��Ŀ����
7����ͼ��ʾ������ת����ϵ�У���Ӧ�����Ͳ���������δȫ���г�����X�����Ǻ�ˮ�к��������Σ�A��BΪ�������嵥�ʣ�BΪ������ɫ���壬I��LΪ�����Ľ������ʣ�HΪ���ɫ���ʣ�YΪ��������ɫҺ�壮
��ش��������⣺
��1��H�Ļ�ѧʽΪFe��OH��3��
��2��C�ĵ���ʽΪ
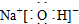 ��
����3����Ӧ�ٵĻ�ѧ����ʽΪ2Al+2NaOH+2H2O�T2NaAlO2+3H2����
��4��Eת��ΪF�����ӷ���ʽΪ2Fe2++Cl2�T2Fe3++2Cl-��
���� X�Ǻ�ˮ�к��������Σ���XΪNaCl��YΪ��������ɫҺ�壬���X��Y�����õ�A��B��C��A��BΪ�������嵥�ʣ�BΪ������ɫ���壬����֪YΪH2O��BΪCl2��AΪH2��CΪNaOH��DΪHCl������I�����ᷴӦ����E����E��F�ת����֪IΪ��۽�������F�����������Ʒ�Ӧ���ɺ��ɫ����H����IΪFe��EΪFeCl2��FΪFeCl2��HΪFe��OH��3������L���������Ʒ�Ӧ����������G����LΪAl��GΪNaAlO2��
��� �⣺X�Ǻ�ˮ�к��������Σ���XΪNaCl��YΪ��������ɫҺ�壬���X��Y�����õ�A��B��C��A��BΪ�������嵥�ʣ�BΪ������ɫ���壬����֪YΪH2O��BΪCl2��AΪH2��CΪNaOH��DΪHCl������I�����ᷴӦ����E����E��F�ת����֪IΪ��۽�������F�����������Ʒ�Ӧ���ɺ��ɫ����H����IΪFe��EΪFeCl2��FΪFeCl3��HΪFe��OH��3������L���������Ʒ�Ӧ����������G����LΪAl��GΪNaAlO2��
��1��H�Ļ�ѧʽΪ��Fe��OH��3���ʴ�Ϊ��Fe��OH��3��
��2��CΪNaOH������ʽΪ��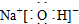 ���ʴ�Ϊ��
���ʴ�Ϊ��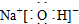 ��
��
��3����Ӧ�ٵĻ�ѧ����ʽΪ��2Al+2NaOH+2H2O�T2NaAlO2+3H2�����ʴ�Ϊ��2Al+2NaOH+2H2O�T2NaAlO2+3H2����
��4��Eת��ΪF�����ӷ���ʽΪ��2Fe2++Cl2�T2Fe3++2Cl-���ʴ�Ϊ��2Fe2++Cl2�T2Fe3++2Cl-��
���� ���⿼�������ƶϣ���X�Ǻ�ˮ�к��������Ρ����ʵ���ɫ״̬������ת����Ϊ�ƶ�ͻ�ƿڣ���Ҫѧ����������Ԫ�ػ�����֪ʶ����Ŀ�Ѷ��еȣ�

| A�� | ������ˮʪ��pH��ֽ��ⶨ��������Һ��pH | |
| B�� | �ⶨ�кͷ�Ӧ�ķ�Ӧ��ʱ��������Һ������������Һ�� | |
| C�� | ������к͵ζ����ⶨδ֪Ũ�ȵļ�Һʱ������ƿ�м���2-3mL��̪��Һ��ָʾ�� | |
| D�� | �ڵ�����Һ�м���ϡ�������һ��ʱ����ٵμ�������Һ������۵�ˮ����� |
| A�� | ���߲������������� | B�� | ǿ����Һ�����������ٶȿ� | ||
| C�� | ������Һ�����϶������ | D�� | ���Ƚ����߲����������� |
| A�� | �÷�Ӧ�ġ�S��0 | |
| B�� | �÷�Ӧ�ġ�H��0 | |
| C�� | �÷�Ӧ���淴Ӧ��һ��������Ҳ���Է����� | |
| D�� | �÷�Ӧ���ر�ЧӦ�����ʱ�ЧӦ |
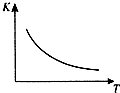 ��ҵ���Ʊ�H2��һ����Ҫ�����ǣ�CO��g��+H2O��g��?CO2 ��g��+H2��g����H=Q kJ/mol����֪�÷�Ӧ��ƽ�ⳣ��K���¶�T�Ĺ�ϵ��ͼ��ʾ������һ�̶��ݻ����ܱ������У�850��ʱ����������Ӧ����������ڸ����ʵ�Ũ�� ��mol/L����ʱ��ı仯��ϵ�������֪��850��ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ��K=1.0����ش��������⣺
��ҵ���Ʊ�H2��һ����Ҫ�����ǣ�CO��g��+H2O��g��?CO2 ��g��+H2��g����H=Q kJ/mol����֪�÷�Ӧ��ƽ�ⳣ��K���¶�T�Ĺ�ϵ��ͼ��ʾ������һ�̶��ݻ����ܱ������У�850��ʱ����������Ӧ����������ڸ����ʵ�Ũ�� ��mol/L����ʱ��ı仯��ϵ�������֪��850��ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ��K=1.0����ش��������⣺| ʱ��/min | CO��g�� | H2O��g�� | CO2��g�� | H2��g�� |
| 0 | 0.200 | 0.300 | 0 | 0 |
| 2 | 0.138 | 0.238 | 0.062 | 0.062 |
| 3 | c1 | c2 | c3 | c4 |
| 4 | c1 | c2 | c3 | c4 |
��2������850��ʱ��Ӧ�����г���H2O��g����K ֵ���䣨���������С�����䡱����
��3���ϱ��� c2Ϊ0.18mol/L��CO��g����ת����Ϊ60%��
| A�� | b-a=n+m | B�� | a-b=n-m | C�� | ������Y��X | D�� | �˵����Y��X |