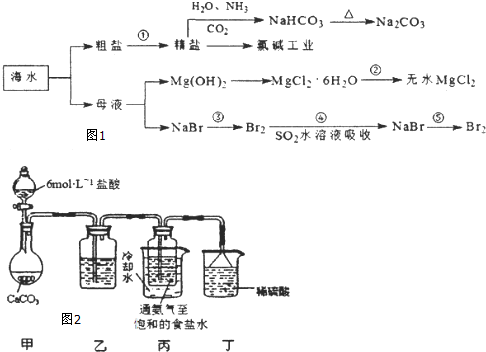
��Ŀ����
���л���ѧ������
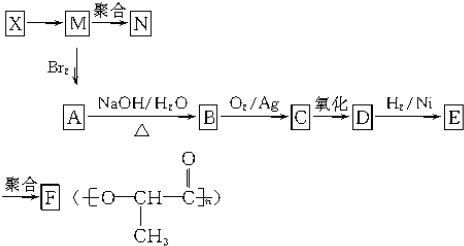
AΪ�л��ϳ��м��壬��һ�������·�����ȥ��Ӧ���ܵõ����ֻ�Ϊͬ���칹��IJ�����е�һ��B��������ȡ�ϳ���֬��Ⱦ�ϵȶ��ֻ�����Ʒ��A�ܷ�����ͼ1��ʾ�ı仯��
��֪����ע��R��R'Ϊ��������ԭ�ӣ�
�Իش�
��1��D�Ľṹ��ʽ�� ��
��2��E��F�ķ�Ӧ������ ��
��3��д��������������A��ͬ���칹��ṹ��ʽ ���������ԣ�H-NMR��ͼ����ͼ2����
��4��д��E���ɸ߾���Ļ�ѧ����ʽ ��
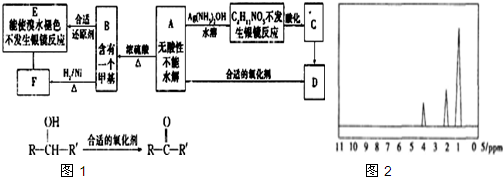
AΪ�л��ϳ��м��壬��һ�������·�����ȥ��Ӧ���ܵõ����ֻ�Ϊͬ���칹��IJ�����е�һ��B��������ȡ�ϳ���֬��Ⱦ�ϵȶ��ֻ�����Ʒ��A�ܷ�����ͼ1��ʾ�ı仯��
��֪����ע��R��R'Ϊ��������ԭ�ӣ�
�Իش�
��1��D�Ľṹ��ʽ��
��2��E��F�ķ�Ӧ������
��3��д��������������A��ͬ���칹��ṹ��ʽ
��4��д��E���ɸ߾���Ļ�ѧ����ʽ

���㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
������A�������Ҳ���ˮ�⣬��A�в����������Ȼ���A�ܷ���������Ӧ�����ɺ�4��Cԭ�ӵIJ����A�Ǻ���4��Cԭ�ӵ�ȩ����A�ܷ�����ȥ��Ӧ����A�к��д��ǻ���A������ȥ��Ӧ����B��B�к���һ������̼̼˫������B�Ľṹ��ʽΪ��CH3CH=CHCHO��A��һ�������·�����ȥ��Ӧ�����ܵõ����ֻ�Ϊͬ���칹��IJ����A�Ľṹ��ʽΪ��CH3CH��OH��CH2CHO��A��������Һ�������ữ����C��C�Ľṹ��ʽΪ��CH3CH��OH��CH2COOH��A�ͺ��ʵ���������Ӧ����D�����������Ϣ֪��D�Ľṹ��ʽΪ��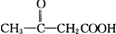 ��B�����������ӳɷ�Ӧ����F��F�Ľṹ��ʽΪ��CH3CH2CH2CH2OH��B����ԭ����E��E��ʹ��ˮ��ɫ��˵������̼̼˫����������������Ӧ��˵������ȩ������E�Ľṹ��ʽΪ��CH3CH=CHCH2OH���ݴ˷������
��B�����������ӳɷ�Ӧ����F��F�Ľṹ��ʽΪ��CH3CH2CH2CH2OH��B����ԭ����E��E��ʹ��ˮ��ɫ��˵������̼̼˫����������������Ӧ��˵������ȩ������E�Ľṹ��ʽΪ��CH3CH=CHCH2OH���ݴ˷������
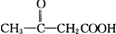 ��B�����������ӳɷ�Ӧ����F��F�Ľṹ��ʽΪ��CH3CH2CH2CH2OH��B����ԭ����E��E��ʹ��ˮ��ɫ��˵������̼̼˫����������������Ӧ��˵������ȩ������E�Ľṹ��ʽΪ��CH3CH=CHCH2OH���ݴ˷������
��B�����������ӳɷ�Ӧ����F��F�Ľṹ��ʽΪ��CH3CH2CH2CH2OH��B����ԭ����E��E��ʹ��ˮ��ɫ��˵������̼̼˫����������������Ӧ��˵������ȩ������E�Ľṹ��ʽΪ��CH3CH=CHCH2OH���ݴ˷���������
�⣺A�������Ҳ���ˮ�⣬��A�в����������Ȼ���A�ܷ���������Ӧ�����ɺ�4��Cԭ�ӵIJ����A�Ǻ���4��Cԭ�ӵ�ȩ����A�ܷ�����ȥ��Ӧ����A�к��д��ǻ���A������ȥ��Ӧ����B��B�к���һ������̼̼˫������B�Ľṹ��ʽΪ��CH3CH=CHCHO��A��һ�������·�����ȥ��Ӧ�����ܵõ����ֻ�Ϊͬ���칹��IJ����A�Ľṹ��ʽΪ��CH3CH��OH��CH2CHO��A��������Һ�������ữ����C��C�Ľṹ��ʽΪ��CH3CH��OH��CH2COOH��A�ͺ��ʵ���������Ӧ����D�����������Ϣ֪��D�Ľṹ��ʽΪ��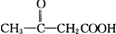 ��B�����������ӳɷ�Ӧ����F��F�Ľṹ��ʽΪ��CH3CH2CH2CH2OH��B����ԭ����E��E��ʹ��ˮ��ɫ��˵������̼̼˫����������������Ӧ��˵������ȩ������E�Ľṹ��ʽΪ��CH3CH=CHCH2OH��
��B�����������ӳɷ�Ӧ����F��F�Ľṹ��ʽΪ��CH3CH2CH2CH2OH��B����ԭ����E��E��ʹ��ˮ��ɫ��˵������̼̼˫����������������Ӧ��˵������ȩ������E�Ľṹ��ʽΪ��CH3CH=CHCH2OH��
��1���������Ϸ�����D�Ľṹ��ʽ��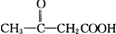 ���ʴ�Ϊ��
���ʴ�Ϊ��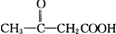 ��
��
��2��EΪCH3CH=CHCH2OH��FΪCH3CH2CH2CH2OH����E��F�ķ�ӦΪE�����������ӳɷ�Ӧ����F���ʴ�Ϊ���ӳɷ�Ӧ��
��3���������ԣ�˵�������Ȼ���H-NMR��ͼ��3�ַ壬��A��ͬ���칹��ṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��4��EΪCH3CH=CHCH2OH�����ɸ߾���Ļ�ѧ����ʽΪ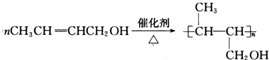 ��
��
�ʴ�Ϊ�� ��
��
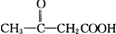 ��B�����������ӳɷ�Ӧ����F��F�Ľṹ��ʽΪ��CH3CH2CH2CH2OH��B����ԭ����E��E��ʹ��ˮ��ɫ��˵������̼̼˫����������������Ӧ��˵������ȩ������E�Ľṹ��ʽΪ��CH3CH=CHCH2OH��
��B�����������ӳɷ�Ӧ����F��F�Ľṹ��ʽΪ��CH3CH2CH2CH2OH��B����ԭ����E��E��ʹ��ˮ��ɫ��˵������̼̼˫����������������Ӧ��˵������ȩ������E�Ľṹ��ʽΪ��CH3CH=CHCH2OH����1���������Ϸ�����D�Ľṹ��ʽ��
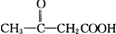 ���ʴ�Ϊ��
���ʴ�Ϊ��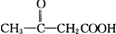 ��
����2��EΪCH3CH=CHCH2OH��FΪCH3CH2CH2CH2OH����E��F�ķ�ӦΪE�����������ӳɷ�Ӧ����F���ʴ�Ϊ���ӳɷ�Ӧ��
��3���������ԣ�˵�������Ȼ���H-NMR��ͼ��3�ַ壬��A��ͬ���칹��ṹ��ʽΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����4��EΪCH3CH=CHCH2OH�����ɸ߾���Ļ�ѧ����ʽΪ
 ��
���ʴ�Ϊ��
 ��
��
���������⿼���л�����ƶϣ����������Ϣ��Ϻϳ�·�߽����Ƶ�����ȷ�л���Ľṹ���������ǽⱾ��ؼ���֪��ͬ���칹��ĺ��壬�Ѷ��еȣ�

��ϰ��ϵ�д�
 ���100��1�ž�ϵ�д�
���100��1�ž�ϵ�д�
�����Ŀ
����ͼ��ʾװ�ý���ʵ�飬װ����ȷ����ƺ������ǣ�������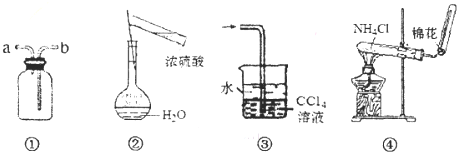

| A��װ�âٿ������ſ������ռ�NO���� |
| B������ͼ����ʾװ�ü���������һ�����ʵ���Ũ�ȵ����� |
| C��װ�âۿ����������Ȼ������ |
| D��ʵ���ҿ���װ�â���ȡNH3 |
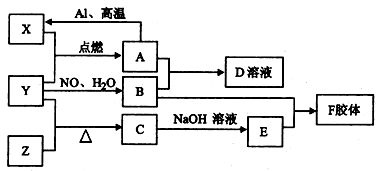
M��N��X��Y��������������ת����ϵ����Ӧ����������������ʡ�ԣ�������ѡ����ܵ��ǣ�������
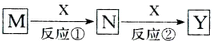

| A��M��Na�� X��O2 |
| B��M��HNO3 X��Fe |
| C��M��Al�� X��NaOH |
| D��M��NH3 X��O2 |
ϡ��ˮ�д�������ƽ�⣺NH3+H20?NH3?H2O?NH4++OH-�ı�������������ʹ����̶�������ǣ�������
| A����Ũ��ˮ |
| B�������¶� |
| C����NH4Cl��Һ |
| D����NaOH��Һ |