��Ŀ����
14���������岻��ȱ�ٵ���Ԫ�أ������������壨FeSO4•7H2O����ҽҩ������Ѫ����ij����С��ⶨ�ò�Ѫ����ÿƬb�ˣ�����Ԫ�صĺ�����ʵ�鲽�����£�
��ش��������⣺
��1���������H2O2�������ǽ�Fe2+ȫ������ΪFe3+��
��2����������õĺ��ɫ����Һ�г�H2O2����ˮ����Ҫ����Fe��OH��3�ͣ�NH4��2SO4���ѧʽ����
��3���������һϵ�д����IJ�������Ϊ���ˡ�ϴ�ӡ����ա���ȴ��������
��4��ʵ�������Ѿ�ȷ��ȡ��Ũ��������100mL 1mol/L��������Һ��������ʱ�õ����������ձ�������������������������100ml����ƿ����ͷ�ιܣ�
��5�����в����ᵼ�����ⶨ����Ԫ�غ���ƫ�ߵ���BC
A����������Ӱ�ˮ������ B�������ϴ�Ӳ���� C����������ղ����
��6����ʵ������ģ���ÿƬ��Ѫ������Ԫ�ص���������Ϊ$\frac{7a}{b}$%���ú���ĸ�Ĵ���ʽ��ʾ����
��7���ۺ�������������ˮ�ľ������仯ѧʽ�ɱ�ʾΪ[Fea��OH��b��SO4��n]mȡһ�������ۺ���������Ʒ���������ᷴӦ��������Һƽ����Ϊ���ݣ�һ����Һ�м���������BaCl2��Һ���õ���ɫ����1.7475g����һ����Һ���Ƚ�Fe3+��ԭΪFe2+������0.02000mol/LK2Cr2O7����Һ�ζ����յ㣬����K2Cr2O7����Һ50.00mL����������ۺ���������Ʒ��a��b�ı�ֵ�� ����֪��Cr2O72һʮ6Fe2+ʮ14H+�T2Cr3+ʮ6Fe3+ʮ7H2O����������̲����֣�
���� ������ͼ��֪����ʵ��ԭ��Ϊ����ҩƷ�е�Fe2+�γ���Һ����Fe2+����ΪFe3+��ʹFe3+ת��Ϊ����������������ת��Ϊ��������ͨ���ⶨ�����������������㲹Ѫ������Ԫ�صĺ�����
��1��˫��ˮ����ǿ�����ԣ�������ͼ��֪������˫��ˮĿ���ǽ�Fe2+ȫ������ΪFe3+��
��2�����ɫ����Ϊ��������������������ܹ��ͼ���İ�ˮ��������泥�
��3�������������ȶ������ֽ⣬������Һ�еij������ֽ����Ҫ������;�����ˡ�ϴ�ӡ����ա���ȴ��������
��4����������һ�����ʵ���Ũ�ȵ���Һ����ѡ��ʹ�õ�������
��5������������������ag�����10Ƭ��Ѫ������Ԫ�ص����ʵ������������ټ����ÿƬ��Ѫ������Ԫ�ص�����������
��6��A����ˮ���㣬���ɵ������������٣��������������٣�
B��ϴ�Ӳ���֣��ᵼ������������ƫ��
C�����ղ���֣�������������ƫ����Ԫ������ƫ�ߣ�
��7�������Ȼ������ɵij���Ϊ���ᱵ���ɼ������������ӵ����ʵ���������Cr2O72-+6Fe2++14H+=2Cr3++6Fe3++7H2O�͵ζ������ظ���ص����ɼ���������ӵ����ʵ��������ݵ���غ�ɼ�������������ӵ����ʵ������������������ӵ����ʵ���֮�ȼ�Ϊa��b�ı�ֵ��
��� �⣺������ͼ��֪����ʵ��ԭ��Ϊ����ҩƷ�е�Fe2+�γ���Һ����Fe2+����ΪFe3+��ʹFe3+ת��Ϊ����������������ת��Ϊ��������ͨ���ⶨ�����������������㲹Ѫ������Ԫ�صĺ�����
��1��˫��ˮ����ǿ�����ԣ�������ͼ��֪������˫��ˮĿ���ǽ�Fe2+ȫ������ΪFe3+��
�ʴ�Ϊ����Fe2+ȫ������ΪFe3+��
��2��������ͼ��֪��������ǽ�Fe3+ת��Ϊ�����������������백ˮ��Ӧ����Fe��OH��3 �ͣ�NH4��2SO4���ʴ�Ϊ��Fe��OH��3 �ͣ�NH4��2SO4��
��3���������һϵ�д�������������������Һ����ת��Ϊ����������Ҫ���ˡ�ϴ�ӵ�����������Ȼ��������������������ȴ�������������������
�ʴ�Ϊ�����ˣ�
��4����ȷ����100mL 1mol/L��������Һ������ʱ��Ҫ����������ƽ��ҩ�ס��������ձ�����ͷ�ιܡ�100mL����ƿ�����Ի���Ҫ100mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ��100mL����ƿ����ͷ�ιܣ�
��5��10Ƭ��Ѫ���������������ʵ���Ϊ��$\frac{a}{160}$mol�����е���Ԫ������Ϊ��2��$\frac{a}{160}$g=$\frac{7a}{10}$g��ÿƬ��Ѫ������Ԫ�ص���������Ϊ��$\frac{\frac{1}{10}��\frac{7a}{10}}{b}$��100%=$\frac{7a}{b}$%��
�ʴ�Ϊ��$\frac{7a}{b}$%��
��6��A����������Ӱ�ˮ�����㣬�����ӳ�������ȫ�����ɵ������������٣��������������٣��ⶨ���ƫ�ͣ���A����
B�������ϴ�Ӳ���֣��ᵼ��������������ƫ��õ���Ԫ����������ƫ��B��ȷ��
C����������ղ���֣��ᵼ�����������������ƫ����Ԫ����������ƫ��C��ȷ��
��ѡBC��
��7��n��SO42-��=$\frac{1.7475g}{233g/mol}$=0.0075mol��n��K2Cr2O7��=0.05L��0.02mol/=0.001mol��
n��Fe2+��=n��K2Cr2O7����6=0.006mol��
�ɵ���غ��֪n��OH-��+n��SO42-����2=n��Fe3+����3��n��OH-��=0.006mol��3-0.0075mol��2=0.003mol��
�õ�a��b=0.006mol��0.003mol=2��1��
�ʴ�Ϊ��2��1��
���� ���⿼��ѧ����ʵ��ԭ����ʵ����������⡢���ʷ����ᴿ��Ԫ�ػ��������ʡ�������ԭ��Ӧ�ζ�����ѧ����ȣ��Ѷ��еȣ����ʵ��ԭ���ǽ���Ĺؼ�����Ҫѧ���߱���ʵ�Ļ���֪ʶ���ۺ�����֪ʶ�������⡢��������������

| A�� | H2 | B�� | N2 | C�� | O2 | D�� | Cl2 |
| A�� | ���ƺ���ϴ�Ӽ���������ʹ�ã���ֹˮ�帻Ӫ����������ˮ��Դ | |
| B�� | ��ʯȼ���������ྻ��Դ�Ǽ����������Ч��ʩ | |
| C�� | ��ֱ�Ӽ����������ķ���ұ���õ��� | |
| D�� | Ǧ�������ڶ��ε�� |
| A�� | ���ʹ������������˺�̫���ܵ�� | |
| B�� | �ڳ����¿�������������������Ũ���� | |
| C�� | ʳ�����ȼҵ�Ļ���ԭ�� | |
| D�� | Na2O2����������ߵĹ����� |
�ף�Al$\stackrel{H_{2}SO_{4}}{��}$Al2��SO4��2$\stackrel{NaOH}{��}$Al��OH��3
�ң�Al$\stackrel{NaOH}{��}$NaAlO2$\stackrel{H_{2}SO_{4}}{��}$Al��OH��3
����
 ��Al��OH��3��
��Al��OH��3��| A�� | �ס������ĵ�ԭ��ͬ���� | B�� | �����ĵ�ԭ������ | ||
| C�� | �����ĵ�ԭ������ | D�� | �������ĵ�ԭ��ͬ���� |
| A�� | �����ȼ����Ϊ890.3 kJ•mol-1�������ȼ�յ��Ȼ�ѧ����ʽ�ɱ�ʾΪ��CH4��g��+2O2��g��=CO2��g��+2H2O��g����H=-890.3 kJ•mol-1 | |
| B�� | ��N2��g��+3H2��g��?2NH3��g����H=-92.4 kJ•mol-1�Ȼ�ѧ����ʽ��֪������Ӧ��ת��6NA����ʱ����Ӧ�ų�����С��92.4 kJ | |
| C�� | HCl��NaOH��Ӧ���к��ȡ�H=-57.3 kJ•mol-1����H2SO4��Ca��OH��2��Ӧ���к��ȡ�H=2����-57.3��kJ•mol-1 | |
| D�� | ��101 kPaʱ��2gH2��ȫȼ������Һ̬ˮ���ų�285.8kJ����������ȼ�յ��Ȼ�ѧ����ʽ��ʾΪ2H2��g��+O2��g��=2H2O��1����H=-571.6 kJ•mol-1 |
| A�� | CH4���ӵ����ģ�ͣ� | B�� | �Ҵ��Ľṹʽ�� | ||
| C�� | ��ϩ�Ľṹ��ʽ��CH2CH2 | D�� | ������ļ���ʽ��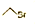 |
| A�� | ����������ˮ��Ӧʱ������0.1mol����ת�Ƶĵ�����Ϊ0.2NA | |
| B�� | 2L0.5mol/L̼������Һ�к��е�CO32-��ΪNA | |
| C�� | ��״����22.4LH2O�ķ�����ΪNA | |
| D�� | 18gD2O��18gH2O�к��еĵ�������Ϊ10NA |
| A�� | c��NH4+��=c��Na+��=c��OH-����c��NH3•H2O�� | B�� | c��NH4+����=c��Na+����c��NH3•H2O����c��OH-�� | ||
| C�� | c��NH4+������c��Na+����c��OH-����c��NH3•H2O�� | D�� | c��NH4+����c��Na+����c��NH3•H2O����c��OH-�� |