û�ѧ��Ӧ����ʽ��ʾ��
��1��A������ ��Һ©�� ��B������ ��ƿ ��
��2�� ϡ���� �� 2H+ + CaCO3= CO2��+ H2O+Ca2+ ��
��3�� CO2��֧��ȼ�գ�����ȼ�����ܶȴ��ڿ��� ��
��4�� Na2O2 �� 2Na2O2 + 2CO2 = 2Na2CO3 + O2 ��

 ��У����ϵ�д�
��У����ϵ�д�