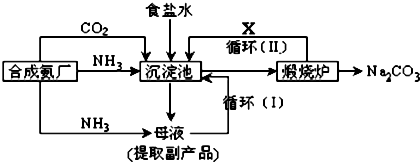
��Ŀ����
19��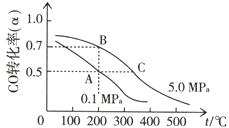 ���ྻú�������о����������൱�ձ飬������Աͨ�������ú������¯�н�����������ˮ�����ķ�����������������ֵ�ߴ�122500��16000kJ•m-3��ú̿��������Ҫ�ɷ���CO��H2��CO��H2����Ϊ��Դ�ͻ���ԭ�ϣ�Ӧ��ʮ�ֹ㷺��
���ྻú�������о����������൱�ձ飬������Աͨ�������ú������¯�н�����������ˮ�����ķ�����������������ֵ�ߴ�122500��16000kJ•m-3��ú̿��������Ҫ�ɷ���CO��H2��CO��H2����Ϊ��Դ�ͻ���ԭ�ϣ�Ӧ��ʮ�ֹ㷺����1����֪��C��s��+O2��g��=CO2��g����H1=-393.5kJ•mol-1��
2H2��g��+O2��g��=2H2O��g����H2=-483.6kJ•mol-1��
C��s��+H2O��g��=CO��g��+H2��g����H3=+131.3kJ•mol-1��
��ӦCO��g��+H2��g��+O2��g��=H2O��g��+CO2��g������H=-524.8kJ•mol-1��
��״���µ�ú̿����CO��H2��33.6L��������ȫ��Ӧ����CO2��H2O����Ӧ������ת��3mol e-��
��2�������¶�650���������ȼ�ϵ�أ�����ú̿����CO��H2��������ȼ����������CO2�Ļ������Ϊ����ȼ������һ��������Li2CO3��Na2CO3���۵�����������ʣ��Խ�������ȼ�ϼ���Ϊ�����Ƴɵģ������������尴���ʵ���֮��Ϊ1��1���뷴Ӧ���õ缫��ӦʽΪO2+4e-+2CO2=2CO32-��
��3���ܱ������г���10mol CO��20mol H2���ڴ��������·�Ӧ���ɼ״���CO��g��+2H2��g��?CH3OH��g����CO��ƽ��ת���ʣ��������¶ȡ�ѹǿ�Ĺ�ϵ��ͼ��ʾ��
����A��B�����ʾ��ijʱ�̴ﵽ��ƽ��״̬����ʱ��A��ʱ���������ΪVAL������¶��µ�ƽ�ⳣ��K=$\frac{{V}^{2}}{100}$L2/mol2��A��B����ʱ�����У�n��A������n��B����=5��4��
����A��C���㶼��ʾ�ﵽ��ƽ��״̬�����Է�Ӧ��ʼ����ƽ��״̬�����ʱ��tA����tC������ڡ�����С�ڡ����ڡ�����
���ڲ��ı䷴Ӧ������������£�Ϊ���CO��ת���ʿɲ�ȡ�Ĵ�ʩ�ǽ��¡���ѹ�����״��ӻ����ϵ�з��������
���� ��1�������Ȼ�ѧ����ʽ��˹����������õ�������n=$\frac{V}{22.4}$�������ʵ�����ϻ�ѧ����ʽ�ĵ���ת�Ƽ��㣻
��2��ȼ�ϵ����ȼ���ڸ���ʧ���ӷ���������Ӧ���������������õ����ӷ�����ԭ��Ӧ��
��3����A��B����ͬ�¶��µ�ƽ�⣬ת���ʱ仯����ƽ�ⳣ�����䣬��ϻ�ѧƽ������ʽ��ʽ����A����ƽ�ⳣ����A��һ����̼ת����Ϊ50%��B��һ����̼ת����Ϊ70%��
��C���¶ȸ����ʴ�ﵽƽ������Ҫ��ʱ��̣�
�����CO��ת���ʿɲ�ȡ�Ĵ�ʩ�Ǹı�������ʹƽ��������У�
��� �⣺��1��C��s��+O2��g��=CO2��g����H1=-393.5kJ/mol��
C��s��+H2O��g��=CO��g��+H2��g����H2=+131.3kJ/mol��
�ɸ�˹���ɢ�-�ڵõ�CO��g��+H2��g��+O2��g��=H2O��g��+CO2��g����H=-524.8KJ/mol��
�ڱ�״���£�33.6L��ú̿�ϳ������ʵ���Ϊ1.5mol����������ȫ��Ӧ����CO2��H2O����Ӧ������2mol�ϳ�����ȫ��Ӧ����ת��4mol������1.5mol�ϳ�����Ӧת�Ƶ���3mol��
�ʴ�Ϊ��-524.8�� 3��
��2��ȼ�ϵ����ȼ���ڸ���ʧ���ӷ���������Ӧ���������������õ����ӷ�����ԭ��Ӧ��������CO2�Ļ������Ϊ����ȼ������һ��������Li2CO3��Na2CO3���۵�����������ʣ��Խ�������ȼ�ϼ���Ϊ�����Ƴɵģ�������Ӧ�������õ����ӷ�����ԭ��Ӧ���缫��ӦΪ��O2+4e-+2CO2=2CO32-��
�ʴ�Ϊ��O2+4e-+2CO2=2CO32-��
��3��CO��g��+2H2��g��?CH3OH��g����ͼ������¶�Խ�ߣ�һ����̼ת���ʼ�С��������У�����Ӧ�Ƿ��ȷ�Ӧ��
������ͼ�������֪AB��ͬ�¶��µ�ƽ�⣬ƽ�ⳣ�����¶ȱ仯������ƽ�ⳣ�����䣬A��һ����̼ת����Ϊ50%��B��һ����̼ת����Ϊ70%���ܱ������г���10mol CO��20mol H2��A��ƽ�ⳣ�����ƽ������ʽ��ʽ���㣻
CO��g��+2H2 ��g��?CH3OH��g��
��ʼ����mol�� 10 20 0
�仯����mol�� 5 10 5
ƽ������mol�� 5 10 5
ƽ�ⳣ��K=$\frac{\frac{5mol}{VL}}{\frac{5mol}{VL}����\frac{10mol}{VL}��^{2}}$=$\frac{{V}^{2}}{100}$L2/mol2��
B��һ����̼ת����Ϊ70%�����ƽ������ʽ��ʽ����
CO��g��+2H2 ��g��?CH3OH��g��
��ʼ����mol�� 10 20 0
�仯����mol�� 7 14 7
ƽ������mol�� 3 6 7
A��B����ʱ�����У�n��A���ܣ�n��B����=��5+10+5������3+6+7��=20��16=5��4��
�ʴ�Ϊ��$\frac{{V}^{2}}{100}$L2/mol2��5��4��
�ڴﵽA��C�����ƽ��״̬�����ʱ�䣬C���¶ȸ߷�Ӧ���ʿ�ﵽ��Ӧ������Ҫ��ʱ��̣�tA��tC��
�ʴ�Ϊ�����ڣ�
�۷�Ӧ�����������С�ķ��ȷ�Ӧ�����CO��ת���ʿɲ�ȡ�Ĵ�ʩ�ǽ��¡���ѹ��������״���
�ʴ�Ϊ�����¡���ѹ�����״��ӻ����ϵ�з��������
���� ���⿼�����Ȼ�ѧ����ʽ��˹���ɵļ���Ӧ�ã���ѧƽ���־�жϣ�ͼ�������Ӱ��ƽ������ط����жϣ���ѧƽ���ƶ�ԭ���ǽ���ؼ�����Ŀ�Ѷ��еȣ�

| A�� | ���д���Cl-����Һ�п϶�������Ag+ | |
| B�� | AgI����Һ�м�������KI��ĩ��ƽ��AgI��s��?Ag+��aq��+I-��aq�������ƶ�����Һ�����ӵ���Ũ�Ȼ��С | |
| C�� | AgCl����Һ������KI������c��I-��=$\frac{{K}_{sp}��AgI��}{{K}_{sp}��AgCl��}$mol•L-1ʱ��AgCl��ʼ��AgI����ת�� | |
| D�� | ��Ũ�Ⱦ�Ϊ0.01 mol•L-1��KCl��KI�Ļ����Һ�еμ�AgNO3��Һ����Cl-��ʼ����ʱ����Һ��I-��Ũ��Ϊ1.25��10-8 mol•L-1 |
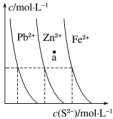 ��ͼΪij�¶��£�PbS��s����ZnS��s����FeS��s���ֱ�����Һ�дﵽ�����ܽ�ƽ�����Һ��S2-Ũ�ȡ�����������Ũ�ȱ仯���������˵����ȷ���ǣ�������
��ͼΪij�¶��£�PbS��s����ZnS��s����FeS��s���ֱ�����Һ�дﵽ�����ܽ�ƽ�����Һ��S2-Ũ�ȡ�����������Ũ�ȱ仯���������˵����ȷ���ǣ�������| A�� | ������������ܶȻ�Ksp��FeS������С | |
| B�� | ��������ֳ����м����ᣬ�����ܽ����PbS | |
| C�� | �������ɵ�ZnS��Һ�е�����������ͬŨ�ȵ�Pb2+��Fe2+����Һ����ZnS������ת��ΪPbS���� | |
| D�� | ��������������������a���Ӧ�Ķ��Dz�������Һ |
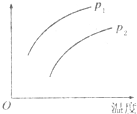 ���ݻ�һ�����ܱ������з������淴Ӧ��A��g��+2B��g��?2C��g������H��0����ʾ���ȣ���ƽ���ƶ���ϵ��ͼ��ʾ������˵����ȷ���ǣ�������
���ݻ�һ�����ܱ������з������淴Ӧ��A��g��+2B��g��?2C��g������H��0����ʾ���ȣ���ƽ���ƶ���ϵ��ͼ��ʾ������˵����ȷ���ǣ�������| A�� | P1��P2��������ָC���������� | |
| B�� | P1��P2��������ָA���������� | |
| C�� | P1��P2��������ָA��ת���� | |
| D�� | P1��P2��������ָ��������ƽ��Ħ������ |
| ʱ��/min Ũ��/��mol/L�� | 0 | 10 | 20 | 30 | 40 | 50 |
| NO | 2.0 | 1.16 | 0.80 | 0.80 | 0.96 | 0.96 |
| N2 | 0 | 0.42 | 0.60 | 0.60 | 0.72 | 0.72 |
| CO2 | 0 | 0.42 | 0.60 | 0.60 | 0.72 | 0.72 |
��2��30min��ֻ�ı�ijһ�����������ϱ��������жϸı������������BC��˫ѡ��
A��������ʵĴ��� B���ʵ���С���������
C��ͨ��һ������NO D������һ�����Ļ���̿
��3����30min�������¶���T2�棬�ﵽƽ��ʱ��������NO��N2��CO2��Ũ��֮��Ϊ5��3��3����ﵽ��ƽ��ʱNO��ת���ʽ��� ������ߡ����͡�������H��0���������������
| A�� | ����12.8gCuSO4�ܽ���100mLˮ�� | |
| B�� | ����25.0gCuSO4•5H2O��������100mLˮ�� | |
| C�� | ����16.0gCuSO4����ˮ��Ȼ��ϡ����100mL | |
| D�� | ����20.0gCuSO4•5H2O��������ˮ��Ȼ��ϡ����100mL |
��H++Z-+XO42-��X2++Z2+H2O��δ��ƽ����
��2M2++R2�T2M3++2R-
��2R-+Z2�TR2+2Z-��
�ɴ��ж�����˵����ȷ���ǣ�������
| A�� | ������ǿ��˳��ΪXO42-��Z2��R2��M3+ | |
| B�� | Z2�ڢ��з���������Ӧ | |
| C�� | RԪ���ڷ�Ӧ���б��������ڢ��б���ԭ | |
| D�� | �����·�Ӧ���ɽ���2M2++Z2�T2M3++2Z- |
��1����֪��C��s��+H2O��g��=CO��g��+H2��g����H=+131.3kJ��mol-l
CO2��g��+H2��g��=CO��g��+H2O��g����H=+41.3kJ��mol-l
��̼��ˮ������Ӧ���ɶ�����̼���������Ȼ�ѧ����ʽΪC��s��+2H2O��g��=CO2��g��+2H2��g����H=+90.0kJ��mol-1���÷�Ӧ�ڸ��£�����¡��������¡����κ��¶ȡ����������������Է����У�
��2������̿��ԭ���ɴ������������NO�ȣ��������ķ�ӦΪC��s��+2NO��g��?N2��g��+CO2 ��g������ij�ܱ������м���һ�����Ļ���̿��NO����T1��ʱ����ͬʱ���õĸ������Ũ�������ʾ��
| ʱ�䣨min�� Ũ�ȣ�mol•L-1�� ���� | 0 | 10 | 20 | 30 | 40 | 50 |
| NO | 1.00 | 0.68 | 0.50 | 0.50 | 0.60 | 0.60 |
| N2 | 0 | 0.16 | 0.25 | 0.25 | 0.30 | 0.30 |
| CO2 | 0 | 0.16 | 0.25 | 0.25 | 0.30 | 0.30 |
��30min��ֻ�ı�ijһ��������Ӧ���´ﵽƽ�⣬�����ϱ��е������жϸı������������AD������ĸ����
A��ͨ��һ������NO B������һ�����Ļ���̿
C��������ʵĴ��� D���ݵ���С���������
��3���о���������ӦCO��g��+H2O��g��?H2��g��+CO2��g����ƽ�ⳣ�����¶ȵı仯�����ʾ��
| �¶�/�� | 400 | 500 | 800 |
| ƽ�ⳣ��K | 9.94 | 9 | 1 |