��Ŀ����
2013�꣬�й�������������Ƶ�����ܵ����㷺��ע�����д�ʩ�����ܸ��ƿ����������ǣ�������
| A��Ӧ�ø�Ч�ྻ����Դת���������ڴ���ȼú��¯�ϰ�װ�͵�ȼ������������������������ |
| B����ǿ��·�ﳾ����ǿ��·�汣����ˮ���� |
| C���ƹ��ʹ������������������������ŷ� |
| D���о���ú�������¼����������͡����͵���Դ�Ĺ�Ӧ�����ᳫ�������������ִ��������������� |
���㣺���������������Ⱦ������
ר�⣺
������A������ú�к���NԪ��ȼ�պ����������������Ⱦ�������ƹ�ʹ�������������������ε�����������Ⱦ��
B��������ˮ���Է���������
C������ú�к�����Ԫ��ȼ�պ��������������Ⱦ�������ƹ�ʹ��ȼú������������SO2��Ⱦ��
D�������˻�ʯȼ�ϵ��������������ܸ��ƿ���������
B��������ˮ���Է���������
C������ú�к�����Ԫ��ȼ�պ��������������Ⱦ�������ƹ�ʹ��ȼú������������SO2��Ⱦ��
D�������˻�ʯȼ�ϵ��������������ܸ��ƿ���������
���
�⣺A��ú�к���NԪ��ȼ�պ����������������Ⱦ�������ƹ�ʹ�������������������ε�����������Ⱦ�����Ը��ƿ�����������A��ȷ��
B����ǿ��·�ﳾ����ǿ��·�汣����ˮ���������Ը��ƿ�����������B��ȷ��
C��ú�к�����Ԫ��ȼ�պ��������������Ⱦ�������ƹ�ʹ��ȼú������������SO2��Ⱦ�����Ը��ƿ�����������C��ȷ��
D��Ӧ�����µ�������Դ������ú��ʯ�͵�ʹ�ã����������������ִ��������������������˻�ʯȼ�ϵ��������������ܸ��ƿ�����������D����
��ѡD��
B����ǿ��·�ﳾ����ǿ��·�汣����ˮ���������Ը��ƿ�����������B��ȷ��
C��ú�к�����Ԫ��ȼ�պ��������������Ⱦ�������ƹ�ʹ��ȼú������������SO2��Ⱦ�����Ը��ƿ�����������C��ȷ��
D��Ӧ�����µ�������Դ������ú��ʯ�͵�ʹ�ã����������������ִ��������������������˻�ʯȼ�ϵ��������������ܸ��ƿ�����������D����
��ѡD��
���������⿼����Դ����������֪ʶ��ͨ��������ѧ����һ����ȷ�˼��ٴ�����Ⱦ�ķ���������������ѧ���������������������ʶ���ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
����˵����ȷ���ǣ�������
| A���ܻ���DEHP��ָ�ڱ����������2���һ�������������������������ʳƷ���Ӽ� |
| B����������������ѹ������ɫҺ��Ϊ�����仯 |
| C��Na2O?SiO2��һ�ּĹ����Σ�������ˮ |
| D��Ϊ�ⶨ�����������Ƶĵ����ԣ��ɽ��������ƹ������ʯӢ�����м����ۻ� |
����ѪҺ�д��ڵ�ƽ�⣺H2CO3?H++HCO3-��ʹѪҺpH������7.35��7.45֮�䣬����ͻᷢ�����ж�����ж�����֪pH�� �仯��ϵ�����ʾ��������˵���д�����ǣ�������
| 1.0 | 17.8 | 20.0 | 22.4 | ||||
| pH | 6.10 | 7.35 | 7.40 | 7.45 |
| A��pH=7��ѪҺ�У�c��HCO3-����c��H2CO3�� | ||||
| B���������������巢�����ж�ʱ��c��H+��?c��OH-����� | ||||
| C�����巢�����ж�ʱ���ɾ�����עһ��Ũ�ȵ�NaHCO3��Һ�ⶾ | ||||
D��
|
���з�Ӧ������Һ��ֻ��һ�����ʵ��ǣ�������
| A��Fe2��SO4��3��Һ�м������Fe�� |
| B��A1��OH��3�м������NaOH��Һ |
| C��ŨH2SO4�м������CuƬ������ |
| D��Ca��C1O��2��Һ��ͨ�����CO2 |
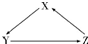 X��Y��ZΪ��ѧ��ѧ�������ʣ���ת����ϵ����ͼ��ʾ����������ʾһ��ת������������ϲ���ȷ���ǣ�������
X��Y��ZΪ��ѧ��ѧ�������ʣ���ת����ϵ����ͼ��ʾ����������ʾһ��ת������������ϲ���ȷ���ǣ�������| ѡ�� | X | Y | Z |
| A | SiO2 | H2SiO3 | Na2SiO3 |
| B | Na | Na2O2 | NaOH |
| C | H2SO3 | SO2 | H2SO4 |
| D | HClO | Cl2 | HCl |
| A��A | B��B | C��C | D��D |
�����£���Ũ�Ⱦ�Ϊ0.1mol/LһԪ��HA��Һ��NaOH��Һ�������ϣ��õ������Һ�٣��û����ҺpH=9������˵����ȷ���ǣ�������
| A������Һ�У�c��A?����c��Na+����c��OH?����c��H+�� |
| B������Һ�У�c��HA��+c��A-��=0.1mol/L |
| C�������£�0.1mol/LHA��Һ��pH=1 |
| D��0.1mol/LHA��Һ�У�c��HA����c��A?�� |
NA��ʾ�����ӵ�������ֵ������˵������ȷ���ǣ�������
| A��1mol Zn����������Ũ�ȵ����ᷴӦת�Ƶ���������2NA |
| B���ڳ��³�ѹ��1molC7H16�к��й��ۼ�������Ŀ��23NA |
| C��0.1molFeCl3��ȫˮ������Fe��OH��3����������ĿΪ0.1NA |
| D��33.6L �����ڱ�״����������HCl���ӵ���ĿΪ1.5NA |
100mL 6mol?L-1��H2SO4��������п��Ӧ����һ���¶��£�Ϊ������Ӧ���ʣ����ֲ�Ӱ����������������������Ӧ���м��������ģ�������
| A�������ƹ��� | B��ˮ |
| C���������Һ | D���������Һ |