��Ŀ����
1����֪���봼��Ӧ���Ƶ���������ѧС���Ա����ᣨ ��ԭ�ϣ���ȡ�������������֪�й����ʵķе������
��ԭ�ϣ���ȡ�������������֪�й����ʵķе������| ���� | �״� | ������ | ��������� |
| �е�/�� | 64.7 | 249 | 199.6 |
��Բ����ƿ�м���12.2g�������20mL�״����ܶ�ԼΪ0.79g•cm-3������С�ļ���3mLŨ���ᣬ���Ⱥ�Ͷ�뼸�����Ƭ��С�ļ���ʹ��Ӧ��ȫ���ñ���������ֲ�Ʒ��
��1��Ũ����������Ǵ�������ˮ�ԣ�
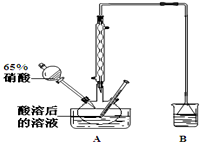
��2��������λͬѧ�ֱ��������ͼ1��ʾ������ʵ���Һϳɱ����������װ�� ���г������ͼ�������������ȥ���������л���ķе㣬��ò����ң���ס����ҡ�������װ�ã�
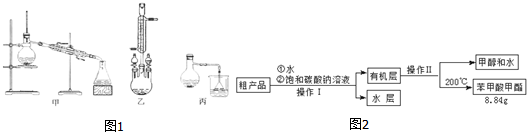
�ֲ�Ʒ�ľ���
��3������������ֲ�Ʒ���������������״������ᡢ�������ˮ�ȣ���������������ͼ���о��ƣ���������ͼ2�з�����������������������ƣ�����I��Һ����������
��4���ܷ���NaOH��Һ���汥��̼������Һ������ܡ���������ԭ��NaOH��ǿ�ʹ���������ˮ�⣬���²�Ʒ��ʧ��
��5��ͨ�����㣬����������IJ���Ϊ65%��
���� ��1��Ũ�����ܶȽϴ����뱽���ᡢ�״���Ϸų�����������Ϊȷ��ʵ��İ�ȫ���Բ���������Ũ���ᣬ�ټ��ϼ״��ķе���ӷ���Ũ��������������ˮ����
��2��������Ӧ���ص��ǣ������ǻ������⣬Ϊ���淴Ӧ��ֻ�����������ܿ�����������ʹ��Ӧ�������������
��3���ɷ������̿�֪����̼�������ռ״�����ȥ�����ᣬ���ͱ�����������ܽ�ȣ��ֲ���Һ���л����к��״������������������ɷ��룻
��4������NaOH��Һ�пɷ���ˮ�ⷴӦ��
��5�����ݼ��㱽��������IJ��ʣ�
��� �⣺��1��������ӦΪ���淴Ӧ��������ˮ����Ũ����������Ǵ�������ˮ�ԣ�
�ʴ�Ϊ����������ˮ�ԣ�
��2��ֻ�����������ܿ�����������ʹ��Ӧ������������������ѡ���ң��ʴ�Ϊ���ң�
��3���ɷ������̿�֪����̼�������ռ״�����ȥ�����ᣬ���ͱ�����������ܽ�ȣ��ֲ���Һ���л����к��״������������������ɷ��룬�����IΪ��Һ������IIΪ ����
�ʴ�Ϊ����Һ������
��4��������NaOH��Һ���汥��̼������Һ����NaOH��ǿ�ʹ���������ˮ�⣬���²�Ʒ��ʧ��
�ʴ�Ϊ����NaOH��ǿ�ʹ���������ˮ�⣬���²�Ʒ��ʧ��
��5����
122 32 136
12.2g 20mL��0.79g/mL x
�״��������Ա�������㱽�������������Ϊx=$\frac{136��12.2g}{122}$=13.6g����������IJ���Ϊ$\frac{8.84g}{13.6g}$��100%=65%��
�ʴ�Ϊ��65%��
���� ���⿼���л�����Ʊ�ʵ�飬Ϊ��Ƶ���㣬�����Ʊ�ԭ�����л�������ʡ����������ᴿΪ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע��ʵ�����ݼ����������ᴿ��Ӧ�ã���Ŀ�ѶȲ���

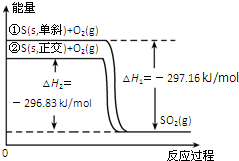
| A�� | ��б��ת��Ϊ������ķ�Ӧ�����ȷ�Ӧ | |
| B�� | ������ȵ�б���ȶ� | |
| C�� | ��ͬ���ʵ�����������ȵ�б�������е������� | |
| D�� | �ٱ�ʾ����1mol O2�еĹ��ۼ������յ��������γ�lmolSO2�еĹ��ۼ����ų���������297.16 kJ |
��������
| �� ѧ �� | H-H | I-I |
| �Ͽ�1mol��ѧ��ʱ���յ�������kJ�� | b | c |
| A�� | �÷�Ӧ�з�Ӧ�������������������������� | |
| B�� | �Ͽ�1 mol H-H ����1 mol I-I �������������ڶϿ�2 mol H-I ���������� | |
| C�� | �Ͽ�2 mol H-I����������Ϊ��a+b+c��kJ | |
| D�� | ���ܱ������м���2 mol H2��2 mol I2����ַ�Ӧ��ų�������С��2a kJ |
| A�� | 16mol | B�� | 6mol | C�� | 12mol | D�� | 4mol |
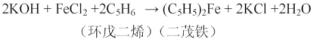
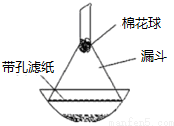
 �������ƿɴ�������Ⱦ�Ϻ��л��ϳɹ�ҵ��
�������ƿɴ�������Ⱦ�Ϻ��л��ϳɹ�ҵ��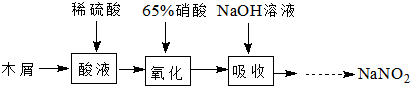
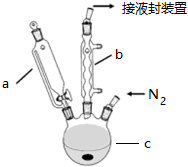
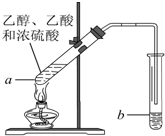 ��ʵ���ҿ������Թ�a�м���3mL �Ҵ���Ȼ������Թܱ���������2mLŨ�����2mL ���ᣬ����ͼ��ʾ�����Ӻ�װ�ý���ʵ�飬����ȡ�����������ش��������⣺
��ʵ���ҿ������Թ�a�м���3mL �Ҵ���Ȼ������Թܱ���������2mLŨ�����2mL ���ᣬ����ͼ��ʾ�����Ӻ�װ�ý���ʵ�飬����ȡ�����������ش��������⣺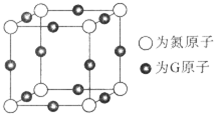 A��B��C��D��E��F��G��ǰ�����ڣ���ϡ�����壩ԭ�������������������Ԫ�أ�A��ԭ�Ӻ������ֻ��һ���˶�״̬��B��C�ļ۵��Ӳ���δ�ɶԵ������� ��2��B��C��Dͬ���ڣ�E�����s��p�ܼ��ĵ��������ࣻF��Eͬ�����ҵ�һ�����ܱ�EС��G��+1�����ӣ�G+���ĸ������ȫ�������ش��������⣺�����������Ԫ�ط��ű�ʾ��
A��B��C��D��E��F��G��ǰ�����ڣ���ϡ�����壩ԭ�������������������Ԫ�أ�A��ԭ�Ӻ������ֻ��һ���˶�״̬��B��C�ļ۵��Ӳ���δ�ɶԵ������� ��2��B��C��Dͬ���ڣ�E�����s��p�ܼ��ĵ��������ࣻF��Eͬ�����ҵ�һ�����ܱ�EС��G��+1�����ӣ�G+���ĸ������ȫ�������ش��������⣺�����������Ԫ�ط��ű�ʾ�� ����λ���á��������
����λ���á��������