��Ŀ����
5����1��ʵ����16g�״�[CH3OH��l��]�������г��ȼ�����ɶ�����̼�����Һ̬ˮʱ�ͷų�363.25kJ����������д���״�ȼ���ȵ��Ȼ�ѧ����ʽ��CH3OH ��l��+$\frac{3}{2}$ O2��g��=CO2��g��+2H2O��l����H=-726.5KJ/mol��
��2���ӻ�ѧ���ĽǶȷ�������ѧ��Ӧ�Ĺ��̾��Ƿ�Ӧ��Ļ�ѧ�����ƻ���������Ļ�ѧ�����γɹ��̣���֪��Ӧ��N2��g��+3H2��g��?2NH3��g����H=a kJ•mol-1���йؼ������������
| ��ѧ�� | H-H | N-H | N��N |
| ���ܣ�kJ•mol-1�� | 436 | 391 | 945 |
��3�����ݸ�˹���ɿ��Զ�ijЩ����ͨ��ʵ��ֱ�Ӳⶨ�Ļ�ѧ��Ӧ�ķ�Ӧ�Ƚ������㣮��֪��
C��s��ʯī��+O2��g���TCO2��g����H1=-393.5kJ•mol-1
2H2��g��+O2��g���T2H2O��l����H2=-571.6kJ•mol-1
2C2H2��g��+5O2��g���T4CO2��g��+2H2O��l����H3=-2599kJ•mol-1
���ݸ�˹���ɣ�����298Kʱ��C��s��ʯī����H2��g������1mol C2H2��g����Ӧ�ķ�Ӧ��Ϊ��
��H=+226.7 KJ/mol��
��4�����������õ������£�NH4+����������Ӧ��������NO3-��������Ӧ�������仯ʾ
��ͼ��ͼ��

��һ����Ӧ�Ƿ��ȷ�Ӧ������ȡ������ȡ�����ԭ���Ƿ�Ӧ��������������������������
���� ��1��ȼ������1mol��ȼ����ȫȼ�������¶�������ų��������������Ȼ�ѧ����ʽ����д������֪����ѧ�������뷴Ӧ�ȳ����ȣ���ע��������ʵľۼ�״̬�����
��2����Ӧ�ȵ��ڷ�Ӧ����ܼ���-��������ܼ������㣻
��3�������ȸ��ݷ�Ӧ�����������д��ѧ����ʽ�����ݸ�˹���ɼ��㷴Ӧ���ʱ䣬�������Ȼ�ѧ����ʽ����д��������д�Ȼ�ѧ����ʽ��
��4������Ӧ������������������������������Ӧ�Ƿ��ȵģ�
��� �⣺��1��16gCH3OH��������ȼ������CO2��Һ̬ˮ���ų�363.25kJ������32g��1molCH3OH��������ȼ������CO2��Һ̬ˮ���ų�726.5kJ������
���H=-726.5KJ/mol����ȼ���ȵ��Ȼ�ѧ����ʽΪ��CH3OH ��l��+$\frac{3}{2}$ O2��g��=CO2��g��+2H2O��l����H=-726.5KJ/mol��
�ʴ�Ϊ��CH3OH ��l��+$\frac{3}{2}$ O2��g��=CO2��g��+2H2O��l����H=-726.5KJ/mol��
��2��N2��g��+3H2��g��???2NH3��g����H=945kJ•mol-1+436kJ•mol-1��3-391kJ•mol-1��6=-93kJ•mol-1=a kJ•mol-1�����a=-93��
�ʴ�Ϊ��-93��
��3����֪����C ��s��ʯī��+O2��g��=CO2��g����H1=-393.5kJ•mol-1��
��2H2��g��+O2��g��=2H2O ��l����H2=-571.6kJ•mol-1��
��2C2H2��g��+5O2��g���T4CO2��g��+2H2O ��l����H2=-2599kJ•mol-1��
2C ��s��ʯī��+H2��g��=C2H2��g���ķ�Ӧ���Ը��ݢ١�2+�ڡ�$\frac{1}{2}$-�ۡ�$\frac{1}{2}$�õ���
���Է�Ӧ�ʱ��H=2����-393.5kJ•mol-1��+��-571.6kJ•mol-1����$\frac{1}{2}$-��-2599kJ•mol-1����$\frac{1}{2}$=+226.7kJ•mol-1��
�ʴ�Ϊ��+226.7 KJ/mol��
��4����Ϊ��H=-273kJ/mol��0����ӦΪ���ȷ�Ӧ����Ӧ����������������������������
�ʴ�Ϊ�����ȣ���Ӧ��������������������������
���� ���⿼�鷴Ӧ�ȵļ��㣬ע�������֪�Ȼ�ѧ����ʽ���ø�˹���ɼ��㷴Ӧ�ȵķ���������ʱע����ᣬ��Ŀ�Ѷ��еȣ�

| A�� | ���Ǻ���̼Ԫ�صĻ����ﶼ���л������� | |
| B�� | ���е��л�����������ܽ���ˮ | |
| C�� | �л������Ԫ�ؽ��٣����л��������� | |
| D�� | ��ȫȼ��ֻ����CO2��H2O���л������� |
�ش��������⣺
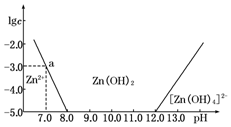
��1����ZnCl2��Һ�м�������������������Һ����Ӧ�����ӷ���ʽ�ɱ�ʾΪZn2++4OH-�T[Zn��OH��4]2-��
��2����ͼ�����ݼ���ɵ�Zn��OH��2���ܶȻ�Ksp=1.00��10-17��
��3��ij��Һ�к�Zn2+��Ϊ��ȡZn2+���Կ�����Һ��pH�ķ�Χ��8.0��pH��12.0��
ij��������ˮ���Ȼ�þ�ʹ�ʯ����ȡ��������þ�������������������ʣ�ͨ���������̽����ᴿ���ƣ������ȼ��������þ��
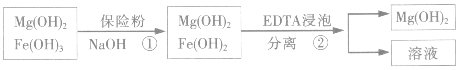
��4��������м��뱣�շۣ�Na2S2O4�������ã�������������ԭΪ����������
��5����֪EDTAֻ������Һ�е�Fe2+��Ӧ����������ˮ�����ʣ�����Mg��OH��2��Ӧ����ȻFe��OH��2������ˮ���������������EDTA�ļ��룬�����ܹ���Fe��OH��2��ȥ����ô��ȸߵ�Mg��OH��2����ӳ����ܽ�ƽ��ĽǶȼ��Խ���Fe��OH��2����Һ�д�������ƽ�⣺Fe��OH��2��s��=Fe2+��aq��+2OH-��aq�������ϵ���EDTAʱ��EDTA�����Fe 2+��ʹƽ�������ƶ���ʹFe��OH��2�����ܽ⣻��
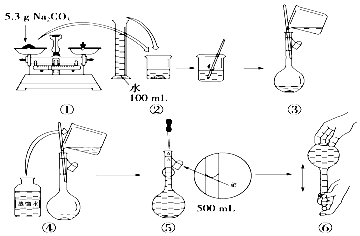
����Ϊ�о���ͬ�����ᴿ���������Ƶ���ȼ���Ĵ��ȴӶ�ȷ������ᴿ������ij�о�С���ȡ������������4���������Ƶõ���ȼ�����к������IJⶨ����������
| ������ȼ�������� | ��ȼ�������� | |||
| ��� | �ᴿ��ϵ�¶�/�� | ����EDTA����/g | ���뱣�շ�����/g | W��Fe��/��10-4g�� |
| 1 | 40 | 0.05 | 0.05 | 7.63 |
| 2 | 40 | 0.05 | 0.10 | 6.83 |
| 3 | 60 | 0.05 | 0.10 | 6.83 |
| 4 | 60 | 0.10 | 0.10 | 6.51 |
��40���60���EDTA����ΪO.05g ��EDTA����Ϊ0.10g �ݱ��շ�����Ϊ0.05g�ޱ��շ�����Ϊ0.10g
A���٢ۢ�B���ڢܢ�C���٢ܢ�D���ڢ�

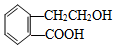 ������ͨ����ͬ��ѧ��Ӧ�ֱ��Ƶ�B��C��D��E�������ʣ�
������ͨ����ͬ��ѧ��Ӧ�ֱ��Ƶ�B��C��D��E�������ʣ�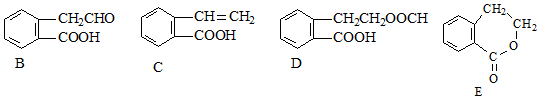
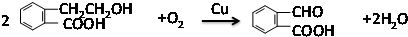 ����Ӧ���ͣ�������Ӧ
����Ӧ���ͣ�������Ӧ ����Ӧ���ͣ�������Ӧ
����Ӧ���ͣ�������Ӧ ��
�� ��
�� ��
��