��Ŀ����
14����ͼ���߿��е�װ�ÿ���������Ũ������ľ̿���ڼ��������·�Ӧ������������������д���пհף�
��1����װ�ÿ�������֤�����е�H2O���ѧʽ������������ˮ����ͭ��Ϊ��ɫ��
��2����װ����A��Һ�����ó�ȥSO2 ����������Һ��ɫ��B��Һ�������Ǽ�����������Ƿ������
��3����װ���е������dz���ʯ��ˮ����ǣ���Ϊ����֤�����е�CO2���ѧΪʽ����
��4�������װ���Т١��ڡ�������������������˳���Ϊ�ڡ��١��ۣ��ҽ�װ��Aǰ���Ʒ����Һʡ�ԣ�����Լ����������CO2��SO2���ѧʽ�������ܼ����������H2O���ѧʽ�������ܼ����ԭ�����������Һ�д���ˮ��
���� ����������Ũ�����̼����������ԭ��Ӧ���ɶ�����������̼��ˮ����ˮ����ͭ���ڼ���ˮ��Ʒ�����ڼ�������������ö��������Ư���ԣ�A�и���������ڳ�ȥ��������B���ڼ�����������Ƿ������C�г���ʯ��ˮ���ڼ��������̼�����ɣ��ɹ۲쵽����ǣ��Դ˽����⣮
��� �⣺��1��װ�â�Ϊ��ˮ����ͭ���Լ���ˮ�Ĵ��ڣ��ɹ۲���ˮ����ͭ��Ϊ��ɫ���ʴ�Ϊ��H2O����ˮ����ͭ��Ϊ��ɫ��
��2�����������Һ�dz�ȥ���������ֹ���Ŷ�����̼�ļ��飬�ɹ۲쵽��Һ��ɫ��B���ڼ�����������Ƿ������
�ʴ�Ϊ����ȥSO2 ����Һ��ɫ��������������Ƿ������
��3�����пɹ۲쵽����ʯ��ˮ����ǣ���֤���ɶ�����̼���ʴ�Ϊ������ʯ��ˮ����ǣ�CO2��
��4�����������������˳���Ϊ�ڡ��ۡ��٣���ڿ��Լ���������Ƕ����������Խ���������������ۼ�������¶�����̼�����Ǣټ���ʱһ������ˮ����Щˮ��һ����ԭ����������е�ˮ��
�ʴ�Ϊ��CO2��SO2��H2O���������Һ�д���ˮ��
���� ���⿼��ѧ��Ũ����������еĺ�̼���ʵķ�Ӧ��Ϊ�߿��������ͺ�Ƶ���㣬��Ŀ�Ѷ��еȣ�ע������Ũ��������ʣ���ȷ����������õ��Լ��Լ�������Ⱥ�˳���ۺ��Խ�ǿ�����������ѧ���ķ�����������ѧʵ��������

 Сѧ��10���ӿ������100��ϵ�д�
Сѧ��10���ӿ������100��ϵ�д�| A�� | 4��3 | B�� | 4��5 | C�� | 5��4 | D�� | 1��1 |
����������ƽȷ��ȡ11.50g�Ȼ��ƹ���
����10mL��Ͳ��ȡ9.8mL����
��������ƿȷ����0.0121mol/L���Ȼ�����Һ100mL
���ù㷺pH��ֽ���ij��Һ��pHΪ5.6���������ݺ������ǣ�
| A�� | ȫ�� | B�� | �٢ڢ� | C�� | �ڢۢ� | D�� | �ڢ� |
| A�� | Ũ������лӷ��� | B�� | Ũ���������ˮ�� | ||
| C�� | Ũ�����Ũ�����е�ˮ���ö����� | D�� | Ũ��������ˮ�� |
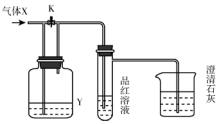 ��ͼ��һ�����������ʵ�ʵ��װ�ã���װ���л���ͨ������X�����رջ���K����Ʒ����Һ��ɫ���ݴ��ж�����X��ϴ��ƿ�ڵ���ҺY���ܵ�����ǣ�������
��ͼ��һ�����������ʵ�ʵ��װ�ã���װ���л���ͨ������X�����رջ���K����Ʒ����Һ��ɫ���ݴ��ж�����X��ϴ��ƿ�ڵ���ҺY���ܵ�����ǣ�������| A | B | C | D | |
| X | ���� | �������� | ������̼ | �Ȼ��� |
| Y | Ũ���� | ����NaHCO3 | Na2SO3��Һ | ����NaHSO3 |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ������ԭ��Ӧ | B�� | ������Ӧ | C�� | ��ԭ��Ӧ | D�� | �û���Ӧ |
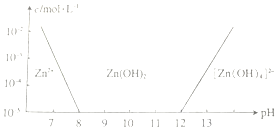 п���仯�������Ź㷺��Ӧ�ã���ش��й�п���仯������й����⣮
п���仯�������Ź㷺��Ӧ�ã���ش��й�п���仯������й����⣮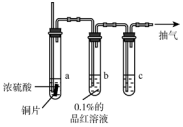 ijʵ��С��ͬѧΪ��̽��ͭ��Ũ����ķ�Ӧ���Խ̲��е�װ�ý��мĽ����ڴ��Թ�a�м���һ�����������ܣ���ͼ��ͼ�мг������ͼ�������û�л�������
ijʵ��С��ͬѧΪ��̽��ͭ��Ũ����ķ�Ӧ���Խ̲��е�װ�ý��мĽ����ڴ��Թ�a�м���һ�����������ܣ���ͼ��ͼ�мг������ͼ�������û�л�������