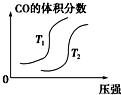
��Ŀ����
8����ͼΪ����250.0mL0.2molLNa2CO3��Һ���̵�ʾ��ͼ���ش��������⣺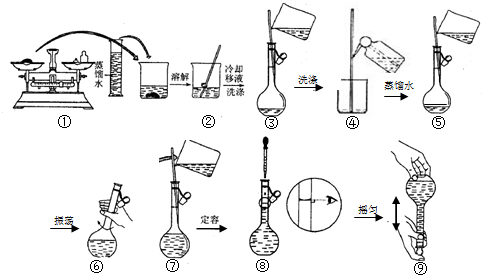
��1��ʵ��ʱҪ�õ��������У��ձ�����������������ƽ��ҩ�ס���Ͳ��250mL����ƿ����ͷ�ιܣ�
��2���ڢڢ�����ʵ�鶼�õ����������������÷ֱ��ǽ��衢�����ܽ⣻��������ֹ��Һ�⽦��
��3�����ƹ����У���������������ȷ�����в���������Ũ��ƫ�����C��
A������ƿ��ԭ������������ˮ��B��û�н��в�������ܺ͢ݣ�C���ڢಽ�۲�Һ��ʱ���ӿ̶��ߣ�D�������ˮ�����˿̶��ߣ�ȡ������ˮʹҺ��ǡ�õ��̶���
��4���ɼ����֪���ڳ��������У�����������ƽ����Na2CO3���������Ϊ5.3g������5mol•L -1Na2CO3��Һ���Ƹ���Һ����Ӧ����Ͳ��ȡ����Һ�����Ϊ10.0mL��
���� ��1���������Ʋ����Ǽ��㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��������Ҫ��������
��2������������Һ���ܽ����Һ�����в��������ý��
��3���������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$������������
��4������m=CVM������Ҫ���ʵ�����������ϡ���������ʵ����ʵ������������ҪŨ��Һ�������
��� �⣺��1�����������м��㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣨������Ͳ��ȡˮ�����ձ��������ò��������裬�����ܽ⣮��ȴ��ת�Ƶ�250mL����ƿ�У����ò�����������ϴ���ձ���������2-3�Σ�����ϴ��Һ��������ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȣ���������������������ƽ��ҩ�ס��ձ�����������250mL����ƿ����ͷ�ιܣ������ṩ��������֪����������250mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ��250mL����ƿ����ͷ�ιܣ�
��2�����������ܽ�ʱ���ã����衢�����ܽ⣻��Һʱ���ã���������ֹ��Һ�⽦��
�ʴ�Ϊ�����裻��ֹ��Һ�⽦��
��3��A������ƿ��ԭ������������ˮ�������ʵ����ʵ�������Һ�����������Ӱ�죬��ҺŨ�Ȳ��䣬��A��ѡ��
B��û�н��в�������ܺ͢ݣ��������ʵ����ʵ���ƫС����ҺŨ��ƫ�ͣ���B��ѡ��
C���ڢಽ�۲�Һ��ʱ���ӿ̶��ߣ�������Һ���ƫС����Һ�����ʵ���Ũ��ƫ�ߣ���Cѡ��
D�������ˮ�����˿̶��ߣ�ȡ������ˮʹҺ��ǡ�õ��̶��ߣ��������ʵ����ʵ���ƫС����Һ�����ʵ���Ũ��ƫ�ͣ���D��ѡ��
�ʴ�Ϊ��C��
��4������250.0mL0.2molLNa2CO3��Һ����Ҫ̼���Ƶ�����=0.2mol/L��0.25mol��106g/mol=5.3g��
����ҪŨ̼������Һ�����ΪV����������Һϡ���ɵã�V��5mol/L=0.2mol/L��250mL�����V=10.0mL��
�ʴ�Ϊ��5.3g��10.0mL��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���ȷ����ԭ���Ͳ��������ǽ���ؼ���ע������C=$\frac{n}{V}$�����������ķ�������Ŀ�ѶȲ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ��������ƽ����8.70gʳ�� | |
| B�� | �ù㷺pH��ֽ�ⶨ��ˮ��pHֵΪ3.5 | |
| C�� | ����������������������������� | |
| D�� | �ü��ȵķ��������Ȼ��ƺ��Ȼ�淋Ĺ������� |
| A�� | ԭ����Ŀ��ȵ��������壬����������Z | |
| B�� | ��ͬ�����£�ͬ�������������壬�����ܶ���С����X | |
| C�� | ��һ�������£��������������Ϊ2.24L�������ǵ����ʵ���һ����Ϊ0.1mol | |
| D�� | ͬ���£������ͬ���������ֱ��2gY�����1gZ���壬����ѹǿ��Ϊ2��1 |
| A�� | �������� | B�� | ���� | C�� | ���� | D�� | ֲ���� |
| A�� | �Ҵ������ѻ�Ϊͬ���칹�� | |
| B�� | ����ú���Եõ����顢���Ͱ�����Ҫ����ԭ�� | |
| C�� | �Ҵ�������֮���ܷ���������Ӧ��������Ӧ��������Ӧ��Ϊ�淴Ӧ | |
| D�� | �����������£�����ˮ��Ļ�ѧ����ʽΪ C12H22O11+H2O��2C6H12O6�������ǣ� |
| A�� | �ᴿ�������ʵĹ�ҵ�Ҵ������õķ��������� | |
| B�� | �����£���������̼ͨ�뱽������Һ����Һ������ | |
| C�� | ������ɳ�ı����ᣬ��ȥ���ʵķ����ǽ����ܽ����ˣ��������ᾧ���� | |
| D�� | ���ͱ���ϡ��Һ�ֱ���Ũ��ˮ��ϣ����߲�����ɫ���� |
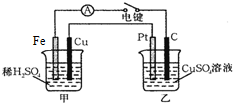 ԭ��غ͵��ض���ʵ���������û����ش��������⣺
ԭ��غ͵��ض���ʵ���������û����ش��������⣺
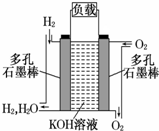 ����Ϊ�ı�δ�������ʮ���¿Ƽ�֮һ��ȼ�ϵ�ؾ�������Ⱦ������������Ч �ʵ��ص㣮��ͼΪ����ȼ�ϵ�صĽṹʾ��ͼ���������ҺΪKOH ��Һ���缫����Ϊ���ɶ��ʯī�����������������ֱ��� �����ϵش�����������ͨ��ȼ�ϵ��ʱ������ڱպϻ�·�в��ϵز����������Իش��������⣺
����Ϊ�ı�δ�������ʮ���¿Ƽ�֮һ��ȼ�ϵ�ؾ�������Ⱦ������������Ч �ʵ��ص㣮��ͼΪ����ȼ�ϵ�صĽṹʾ��ͼ���������ҺΪKOH ��Һ���缫����Ϊ���ɶ��ʯī�����������������ֱ��� �����ϵش�����������ͨ��ȼ�ϵ��ʱ������ڱպϻ�·�в��ϵز����������Իش��������⣺ ��
��