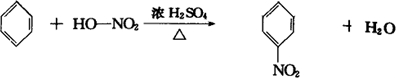
��Ŀ����
3����1��CH3+��-CH3��������CH3-������Ҫ���л���Ӧ�м��壬�й����ǵ�˵����ȷ����CDA�����Ǿ��ɼ���ȥ��һ����ԭ������
B�����ǻ�Ϊ�ȵ����壬̼ԭ�Ӿ���ȡsp2�ӻ�
C��CH3-��NH3��H3O+��Ϊ�ȵ����壬���ι��;�Ϊ������
D������CH3-��һ��CH3+��һ��CH3-��Ͼ��ɵõ�CH3CH3
��2��п��һ����Ҫ�Ľ�����п���仯�������Ź㷺��Ӧ�ã�
��������п[CH2OH��CHOH��4COO]2Zn��Ŀǰ�г������еIJ�п����д��Zn2+��̬�����Ų�ʽ1s22s22p63s23p63d10�������Ƿ���[CH2OHCHOH��CHOH��3COOH]��̼ԭ���ӻ���ʽ��sp2��sp3��
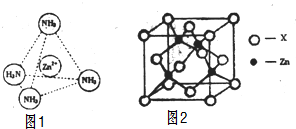
��3��Zn2+����NH3�γ�������[Zn��NH3��4]2+��Zn2+λ�������������ģ�NH3λ����������Ķ��㣬����ͼ1�б�ʾ��[Zn��NH3��4]2+��Zn2+��N֮��Ļ�ѧ����
��4����ͼ2��ʾп��ij�ǽ���Ԫ��X�γɵĻ����ᄃ��������Zn��Xͨ�����ۼ���ϣ��û�����Ļ�ѧʽΪZnX��
���� ��1��A������ȥ��һ����ԭ�Ӳ��ܵõ�CH3+��CH3-��
B��ԭ��������ȡ��۵����������������������ȵ�����Ϊ�ȵ����壻-CH3��������CH3-��Cԭ�Ӿ��γ�3���Ҽ�������̼ԭ����1�������ӣ�CH3-��Cԭ���ɶԹ¶Ե��ӣ��ӻ������Ŀ��Ϊ4��
C��CH3-��NH3��H3O+������4��ԭ�ӡ�10�����ӣ���Ϊ�ȵ����壬�ռ�ṹ���ƣ�
D������-CH3��һ��CH3+��CH3-��϶��ܵõ�CH3CH3��
��2��Znԭ�Ӻ�������Ų�ʽΪ1s22s22p63s23p63d104s2��ʧȥ4s�ܼ�2�������γ�Zn2+��
�Ȼ���Cԭ���γ�3���Ҽ�������̼ԭ���γ�4���Ҽ�����û�й¶Ե��ӣ��ӻ������ĿΪ�ֱ�Ϊ3��4��
��3��Zn2+�����пչ����NH3�����й¶Ե��ӣ�����ͨ����λ���γ�������[Zn��NH3��4]2+��
��4�����ݾ�̯�����㾧����Znԭ����Ŀ��Xԭ����Ŀ������ȷ����ѧʽ��
��� �⣺A��������ӱ��CH3+��-CH3��CH3-ʱ��ʧȥ�ķֱ����⸺���ӡ���ԭ�Ӻ������ӣ���A����
B��CH3+��-CH3��CH3-�ֱ����6����7����8���۵��ӣ����ǵȵ����壬-CH3��������CH3-��Cԭ�Ӿ��γ�3���Ҽ�������̼ԭ����1�������ӣ�CH3-��Cԭ���ɶԹ¶Ե��ӣ��ӻ������Ŀ��Ϊ4������̼ԭ�Ӳ�ȡsp3�ӻ�����B����
C��CH3-��NH3��H3O+������8���۵��ӡ�4��ԭ�ӣ���Ϊ�ȵ����壬���ι��;�Ϊ�����Σ���C��ȷ��
D������-CH3��һ��CH3+��CH3-��϶��ܵõ�CH3CH3����D��ȷ��
��ѡCD��
��2��Znԭ�Ӻ�������Ų�ʽΪ1s22s22p63s23p63d104s2��ʧȥ4s�ܼ�2�������γ�Zn2+��Zn2+��̬�����Ų�ʽΪ��1s22s22p63s23p63d10��
�Ȼ���Cԭ���γ�3���Ҽ�������̼ԭ���γ�4���Ҽ�����û�й¶Ե��ӣ��ӻ������ĿΪ�ֱ�Ϊ3��4��������̼ԭ���ӻ���ʽΪ��sp2��sp3�ӻ���
�ʴ�Ϊ��1s22s22p63s23p63d10��sp2��sp3��
��3��Zn2+�����пչ����NH3�����й¶Ե��ӣ�����ͨ����λ���γ�������[Zn��NH3��4]2+����ͼ��ʾ��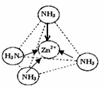 ��
��
�ʴ�Ϊ�� ��
��
��4��������Znԭ����ĿΪ4��Xԭ����ĿΪ8��$\frac{1}{8}$+6��$\frac{1}{2}$=4��Zn��Xԭ����Ŀ֮��Ϊ1��1���ʻ�ѧʽΪZnX��
�ʴ�Ϊ��ZnX��
���� �����Ƕ����ʽṹ�����ʵĿ��飬�漰��������Ų�ʽ���ռ乹�����ӻ���ʽ���жϡ��ȵ����塢������������ȣ�ּ�ڿ���ѧ���Ի���֪ʶ��������Ӧ�ã��Ѷ��еȣ�

| A�� | �����ܲ��������ЧӦ | B�� | ���岻��ͨ����ֽ | ||
| C�� | ������۲����� | D�� | ���岻�ȶ������ú����ײ������� |
| A�� | �������е���AgNO3��Һ�������е���Ԫ�أ�Br-+Ag+�TAgBr�� | |
| B�� | ʵ����һ���ô�ˮ�͵�ʯ����Ȳ��CaC2+2H2O�TC2H2��+Ca��OH��2 | |
| C�� | �ױ���Ũ�����Ũ����Ļ���ﷴӦ��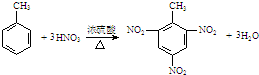 | |
| D�� | ʵ������Һ��ͱ����屽�� |
| A�� | ������ͨ������С�ڵ�����ʾ���ӵĶ��� | |
| B�� | ��ͬһ�ܼ����˶��ĵ��ӣ����˶�״̬�϶���ͬ | |
| C�� | �ܲ�����Խ��s�����Ƶİ뾶Խ�� | |
| D�� | ���ӽ��ڼ���̬ԾǨ����̬ʱ�Ż����ԭ�ӹ��� |
| A�� | HCO3-+H2O?H3O++CO32- | B�� | PO43-+3H2O?H3PO4+3OH- | ||
| C�� | NH4++H2O?NH3H2O+H+ | D�� | H2O+H2O?H3O++OH- |
| A�� | b=2a | B�� | V����ϩ��=0.5aL | C�� | n��H2O��=n��CO2�� | D�� | b=3a |
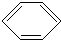 +Cl2
+Cl2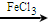
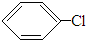 +HCl
+HCl