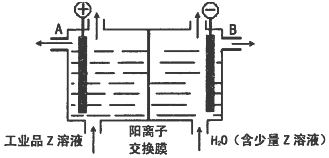
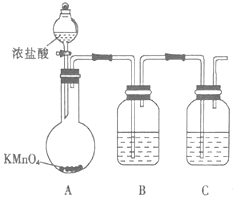
��Ŀ����
5��LiPF6������ӵ���й㷺Ӧ�õĵ���ʣ�ij������LiF��PCl5Ϊԭ�ϣ����·�Ӧ�Ʊ�LiPF6����������ͼ��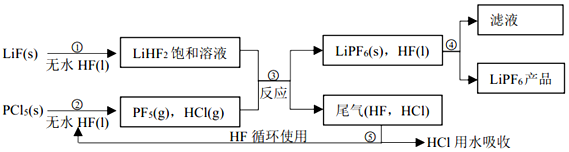
��֪��HCl�ķе���-85.0�棬HF�ķе��� 19.5�森
��1���ڢٲ���Ӧ����ˮ HF �������Ƿ�Ӧ��ܼ�����Ӧ�豸�����ò������ʵ�ԭ����SiO2+4HF�TSiF4��+2H2O���û�ѧ����ʽ��ʾ����
��2��������������ˮ�����½��У��ڢ۲���Ӧ��PF5����ˮ�⣬�����Ϊ�����ᣬд��PF5ˮ��Ļ�ѧ����ʽ��PF5+4H2O�TH3PO4+5HF��
��3���ڢܲ�������õķ����ǹ��ˣ��ڢݲ�����β���� HF��HCl ���õķ�����������
���� ��1��������Ŀ�е����̿��Կ���������+Һ���������+������Һ��������ˮHF�������Ƿ�Ӧ����ܼ�����������Ҫ�ɷ��к��ж������裬�ܺ�HF������Ӧ��
��2������Ԫ����ɿ�֪��������ֱ���H3PO4��HF��
��3������Һ��Ͳ�����Һ�������ù��˷��룬HF���Ӽ京��������е����HCl���ɲ������������룮
��� �⣺��1��������Ŀ�е����̿��Կ���������+Һ���������+������Һ��������ˮHF�������Ƿ�Ӧ����ܼ�����������Ҫ�ɷ��к��ж������裬�ܺ�HF������Ӧ����ѧ����ʽΪSiO2+4HF�TSiF4��+2H2O���ʴ�Ϊ����Ӧ��ܼ���SiO2+4HF�TSiF4��+2H2O��
��2��������Ŀ�е���Ϣ��PF5����ˮ�⣬�����Ϊ�����ᡱ�������Ԫ����ɿ�֪��������ֱ���H3PO4��HF�����Է�Ӧ�ķ���ʽΪPF5+4H2O�TH3PO4+5HF��
�ʴ�Ϊ��PF5+4H2O�TH3PO4+5HF��
��3���ڢܲ�������ǹ��壨LiPF4��s������Һ�壨HF��l���������Բ��ù��˵ķ���������β����HF��HCl���������ö��߷е�IJ��죨HF����֮�������������з��룬���Բ�����������
�ʴ�Ϊ�����ˣ�������
���� ����Ϊ����ѧ�����⣬�����Ǹ߿��еij������ͣ������е��Ѷȵ����⣮�����ۺ���ǿ���漰�����ƶϡ������ᴿ������ʽ��д����ѧ�����֪ʶ��ע�����������Ϣ��ʹ�ã����ض�ѧ��ʵ�������������ͽ��ⷽ����ָ��������������ѧ���淶���Ͻ���ʵ����ƺ�����������

 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�| ʵ����� | ��Ʒ������/g | NaOH��Һ�����/mL | ���������/L |
| �� | 7.4 | 40.00 | 1.68 |
| �� | 14.8 | 40.00 | 3.36 |
| �� | 22.2 | 40.00 | 1.12 |
| �� | 37.0 | 40.00 | 0 |
��2��3.7g����Ʒ����ͬ��ʵ��ʱ�����ɵİ����ڱ�״�������Ϊ0.84L��
��3��������û�����У�NH4��2SO4��NH4HSO4�����ʵ���֮�ȣ�
��4���������NaOH��Һ�����ʵ���Ũ��Ӧѡ��ڢ������ݣ����ɴ˼���NaOH��Һ�����ʵ���Ũ�ȣ�д��������̣�
 ��������Ԫ�ص����ʻ�ṹ��Ϣ���£�
��������Ԫ�ص����ʻ�ṹ��Ϣ���£�| Ԫ�ر�� | �����Ϣ |
| X | �ؿ��к������Ľ���Ԫ�أ�Ԫ�����Ϊ+3�ۣ� |
| Y | ԭ�������������ǵ��Ӳ�����2����������������X������������2���� |
| Z | ͬ��������Ԫ����ԭ�Ӱ뾶��С�������µ��ʳ�Һ̬�� |
| M | �ܴӺ�ˮ����ȡ�Ľ���Ԫ�أ����ʿ��ڵ����������̼��ȼ�գ� |
 ������Ԫ������������Ӧ��ˮ���������ǿ���ǣ�д����ʽ��Mg��OH��2��Ԫ����Ԫ�ط��ű�ʾ����ͬ������
������Ԫ������������Ӧ��ˮ���������ǿ���ǣ�д����ʽ��Mg��OH��2��Ԫ����Ԫ�ط��ű�ʾ����ͬ��������2��Y����������Ũ���������������Ļ�ѧʽΪSO2��
��3��������Z���ʱ�Y���ʻ�����ǿ��һ����ѧ����ʽ��S2-+Br2=S+2Br-��
��4�������£�������X�ĵ��ʳ���������Ӧ���ǣ���ѡ����ţ�bce��
a��CuSO4��Һ b��Fe2O3 c��Ũ���� d��NaOH��Һ e��Na2CO3����
��5����Ԫ����ZԪ���γɻ�����FeZ3��FeZ3��Һ����ͼ��ʾ�������飮װ��ͨ������ӵ�Դ���������������������̼���ߵ�ú����ɫ�������һ̼��������Һ�����ֵ���������Һ���dz��ɫ��
��ȡ������Ʒ�����������ϡ���ᣬ�����ݲ��������岿���ܽ�
����ȡ������Ʒ��������Ũ���Ტ���ȣ������ݲ���������ȫ���ܽ�
�ú�ɫ��ĩ����Ϊ��������
| A�� | Fe��MnO2 | B�� | C��FeS | C�� | Si��FeO | D�� | CuO��Ag2S |
| A�� | ����̪ݼ���ķ��ӣ�ֱ��Ϊ1.3��10-9m����ˮ���γɵķ�ɢϵ�ܲ��������ЧӦ | |
| B�� | ��������ϸ�ڲ����Լ�ƿ���� | |
| C�� | ճ�������ʵ��Թܿ��þƾ�ϴ�� | |
| D�� | ���ع��͡�������Ƶÿ������ |
 ��Al��NaOH��Һ��Ӧ�����ӷ���ʽΪ2Al+2NaOH+2H2O=2NaAlO2+3H2����
��Al��NaOH��Һ��Ӧ�����ӷ���ʽΪ2Al+2NaOH+2H2O=2NaAlO2+3H2����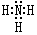 ��D������Һ̬X�з�����������A2C�ķ�Ӧ��д����Ӧ�Ļ�ѧ����ʽ2Na+2NH3=2NaNH2+H2����
��D������Һ̬X�з�����������A2C�ķ�Ӧ��д����Ӧ�Ļ�ѧ����ʽ2Na+2NH3=2NaNH2+H2����