��Ŀ����
ij��ѧ��ȤС���ú�����������ͭ�ĺϽ���ȡ�������Ȼ�����Һ���̷�����͵������壬��̽����ҵ���ϵ������á���ʵ�鷽�����£�
�ش��������⣺
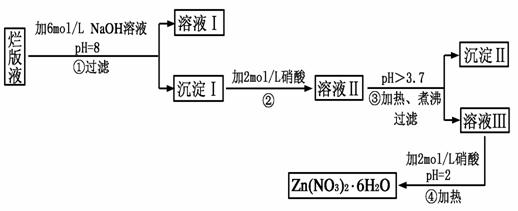
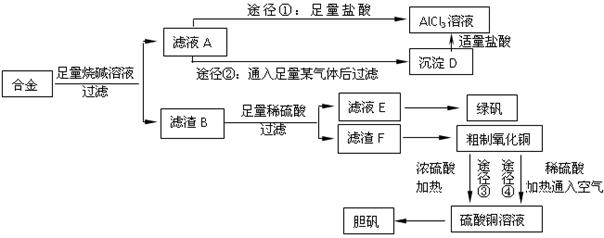
��1��д���Ͻ����ռ���Һ��Ӧ�����ӷ���ʽ ��������Ϊ�Ͻ����ռ���Һ�γ���ԭ��أ�����Ϊԭ��ظ����������� ��
��2������ҺA��AlCl3��Һ��;���Тٺ͢����֣�����Ϊ�������� ������ʵ�鷽���ദ�����˹��˲������������õ��IJ��������� �Ͳ����������в������������� ��
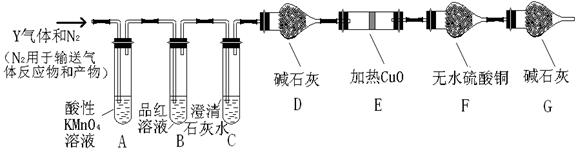
��3���ô�������ͭͨ������;����ȡ��������;������ȣ�;�������Ծ��е������ŵ���: �� ��
��4��ͨ��;����ʵ���ô�������ͭ��ȡ������������е�ʵ��������裺���ܡ�����ͨ���������ˡ� ����ȴ�ᾧ�� ����Ȼ������С�����ͨ���������������Ϊ �������ӷ���ʽ��ʾ����
��5���ڲⶨ���õ�����CuSO4��xH2O���нᾧˮxֵ��ʵ������У������������ٽ��� �Ρ����ⶨ���xֵƫ�ߣ����ܵ�ԭ���� ��
a�������¶ȹ��� b����������Ŀ����ϴ�
c�����Ⱥ���ڿ�������ȴ d���������岿�ַ绯
e������ʱ��������ɽ����� f��������������δ�����ʪ��

�ش��������⣺
��1��д���Ͻ����ռ���Һ��Ӧ�����ӷ���ʽ ��������Ϊ�Ͻ����ռ���Һ�γ���ԭ��أ�����Ϊԭ��ظ����������� ��
��2������ҺA��AlCl3��Һ��;���Тٺ͢����֣�����Ϊ�������� ������ʵ�鷽���ദ�����˹��˲������������õ��IJ��������� �Ͳ����������в������������� ��
��3���ô�������ͭͨ������;����ȡ��������;������ȣ�;�������Ծ��е������ŵ���: �� ��
��4��ͨ��;����ʵ���ô�������ͭ��ȡ������������е�ʵ��������裺���ܡ�����ͨ���������ˡ� ����ȴ�ᾧ�� ����Ȼ������С�����ͨ���������������Ϊ �������ӷ���ʽ��ʾ����
��5���ڲⶨ���õ�����CuSO4��xH2O���нᾧˮxֵ��ʵ������У������������ٽ��� �Ρ����ⶨ���xֵƫ�ߣ����ܵ�ԭ���� ��
a�������¶ȹ��� b����������Ŀ����ϴ�
c�����Ⱥ���ڿ�������ȴ d���������岿�ַ绯
e������ʱ��������ɽ����� f��������������δ�����ʪ��
��1��2Al+2OH��+6H2O��2[Al��OH��4]��+3H2�� ��2�֣���Al��1�֣�
��2��;���ڣ�2�֣����ձ���©����2�֣���������1�֣�
��3��������������;�������������٣�1�֣�;���ܲ��������Ⱦ���������壨1�֣�
��4������Ũ����1�֣����ˣ�1�֣�2Cu+O2+4H+��2Cu2++2H2O��2�֣�
��5��4��2�֣���6��aef��2�֣�
��2��;���ڣ�2�֣����ձ���©����2�֣���������1�֣�
��3��������������;�������������٣�1�֣�;���ܲ��������Ⱦ���������壨1�֣�
��4������Ũ����1�֣����ˣ�1�֣�2Cu+O2+4H+��2Cu2++2H2O��2�֣�
��5��4��2�֣���6��aef��2�֣�
��

��ϰ��ϵ�д�
�����Ŀ
|
����ʵ���������������ȷ���ǣ� ��  �ٽ�NaNO3��KCl�Ļ��Һ���Ȳ�Ũ�����о������������ȹ���ʱ���ɷ����NaCl���� �ں����е�Ԫ�صķ��뼰����ʱ����Ҫ���ҵĽ�ȡҺ�м�������ϡ�������������Һ ��ֽ���������������Ӻ�ͭ����ʵ���У����������ֽ�����ɺ��ܽ������չ������ �ܾ���������������ˮ������������Na2CO3��Һ��FeSO4��Һ����ʹ������Һ���� ���ü��ȷ����Է��������غ͵ⵥ�ʵĻ�����Ϊ�ⵥ������������ ������һ�����ʵ���Ũ�ȵ���Һʱ������ȡҺ̬���ʵ���Ͳ��ˮϴ�ӣ�ϴ��Һ��������ƿ �߽�������ˮ�ε����۵⻯����ֽ�ϳ�����ͼ����˵����Ũ����ˮ�ܽ�I-������I2����Ũ����ˮ�ܽ�I2��һ�������ɵ�Ļ�����  �ཫ����CO2ͨ��Ca(ClO)2��Һ�ó�����Һ��˵��H2CO3�����Ա�HClO�� 
|