��Ŀ����
2��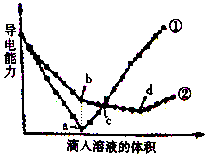 ��������ͬ��Ba��OH��2��Һ�У��ֱ�������ʵ���Ũ����ȵ�H2SO4��NaHSO4��Һ���䵼�������������Һ����仯��������ͼ��ʾ��
��������ͬ��Ba��OH��2��Һ�У��ֱ�������ʵ���Ũ����ȵ�H2SO4��NaHSO4��Һ���䵼�������������Һ����仯��������ͼ��ʾ����ش���������
��1��д��NaHSO4��Һ�У�NaHSO4�ĵ��뷽��ʽNaHSO4=Na++H++SO42-��
��2��ͼ�Т١��ڷֱ�����μ�������Һ�ı仯���ߢ�H2SO4��NaHSO4��
��3��b�㣬��Һ�д������ڵ�������Na+��OH-��
��4��a��b�����Ӧ����Һ�������ԡ����ԣ�����ԡ������ԡ����ԡ���
��5�����ߢ���d�㵼������������ԭ����Ba��OH��2+2NaHSO4=BaSO4��+Na2SO4+2H2O���û�ѧ����ʽ��ʾ��
��6��c�㣬����Һ�к�����ͬ������������SO42-��
���� Ba��OH��2��Һ��H2SO4��NaHSO4��Һ��Ӧ����ʽ�ֱ�ΪH2SO4+Ba��OH��2=BaSO4��+2H2O��NaHSO4+Ba��OH��2=BaSO4��+NaOH+H2O��2NaHSO4+Ba��OH��2=BaSO4��+Na2SO4+2H2O����Һ��������������Ũ�ȳ����ȣ�����ͼ֪�����ߢ���a����Һ���������ӽ�0��˵���õ���Һ����Ũ����С��Ӧ��ΪBa��OH��2��Һ��H2SO4�ķ�Ӧ�������ߢ�ΪBa��OH��2��Һ��NaHSO4��Һ�ķ�Ӧ������ͼ֪��a��ΪBa��OH��2��Һ��H2SO4ǡ�÷�Ӧ��H2SO4��NaHSO4��Һ�����ʵ���Ũ����ȣ���b����Һ����ΪNaOH��c�㣬����ϡ�������������Ϊ���ᣬ���з�Ӧ������ΪNaOH��Na2SO4��a������������������ǡ����ȫ��Ӧ����Һ��ֻ��ˮ��d���������ΪNa2SO4��
��� �⣺Ba��OH��2��Һ��H2SO4��NaHSO4��Һ��Ӧ����ʽ�ֱ�ΪH2SO4+Ba��OH��2=BaSO4��+2H2O��NaHSO4+Ba��OH��2=BaSO4��+NaOH+H2O��2NaHSO4+Ba��OH��2=BaSO4��+Na2SO4+2H2O����Һ��������������Ũ�ȳ����ȣ�����ͼ֪�����ߢ���a����Һ���������ӽ�0��˵���õ���Һ����Ũ����С��Ӧ��ΪBa��OH��2��Һ��H2SO4�ķ�Ӧ�������ߢ�ΪBa��OH��2��Һ��NaHSO4��Һ�ķ�Ӧ�����ٴ����μ�H2SO4��Һ�ı仯���ߣ�
��1��NaHSO4��ǿ����ʣ�����Һ�У�NaHSO4�ĵ��뷽��ʽΪNaHSO4=Na++H++SO42-��
�ʴ�Ϊ��NaHSO4=Na++H++SO42-��
��2�����ߢ�ΪBa��OH��2��Һ��NaHSO4��Һ�ķ�Ӧ���ٴ����μ�H2SO4��Һ�ı仯���ߣ��ʴ�Ϊ��H2SO4��NaHSO4��
��3��a��ΪBa��OH��2��Һ��H2SO4ǡ�÷�Ӧ��H2SO4��NaHSO4��Һ�����ʵ���Ũ����ȣ���b����Һ����ΪNaOH���ʴ�Ϊ��Na+��OH-��
��4��a��ΪBa��OH��2��Һ��H2SO4ǡ�÷�Ӧ�������ᱵ��ˮ����ʾ���ԣ�H2SO4��NaHSO4��Һ�����ʵ���Ũ����ȣ���b����Һ����ΪNaOH����ʾ���ԣ�
�ʴ�Ϊ�����ԣ����ԣ�
��5��d���������ΪNa2SO4����ʱ������Ӧ��Ba��OH��2+2NaHSO4=BaSO4��+Na2SO4+2H2O�������ƶ�����Ũ����С�����������������ʴ�Ϊ��Ba��OH��2+2NaHSO4=BaSO4��+Na2SO4+2H2O��
��6��c�㣬����ϡ�������������Ϊ���ᣬ���з�Ӧ������ΪNaOH��Na2SO4������Һ�к�����ͬ����SO42-���ʴ�Ϊ��SO42-��
���� ���⿼���������Һ�����жϣ�Ϊ��Ƶ���㣬���ؿ���ѧ�������жϼ�ʶͼ��������ȷ�����ķ�Ӧ��������Һ�����ʳɷ��ǽⱾ��ؼ���ע�⣺��Һ��������������Ũ�ȳ����ȣ���Ŀ�Ѷ��еȣ�

| A�� | ��ȡˮ��Ͳ�����ԭ���о��д��� | |
| B�� | ��ͨ�����ǵ�ľ�Ե�壬�������Ĺ��������ǻ����̶����۵� | |
| C�� | ��ͨ�����׳�ˮ���� | |
| D�� | �����ι�ҵʹ�õ�ÿһ��ԭ�϶����й� |
| A�� | ����ȼ�ϵ�������Խ����е�������Ӧʽ��O2+4H++4e-�T2H2O | |
| B�� | ��������������̼����������ⱥ���Ȼ�����Һ�����ӷ���ʽΪ��2C1-+2H2O$\frac{\underline{\;���\;}}{\;}$H2��+Cl2��+2OH- | |
| C�� | ��ͭ����ʱ�����Դ�����������Ǵ�ͭ���缫��ӦʽΪ��Cuһ2e-=Cu2+ | |
| D�� | ��ӦHCl��aq��+NaOH��aq���TNaCl��aq��+H2O��l����H��0�������������������ԭ��� |
��10L���ܱ������а����ʵ���֮��1��2����CO��H2��������10molCO������Ӧ��CO��g��+2H2��g��?CH3OH��g����
���ƽ��ʱCO��ת�����S�¶ȱ仯��ѹǿ�ı仯��ͼ��ʾ��
p2��T2ʱ��n��CO����ʱ��ı仯�����ʾ��
| t/min | 0 | 1 | 3 | 5 |
| n��CO��/mol | 10 | 7 | 5 | 5 |
��2������Ϊp1��p2���������������=����
��3���ϳɼ״��ķ�ӦΪ���ȣ�����ȡ������ȡ�����Ӧ��A��B��C�����ƽ�ⳣ��KA��KB��KC�Ĵ�С��ϵΪKa=Kb��Kc��
��4������ҵ����CH3OH�����д�ʩ�У������ӵ�λʱ����CH3OH�����ķ����Т٢ۢܢݢޣ�
������ѹǿ �ڽ����¶� �������¶�
�ܼ������ ���ʵ�����һ����̼�ı����� ��ʱ����CH3OH
��5����֪̼��ȼ����ΪQ1kJ/mol��̼��������Ӧ����һ����̼������Ͳ�����ȣ��ķ�Ӧ��Ϊ-Q2J/mol���������̼�ڶ�������������ʱ�ֽ���Ȼ�ѧ����ʽ������������ȣ�Ϊ��2CO2��g��=2CO��g��+O2��g����H=+2��Q1-Q2��kJ/mol��
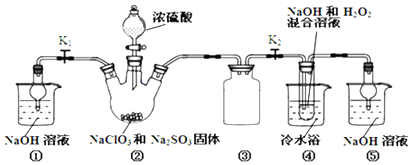
���������գ�
��1��װ�â��в���ClO2�Ļ�ѧ����ʽΪ2NaClO3+Na2SO3+H2SO4=2ClO2+2Na2SO4+H2O��װ�â��з�����Ӧ�����ӷ���ʽΪ2ClO2+H2O2+2OH-=2ClO2-+2H2O+O2����
��2��װ�â۵������Ƿ�ֹ������װ�âٺ͢��ڱ�ʵ���е��������շ�Ӧ���������壬��ֹ����Ⱦ������
��3��װ�âܷ�Ӧ�����Һ�л���������NaOH���ʣ��Ӹ���Һ�����ˮNaClO2����IJ�������Ϊ���ټ�ѹ��55�����������ᾧ���ڳ��ȹ��ˣ�����50�����ҵ���ˮϴ�ӣ��ܵ���60�����õ���Ʒ�����������50�����ҵ���ˮϴ�ӵ�ԭ���Ƿ�ֹ����NaClO2•3H2O������¶ȹ��߲�Ʒ�ֽ⣮
��4���������ƴ��Ȳⶨ����ȷ��ȡ��������������Ʒ10.0g���ձ��У�������������ˮ�����ĵ⻯�ؾ��壬�ٵ���������ϡ���ᣬ��ַ�Ӧ�������û��Һ���250mL������Һ����ȡ25.00mL����Һ����2.0 mol•L-1Na2S2O3��Һ�ζ���I2+2S2O32-=2I-+S4O62-�����Ե�����Һ��ָʾ�����ﵽ�ζ��յ�ʱ������Ϊ������һ�α�Һ����ƿ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ���ظ��ζ�3�Σ�������������ʾ�������Ʒ��NaClO2����������Ϊ90.5%��
| ʵ����� | �ζ�ǰ����/mL | �ζ������/mL |
| 1 | 0.00 | 19.96 |
| 2 | 3.26 | 23.30 |
| 3 | 1.10 | 23.40 |
| A�� | ����32.8% | B�� | С��32.8% | C�� | ����32.8% | D�� | Լ����32.8% |
��1����ӦC��s��+H2O��g��?CO��g��+H2��g����H=+131.3kJ•mol-1���ﵽƽ����������ʱ���������������H2���ʵĴ�ʩ��BC��
A������̼�������� B�������¶�
C����CO���ռ���ȥCO���� D���������
��2����֪��C��s��+CO2��g��?2CO��g����H=+172.5kJ•mol-1
��Ӧ CO��g��+H2O��g��?CO2��g��+H2��g�� �ġ�H=-41.2kJ•mol-1��
��3����һ���¶��£���CO��g����H2O��g����0.16mol�ֱ�ͨ�뵽���Ϊ2.0L�ĺ����ܱ������У��������·�Ӧ��CO��g��+H2O��g��?CO2��g��+H2��g�����õ��������ݣ�
| t/min | 2 | 4 | 7 | 9 |
| n��H2O��/mol | 0.12 | 0.11 | 0.10 | 0.10 |
�������������䣬�ٳ���0.1mol CO��g����0.1mol H2O��g�����ﵽƽ��ʱCO������������䣨���������С�����䡱����
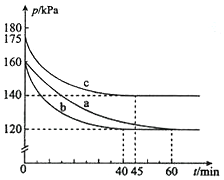 ������AX3�͵���X2��һ�������·�Ӧ�����ɻ�����AX5���ش��������⣮
������AX3�͵���X2��һ�������·�Ӧ�����ɻ�����AX5���ش��������⣮