��Ŀ����
18��A��B��C��D��M��Ϊ��ѧ��ѧ�������ʣ����ǵ�ת����ϵ��ͼ��ʾ������������ͷ�Ӧ������ȥ��������A��H2O�ķ�Ӧ�ڳ����½��У������ƶϲ���ȷ���ǣ�������
| A�� | ��MΪ���г�������ζ�����壬��D����Ϊ��ʽ�Σ���M��A��=M��C�� | |
| B�� | ��MΪ��������D����Һ��ͨ��A���ܵ�C | |
| C�� | ��CΪ���Cת��ΪD�ķ�Ӧ��CO32-+H2O+CO2�T2HCO3-����A����Ϊ���� | |
| D�� | ��MΪ���������Σ���A��ˮ��Ӧ���ܲ���������Ҳ���ܲ������� |
���� A��MΪ���г�������ζ�����壬Ϊ���⣬A��ΪNa2O2��BΪNaOH��CΪNa2S��DΪNaHS��
B����MΪ����A��ΪNO2��BΪHNO3��CΪFe��NO3��3��DΪFe��NO3��2��
C����CΪ���BΪNaOH��MΪCO2��
D��MΪ���������Σ�A��ΪNa��Na2O2��DΪAl��0H��3��BΪNaOH��CΪƫ�����Σ�
��� �⣺A��MΪ���г�������ζ�����壬Ϊ���⣬A��ΪNa2O2��BΪNaOH��CΪNa2S��DΪNaHS������M��A��=M��C������A��ȷ��
B����MΪ����A��ΪNO2��BΪHNO3��CΪFe��NO3��3��DΪFe��NO3��2����NO2����ˮ��Ӧ�������ᣬ��Fe��NO3��2��Һ��ͨ���������������Fe��NO3��3����B��ȷ��
C����CΪ���BΪNaOH��MΪCO2��A��������������C����
D��MΪ���������Σ�A��ΪNa��Na2O2��DΪAl��0H��3��BΪNaOH��CΪƫ�����Σ�A��ˮ��Ӧ���ܲ���������Ҳ���ܲ�����������D��ȷ��
��ѡC��
���� ���⿼��������ƶϣ��ۺϿ���Ԫ�ػ���������ʣ���Ŀ�ѶȽϴ����ñ���Ĺؼ�֮�����ڰ��պ����ʵ����ʣ�ѡ��D���ơ�����������ˮ�ķ�Ӧ��

��ϰ��ϵ�д�
 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�
�����Ŀ
8��a��b��c��d��e�ֱ���Cu��Ag��Fe��Al��Mg���ֽ����е�һ�֣���֪��1��a��d��e������ϡ���ᷴӦ������������2��b�ܴ�c������Һ���û���c���ʣ���3��a��ǿ����Һ��Ӧ�ų����壻��4��a��e����Ũ�����Ũ�����з����ۻ����ɴ˿��ж�a��b��c��d��e�����ǣ�������
| A�� | Fe��Cu��Ag��Al��Mg | B�� | Mg��Cu��Al��Ag��Fe | C�� | Al��Cu��Ag��Mg��Fe | D�� | Mg��Ag��Cu��Al��Fe |
9����m g��ij�ֿ�ȼ��A�ڹ����������г��ȼ�գ��ٽ�ȼ�յIJ���ͨ������Na2O2��ֵ����գ����ֹ������ӵ���������m g�����ȼ��A�����ǣ�������
| A�� | һ����̼ | B�� | �Ҵ���C2H5OH�� | C�� | ���ᣨHCOOH�� | D�� | �״���CH3OH�� |
6����1L1.5mol•L-1�������������ձ�״����22.4LCO2��������Һ�е�CO32-��HCO3-�����ʵ���Ũ��֮��Ϊ��������
| A�� | 1��3 | B�� | 1��2 | C�� | 1��1 | D�� | 3��2 |
13������������Ԫ��X��Y��Z��W�����ڱ��е����λ�����������Ԫ�ص�ԭ������������֮��Ϊ20������˵����ȷ���ǣ�������
| X | Y | Z |
| W |
| A�� | Wλ�ڵ������ڢ�A�� | |
| B�� | Ԫ��������ۣ�Z��Y��X | |
| C�� | ����̬�⻯���ȶ��ԣ�W��Y��X | |
| D�� | ��ҵ�ϴ�����Y���⻯��������Ʊ��京���� |
3�������£���20mL 0.2mol��L-1��Ԫ��H2A��Һ�еμ�0.2mol��L-lNaOH��Һ���й������ʵ����仯��ͼ������������ȷ���ǣ�������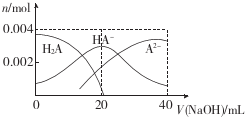

| A�� | ��V��NaOH��=20 mLʱ����Һ�и�����Ũ�ȵĴ�С˳��Ϊc��Na+����c��HA-����c�� A2-����c�� OH-��������H+�� | |
| B�� | �������Ũ�ȵ�NaOH��Һ��H2A��Һ��Ϻ�����Һ��ˮ�ĵ���̶ȱȴ�ˮ�еĴ� | |
| C�� | ��Ũ��H2A��NaHA�Ļ����Һ�����ۼ���������ǿ���ǿ���Һ��pH�仯������ | |
| D�� | ��V��NaOH��=40 mLʱ�������¶ȣ�$\frac{c��N{a}^{+}��}{c��{A}^{2-}��}$��С |
10��Ŀǰ�����ڶ�����̼�Ƿ�Ϊ������Ⱦ���в�ͬ�Ĺ۵㣺�ٶ�����̼����Ҫ�Ļ���ԭ�� �ڶ�����̼��ֲ�������õı���ԭ�� �۶�����̼����ɫ����ζ���������� �ܳ�������̼�⣬���顢һ��������Ҳ���������壮��Ϊ��������̼���Ǵ�����Ⱦ��������ǣ�������
| A�� | �٢� | B�� | �ڢ� | C�� | �ۢ� | D�� | �٢� |
7��һ�������£��ü�������������������NOx������Ⱦ����֪��
��CH4��g��+4NO2 ��g���T4NO��g��+CO2 ��g��+2H2 O��g����H1=-574kJ•mol-1
��CH4 ��g��+4NO��g���T2N2 ��g��+CO2 ��g��+2H2 O��g����H2=-1160kJ•mol-1
����ѡ���ȷ���ǣ�������
��CH4��g��+4NO2 ��g���T4NO��g��+CO2 ��g��+2H2 O��g����H1=-574kJ•mol-1
��CH4 ��g��+4NO��g���T2N2 ��g��+CO2 ��g��+2H2 O��g����H2=-1160kJ•mol-1
����ѡ���ȷ���ǣ�������
| A�� | CH4 ��g��+2NO2 ��g���TN2 ��g��+CO2 ��g��+2H2 O��g����H=-867 kJ•mol-1 | |
| B�� | CH4 ��g��+4NO2 ��g���T4NO��g��+CO2 ��g��+2H2O��l����H3����H1 | |
| C�� | ����0.2 mol CH4��ԭNO2��N2����Ӧ�зų�������һ��Ϊ173.4kJ | |
| D�� | ���ñ�״����2.24 L CH4��ԭNO2��N2������������ת�Ƶĵ���Ϊ0.8mol |
8��NA���������ӵ�����������˵����ȷ���ǣ�������
| A�� | ��״���£�22.4LSO3����ԭ����Ϊ4NA | |
| B�� | 1.0LŨ��Ϊ1.0mol/L��һԪ����Һ�к���NA�������� | |
| C�� | 7.8g Na2O2�������ˮ�У���Ӧת�Ƶĵ�����Ϊ0.2NA | |
| D�� | ͬ��ͬѹ�£�ԭ������ΪNA�������ͺ������в�ͬ����� |