��Ŀ����
3��±�����ڼ��Դ���Һ���ܷ�����ȥ��Ӧ������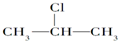 +NaOH$��_{��}^{��}$CH3-CH=CH2+NaCl+H2O
+NaOH$��_{��}^{��}$CH3-CH=CH2+NaCl+H2O�÷�ӦʽҲ�ɱ�ʾΪ��
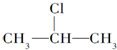 $��_{-NaCl��-H_{2}O}^{NaOH��������}$CH3-CH=CH2
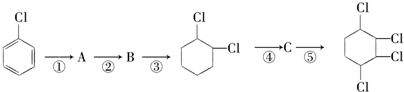
$��_{-NaCl��-H_{2}O}^{NaOH��������}$CH3-CH=CH2�����ǰ˸��л��������ת����ϵ����ش��������⣺

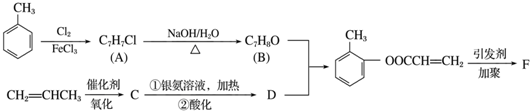
��1������ϵͳ��������������A��������2��3-�������飻
��2��������ͼ�У�����ȡ����Ӧ��Ӧ�����Ǽӳɷ�Ӧ��Ӧ���Ӧ���ͣ���
��3��������E����Ҫ�Ĺ�ҵԭ�ϣ�д����D����E�Ļ�ѧ����ʽ��CH3CBr��CH3��CBr��CH3��2+2NaOH$��_{��}^{��}$CH2=C��CH3��C��CH3��=CH2+2NaBr+2H2O��
��4��C2�Ľṹ��ʽ�ǣ�CH3��2C=C��CH3��2��F1�Ľṹ��ʽ��BrCH2C��CH3��=C��CH3��CH2Br��
��5����д����E�ϳɸ߷��ӻ�����Ļ�ѧ����ʽ��
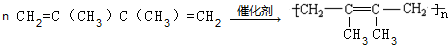 ��
��
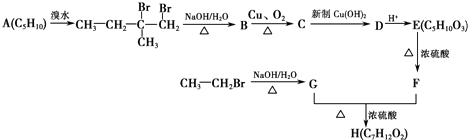
���� A�ڹ�������������������ȡ����Ӧ����B��B���������ƴ���Һ�����������·�����ȥ��Ӧ����C1��C2��C2���巢���ӳɷ�Ӧ����D��D���������ƴ���Һ�����������·�����ȥ��Ӧ����E��E������Է���1��2-�ӳɷ�Ӧ����F2������1��4-�ӳɷ�Ӧ����F1����EΪCH2=C��CH3��C��CH3��=CH2��F2ΪCH2=C��CH3��CBr��CH3��CH2Br��F1ΪBrCH2C��CH3��=C��CH3��CH2Br�����ƿɵ�DΪCH3CBr��CH3��CBr��CH3��2��C2Ϊ��CH3��2C=C��CH3��2��C1Ϊ��CH3��2CHC��CH3��=CH2��
��� �⣺A�ڹ�������������������ȡ����Ӧ����B��B���������ƴ���Һ�����������·�����ȥ��Ӧ����C1��C2��C2���巢���ӳɷ�Ӧ����D��D���������ƴ���Һ�����������·�����ȥ��Ӧ����E��E������Է���1��2-�ӳɷ�Ӧ����F2������1��4-�ӳɷ�Ӧ����F1����EΪCH2=C��CH3��C��CH3��=CH2��F2ΪCH2=C��CH3��CBr��CH3��CH2Br��F1ΪBrCH2C��CH3��=C��CH3��CH2Br�����ƿɵ�DΪCH3CBr��CH3��CBr��CH3��2��C2Ϊ��CH3��2C=C��CH3��2��C1Ϊ��CH3��2CHC��CH3��=CH2��
��1��������A�������ǣ�2��3-�������飬�ʴ�Ϊ��2��3-�������飻
��2��������ͼ�У���Ӧ������ȡ����Ӧ����Ӧ�����ڼӳɷ�Ӧ���ʴ�Ϊ��ȡ����Ӧ���ӳɷ�Ӧ��
��3��D����E�Ļ�ѧ����ʽ��CH3CBr��CH3��CBr��CH3��2+2NaOH$��_{��}^{��}$CH2=C��CH3��C��CH3��=CH2+2NaBr+2H2O��
�ʴ�Ϊ��CH3CBr��CH3��CBr��CH3��2+2NaOH$��_{��}^{��}$CH2=C��CH3��C��CH3��=CH2+2NaBr+2H2O��
��4��C2�Ľṹ��ʽ�ǣ���CH3��2C=C��CH3��2��F1�Ľṹ��ʽ�ǣ�BrCH2C��CH3��=C��CH3��CH2Br��
�ʴ�Ϊ����CH3��2C=C��CH3��2��BrCH2C��CH3��=C��CH3��CH2Br��
��4����E�ϳɸ߷��ӻ�����Ļ�ѧ����ʽ��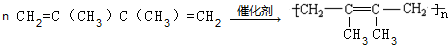 ��
��
�ʴ�Ϊ��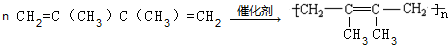 ��
��
���� ���⿼���л�����ƶϣ��Ѷ��еȣ��������չ����ŵ�������ת���ǹؼ�����ѧ������������һ����Ҫ��

| A�� | KOH | B�� | NaCl | C�� | O2 | D�� | NH3 |
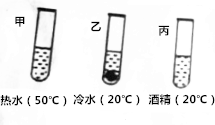 ������Һ�й�֪ʶ���ش��������⣺
������Һ�й�֪ʶ���ش��������⣺��1��������ˮ����������Ϊ0.9%���Ȼ�����Һ��ʵ������Ҫ����������ˮ200g��Ӧ��ȡ�Ȼ��ƹ���1.8g��ʵ����Ҫ�õ��IJ����������ձ�����Ͳ������������ͷ�ιܣ�
��2���ڼס��ҡ�����֧�Թ��и�����2gX���壮�ٷֱ����������Ϊ10.0g����ˮ����ˮ���ƾ�����ֽ��農�ú����ͼ��ʾ��
| �¶�/�� | 10 | 30 | 50 | 70 | |
| �ܽ��/g | NaCl | 35.8 | 36.3 | 37.0 | 37.8 |
| KNO3 | 20.9 | 45.8 | 85.5 | 138 | |
��3�����ݱ����ṩ����Ϣ�ýᾧ�ķ�������NaCl��KNO3�Ļ���ȡ100g�������100g��ˮ�У���ȫ�ܽ����ȴ��10�棬Ҫ�õ�������KNO3���壬�������KNO3������m��ȡֵ��Χ��85.5 g��m��64.2g��
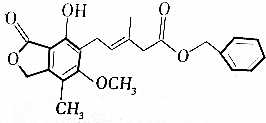
| A�� | �����ʵĻ�ѧʽ��C23H24O6 | |
| B�� | 1mol�û�������������9molH2�ӳ� | |
| C�� | ����KMnO4��Һ����ˮ��������ʷ���������ԭ��Ӧ����ɫ | |
| D�� | ��������FeCl3��Һ���ÿ��Ժ�ɫ���÷�Ӧ�����ڼ������еķ��ǻ� |
��1�������£�Ũ�Ⱦ�Ϊ0.1mol•L-1������������Һ��pH���±���
| ���� | CH3COONa | NaHCO3 | Na2CO3 | NaClO | NaCN |
| pH | 8.8 | 9.7 | 11.6 | 10.3 | 11.0 |
�ڸ��ݱ��������жϣ�Ũ�Ⱦ�Ϊ0.01mol•L-1�������������ʵ���Һ�У�������ǿ����C��
�����ţ���ͬ��������Һ�ֱ�ϡ��100����pH�仯��С����A
A��HCN B��HClO C��H2CO3 D��CH3COOH
�۸����ϱ����ݣ������0.1mol•L-1��NaCN��Һ��ˮ����������������ӵ�Ũ��10-3mol/L
��Ҫ������ˮ��HClO��Ũ�ȣ�������ˮ�м���������̼������Һ������ƽ��ԭ��ԭ��HClO��̼���Ʋ���Ӧ���������̼���Ʒ�Ӧ���ɶ�����̼�����Ե�����ˮ��HClOŨ������2Cl2+CO32-+H2O=CO2��+2Cl-+2HClO��д���ӷ���ʽ���ñ�Ҫ������˵��������0�֣���
��2����0.1mol•L-1��NaClO��Һ�У�д������Ũ�ȵĴ�С��ϵc��Na+����c��ClO-����c��OH-����c��H+����
| A�� | ��ȼú��ȼú������������ | |
| B�� | �ƹ���Ȼ�����״�����Ϊ������ȼ�� | |
| C�� | �Ժ�SO 2��NO 2�ķ������������ſ� | |
| D�� | �˹��ռ�����������ĵ��������� |
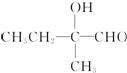 +2Cu��OH��2+OH-$\frac{\underline{\;\;��\;\;}}{\;}$
+2Cu��OH��2+OH-$\frac{\underline{\;\;��\;\;}}{\;}$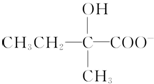 +Cu2O��+3H2O��
+Cu2O��+3H2O��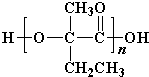 ��
��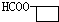 ��
��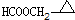 ��
��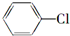 �Ʊ�
�Ʊ�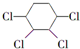
 B��
B�� ��
�� +2NaOH$��_{��}^{��}$
+2NaOH$��_{��}^{��}$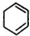 +2NaCl+2H2O
+2NaCl+2H2O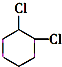 �ĺ�����Ԫ̼����ͬ���칹�壺
�ĺ�����Ԫ̼����ͬ���칹�壺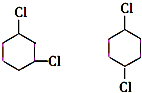
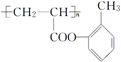 ��
��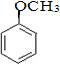 ����ṹ��ʽ����
����ṹ��ʽ����