��Ŀ����
7�����������ǵij��д������㣬�������ǵĽ�������Σ���������еĻҳ������ᡢ����ȿ�������ɵ����ܽ�ϵͳ��ɵ��Ӿ��ϰ��Ľ�������ˮ������Ӿ硢����ʪ������ʱ�����ͻ�ת��Ϊ���������й�˵������ȷ���ǣ�������| A�� | �����쳵������Ĺ���ͨ·���ڶ�������� | |
| B�� | ������̼����������Ϳ���������������������Ҫ�ɷ� | |
| C�� | ����Ķ��죬�����ϵĻ��ð����������ˮ���� | |
| D�� | ���ٻ��������Ͽ��ֵ�����ЧӦ�����������ķ��� |
���� A����ɢ������ֱ����10-9m��10-7m֮��ķ�ɢϵ���ڽ��壬����ɷ�Ϊ���ܽ���Һ�ܽ������ܽ��������������ܽ��������ơ��̵ȣ�
B��������̼�ǿ����ijɷ֣�
C��ˮ�ӷ���
D�����ֿ��Ե�ס�����еĿ����
��� �⣺A���������ڳ��������ܽ�����һ��ǿ��ͨ����Һ����Թ۲쵽���������A��ȷ��
B��������̼�ǿ����ijɷ֣����������ijɷ֣���B����
C��ˮ�ӷ���ð����������ˮ��������C��ȷ��
D�����ֿ��Ե�ס�����еĿ����������Ӧ���ٻ�������D��ȷ��
��ѡB��
���� ������Ҫ���������������ʵ�����ǽ���Ĺؼ���ע��֪ʶ�Ļ��ۺͽ�������ʣ���Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
17����״���£������ΪV L��Բ����ƿ�г��������������ں�������ˮ��ˮ���У���ͼ��ʾ����ʵ����Ϻ���������ƿ��ˮ����ȡ����������Һ���ܶ�Ϊ��g•cm-3������˵��������ǣ�������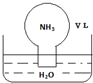

| A�� | ˮ���ջ����������ƿ | |
| B�� | ������Һ�����ʵ���Ũ��Ϊ$\frac{1}{22.4}$ mol•L-1 | |
| C�� | ������Һ����������Ϊ$\frac{17}{22400}$�� | |
| D�� | ������Һ�У�n��NH3•H2O��+n��NH4+��=$\frac{V}{22.4}$mol |
18���������ݻ���ͬ���ܱ������У�һ��ʢ��HCl���壬��һ��ʢ��H2��Cl2�Ļ�����壬��ͬ��ͬѹ�£��������ڵ�����һ�����ܾ�����ͬ�ģ�������
| A�� | �ܶ� | B�� | ��ɫ | C�� | ԭ���� | D�� | ������ |
19����������һ�����オ�����ϣ�����Ľṹ��ʽΪHOCH��CH3��COOH������˵����ȷ���ǣ�������
| A�� | ���������Է���������72 | |
| B�� | ����ķ���ʽ��C2H6O3 | |
| C�� | 1mol����������Ľ����ƿɷų�����44.8L����״���� | |
| D�� | 1mol�����������̼��������Һ��Ӧ�ɷų�������̼22.4L����״���� |
 A��B��C��D��E��F����ѧ��ѧ���������ʣ�����֮��ת����ϵ��ͼ��ʾ������A��BΪ���ʣ�FΪ��ɫ������DΪ���壮
A��B��C��D��E��F����ѧ��ѧ���������ʣ�����֮��ת����ϵ��ͼ��ʾ������A��BΪ���ʣ�FΪ��ɫ������DΪ���壮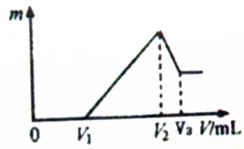 ��0.1mol��þ�������������2mol/L H2SO4��Һ�У�Ȼ���ٵμ�0.1mol/L NaOH��Һ����ش�
��0.1mol��þ�������������2mol/L H2SO4��Һ�У�Ȼ���ٵμ�0.1mol/L NaOH��Һ����ش�