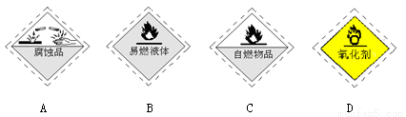
��Ŀ����
��֪298 K��101 kPaʱ���з�Ӧ��
��2H2(g)+O2(g) �� 2H2O(l) ��H ����571.6 kJ��mol��1
��C2H4(g)+3O2(g) �� 2CO2(g)+2H2O(l) ��H ����1 411.0 kJ��mol��1
��C2H5OH(l)+3O2(g) �� 2CO2(g)+3H2O(l) ��H ����1 366.8 kJ��mol��1
��C2H4(g)+H2O(l) ��C2H5OH(l)�Ħ�H Ϊ( )
A����44.2 kJ��mol��1 B����44.2 kJ��mol��1 C����330 kJ��mol��1 D����330 kJ��mol
A
��������
����������ڣ��ۣ������ɵã�C2H4(g)+H2O(l) ��C2H5OH(l)�Ħ�H=��44.2 kJ/mol,���ѡ����A��
���㣺�����Ȼ�ѧ����ʽ����д��֪ʶ��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����11�֣��±���Ԫ�����ڱ���һ����, ��Ա��еĢ١�����Ԫ��,��д���пհ�:
���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0�� |
2 |
|
|
| �� | �� | �� |
|
|
3 | �� |
| �� |
|
| �� | �� | �� |
4 | �� |
|
|
|
|
| �� |
|
��1�� ������������ˮ�����У�������ǿ�Ļ�����ķ���ʽ�ǣ� ��
������ǿ�Ļ�����ĵ���ʽ�ǣ� ��
��2�� ����������������������Ԫ���� ��д���������������������Ʒ�Ӧ�����ӷ���ʽ ��
��3�� �õ���ʽ��ʾԪ�آ���Ļ�������γɹ��̣� ��
�û��������� (�� �����ۡ������ӡ�)�����
��4����ʾ����ߵĻ�����ĵ���ʽ ���û��������� ������ԡ����Ǽ��ԡ������γɵġ�
��5���ۡ��ޡ�������Ԫ���γɵ����ӣ����Ӱ뾶�ɴ�С��˳����_____________________
��Ҫ�������ӷ��ű�ʾ����
��6��Ԫ�آ۵��⻯�ﳣ���º�Ԫ�آߵĵ��ʷ�Ӧ�����ӷ���ʽΪ�� ��
��7��д��������������ˮ����͢ٵĵ��ʷ�Ӧ�Ļ�ѧ����ʽ�� ��