��Ŀ����
��1�����á���������������=����д���пո�
����֪��Zn��s��+CuSO4��aq���TZnSO4��aq��+Cu��s����H=-216kJ?mol-1����E��Ӧ�� E������
����A��B���������缫��������Һ���������Һ���γ�ԭ��أ�������A�������·����B���������������A B
�۶Ʋ���������ĸ�ʴ�ٶȱȽϣ���п���� ��������
���е��������pH��Ba��OH��2��NH3?H2O������Һ���õ�Ũ�ȵ��������ζ���ǡ���к�ʱ����ȥ�������ֱ�ΪV1��V2�����У�V1 V2
�ݳ����£����������pH�Ĵ����������Һ�У�c��CH3COO-�� c��SO42-��
��2���á���������С�����䡱��д���пո�
����ʯīΪ�缫���Na2SO4��Һʱ������������������Һ��pH ��
��ij�¶��£����淴ӦA��s��+B��g��?2C��g������H��0��ƽ�ⳣ��ΪK��������������ʱ���¶����ߣ�ƽ�ⳣ��K ��
��ˮ�ĵ���ƽ��ΪH2O?H+��OH-����H��0��������£���ˮ�м�������KOH���壬Kw ��
����0.10mol?L-1 ��ˮ�У���������NH4Cl������� NH3?H2O�ĵ���̶� ��
����AgCl����Һ�м������KI��Һ����ɫ����ת��Ϊ��ɫ�������������KI��Һ��ַ�Ӧ����Һ��Ksp��AgCl�� ��
����֪��Zn��s��+CuSO4��aq���TZnSO4��aq��+Cu��s����H=-216kJ?mol-1����E��Ӧ��
����A��B���������缫��������Һ���������Һ���γ�ԭ��أ�������A�������·����B���������������A
�۶Ʋ���������ĸ�ʴ�ٶȱȽϣ���п����
���е��������pH��Ba��OH��2��NH3?H2O������Һ���õ�Ũ�ȵ��������ζ���ǡ���к�ʱ����ȥ�������ֱ�ΪV1��V2�����У�V1
�ݳ����£����������pH�Ĵ����������Һ�У�c��CH3COO-��
��2���á���������С�����䡱��д���пո�
����ʯīΪ�缫���Na2SO4��Һʱ������������������Һ��pH
��ij�¶��£����淴ӦA��s��+B��g��?2C��g������H��0��ƽ�ⳣ��ΪK��������������ʱ���¶����ߣ�ƽ�ⳣ��K
��ˮ�ĵ���ƽ��ΪH2O?H+��OH-����H��0��������£���ˮ�м�������KOH���壬Kw
����0.10mol?L-1 ��ˮ�У���������NH4Cl������� NH3?H2O�ĵ���̶�
����AgCl����Һ�м������KI��Һ����ɫ����ת��Ϊ��ɫ�������������KI��Һ��ַ�Ӧ����Һ��Ksp��AgCl��
���㣺��Ӧ�Ⱥ��ʱ�,�����ĵ绯ѧ��ʴ�����,���������ˮ��Һ�еĵ���ƽ��,���ܵ���ʵ��ܽ�ƽ�⼰����ת���ı���,���ԭ��
ר�⣺�����������������
��������1���ٸ��ݷ��ȷ�Ӧ����Ӧ���������Ҫ��������������������жϣ�
����ԭ����У��������������ã�
����ԭ����У�������������
�ܰ�ˮ��������е��������pH��Ba��OH��2��NH3?H2O������Һ�У�NH3?H2O�����ʵ���Զ����Ba��OH��2��
�ݸ��ݵ���غ���������
��2������ʯīΪ�缫���Na2SO4��Һʱ��ͨ������OH-�ŵ����жϣ�
�߸�������Ӧ������������
��ˮ�����ӻ�Kw ֻ���¶ȵ�Ӱ�죻
���������NH4Cl�����������C��NH4+�����ݴ˷�����
��Ksp��AgCl��ֻ���¶ȵ�Ӱ�죮
����ԭ����У��������������ã�
����ԭ����У�������������
�ܰ�ˮ��������е��������pH��Ba��OH��2��NH3?H2O������Һ�У�NH3?H2O�����ʵ���Զ����Ba��OH��2��
�ݸ��ݵ���غ���������
��2������ʯīΪ�缫���Na2SO4��Һʱ��ͨ������OH-�ŵ����жϣ�
�߸�������Ӧ������������
��ˮ�����ӻ�Kw ֻ���¶ȵ�Ӱ�죻
���������NH4Cl�����������C��NH4+�����ݴ˷�����
��Ksp��AgCl��ֻ���¶ȵ�Ӱ�죮
���
�⣺��1�����ڷ��ȷ�Ӧ�У���Ӧ���������Ҫ��������������������ʴ�Ϊ������
����ԭ����У�������������������AΪ������BΪ��������ԭ����и��������������ã��ʴ�Ϊ������
����ԭ����У������������������п���������������������������������������������ױ���ʴ���ʴ�Ϊ������
�ܰ�ˮ��������������pH��Ba��OH��2��NH3?H2O������Һ�У�NH3?H2O�����ʵ���Զ����Ba��OH��2�����õ�Ũ�ȵ��������ζ���ǡ���к�ʱ����ȥ������NH3?H2O����Ϊ������
�ݳ����£����������pH�Ĵ����������Һ�У�C��H+����ͬ��C��OH-��Ҳ��ͬ�����ݵ���غ㣬�ڴ�����Һ�У�C��H+��=c��CH3COO-��+C��OH-������������Һ�У�C��H+��=2c��SO42-��+C��OH-������2c��SO42-��=c��CH3COO-������c��SO42-����c��CH3COO-�����ʴ�Ϊ������
��2������ʯīΪ�缫���Na2SO4��Һʱ��������������ˮ��OH-�ŵ磬����H+����pH��С���ʴ�Ϊ����С��
�߿��淴ӦA��s��+B��g��?2C��g����H��0������Ӧ���ȣ������¶ȣ�ƽ�����ƣ�ƽ�ⳣ��k��С���ʴ�Ϊ����С��
��ˮ�����ӻ�Kw ֻ���¶ȵ�Ӱ�죬�¶Ȳ��䣬Kw ���䣬�ʴ�Ϊ�����䣻
���������NH4Cl�����������C��NH4+����NH3?H2O�ĵ���ƽ�����ƣ�NH3?H2O�ĵ���̶ȼ�С���ʴ�Ϊ����С��
��Ksp��AgCl��ֻ���¶ȵ�Ӱ�죬�¶Ȳ��䣬Ksp���䣬�ʴ�Ϊ�����䣻
����ԭ����У�������������������AΪ������BΪ��������ԭ����и��������������ã��ʴ�Ϊ������
����ԭ����У������������������п���������������������������������������������ױ���ʴ���ʴ�Ϊ������
�ܰ�ˮ��������������pH��Ba��OH��2��NH3?H2O������Һ�У�NH3?H2O�����ʵ���Զ����Ba��OH��2�����õ�Ũ�ȵ��������ζ���ǡ���к�ʱ����ȥ������NH3?H2O����Ϊ������
�ݳ����£����������pH�Ĵ����������Һ�У�C��H+����ͬ��C��OH-��Ҳ��ͬ�����ݵ���غ㣬�ڴ�����Һ�У�C��H+��=c��CH3COO-��+C��OH-������������Һ�У�C��H+��=2c��SO42-��+C��OH-������2c��SO42-��=c��CH3COO-������c��SO42-����c��CH3COO-�����ʴ�Ϊ������
��2������ʯīΪ�缫���Na2SO4��Һʱ��������������ˮ��OH-�ŵ磬����H+����pH��С���ʴ�Ϊ����С��
�߿��淴ӦA��s��+B��g��?2C��g����H��0������Ӧ���ȣ������¶ȣ�ƽ�����ƣ�ƽ�ⳣ��k��С���ʴ�Ϊ����С��
��ˮ�����ӻ�Kw ֻ���¶ȵ�Ӱ�죬�¶Ȳ��䣬Kw ���䣬�ʴ�Ϊ�����䣻
���������NH4Cl�����������C��NH4+����NH3?H2O�ĵ���ƽ�����ƣ�NH3?H2O�ĵ���̶ȼ�С���ʴ�Ϊ����С��
��Ksp��AgCl��ֻ���¶ȵ�Ӱ�죬�¶Ȳ��䣬Ksp���䣬�ʴ�Ϊ�����䣻
�����������ۺϿ�����������ʵĵ���ƽ�⡢��ѧƽ����ƶ����ܽ�ƽ�ⳣ��ֻ���¶ȵ�Ӱ�죬�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
���и�������������ͬ���칹����ǣ�������
| A�����ʯ��C60 |
B��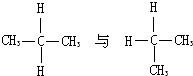 |
C��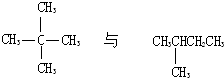 |
| D��CH4��CH3CH3 |
���¶ȡ��ݻ���ͬ�� 3 ���ܱ������У�����ͬ��ʽͶ�뷴Ӧ����ֺ��¡����ݣ���÷�Ӧ�ﵽƽ��ʱ���й��������£�
����֪N2��g��+3H2 ��g��?2NH3��g����H=-92.4kJ?mol-1��
����˵������ȷ���ǣ�������
����֪N2��g��+3H2 ��g��?2NH3��g����H=-92.4kJ?mol-1��
| ���� | �� | �� | �� |
| ��Ӧ��Ͷ���� | 1mol N2��3mol H2 | 2mol NH3 | 4mol NH3 |
| NH3 ��Ũ�ȣ�mol?L-1 �� | c1 | c2 | c3 |
| ��Ӧ�������仯 | �ų� akJ | ���� bkJ | ���� ckJ |
| ��ϵѹǿ��Pa�� | p1 | p2 | p3 |
| ��Ӧ��ת���� | ��1 | ��2 | ��3 |
| A��2p2��p3 |
| B��a+b=92.4 |
| C��2c1��c3 |
| D����1+��2=1 |
����ʱ��Ũ�ȶ�Ϊ0.1mol/L��HA��HC��HD����һԪ����Һ������HA��Һ��c��H+��=0.01mol/L��HC��pHֵΪ3��HD��Һ��c��OH-��=10-13mol/L�����������Ӧ�����������ʵ���Ũ�Ⱥ��¶���ͬ�������£���Һ��pHֵ�ɴ�С��˳��Ϊ��������
| A��NaA��NaC��NaD |
| B��NaC��NaA��NaD |
| C��NaD��NaA��NaC |
| D��NaC��NaD��NaA |
�����й��������ʵıȽ��У���ȷ���ǣ�������
�����ȶ��ԣ�CH4��NH3��H2O
�ڻ�ԭ�ԣ�I-��Br-��Cl-
�����ԣ�H3PO4��H2SO4��HClO4
�ܷе㣺HI��HBr��HCl��HF��
�����ȶ��ԣ�CH4��NH3��H2O
�ڻ�ԭ�ԣ�I-��Br-��Cl-
�����ԣ�H3PO4��H2SO4��HClO4
�ܷе㣺HI��HBr��HCl��HF��
| A���٢� | B���ڢ� | C���٢� | D��ֻ�Т� |
����˵����ȷ���ǣ�������
| A�������ӳ��������壬����109��28�䣬1mol�������ۼ�6mol |
| B���������ӳ�ֱ���ͣ��������ij�����������������ԭ������ͬ |
| C��S8�Ƿ��Ӿ��壬8����ԭ����ͬһ��ƽ���ϣ�Ħ������Ϊ256g |
| D��ʯī����ʲ�״�ṹ��ÿ��̼ԭ��ֻ��3���۵����γɹ��ۼ� |