��Ŀ����
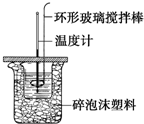 ������ͼ��ʾװ�òⶨ�к��ȵ�ʵ�鲽�����£�
������ͼ��ʾװ�òⶨ�к��ȵ�ʵ�鲽�����£�������Ͳ��ȡ50mL 0.50mol?L-1���ᵹ��С�ձ��У���������¶ȣ�������һ��Ͳ��ȡ50mL 0.55mol?L-1NaOH��Һ������ͬһ�¶ȼƲ�����¶ȣ��۽�NaOH��Һ����С�ձ��У��跨ʹ֮��Ͼ��ȣ���û��Һ����¶ȣ��ش��������⣺
��1��Ϊʲô����NaOH��ҺҪ�Թ�����
��2������NaOH��Һ����ȷ������
a���ز������������� b��һ��Ѹ�ٵ��� c����������������
��3��ʹ������NaOH��Һ��Ͼ��ȵ���ȷ������
A�����¶ȼ�С�Ľ���
B���ҿ�ӲֽƬ�ò���������
C����������ձ�
D���������¶ȼ��ϵĻ��β����������������½���
��4���ֽ�һ������ϡ����������Һ��ϡ����������Һ��ϡ��ˮ�ֱ��1L 1mol?L-1��ϡ����ǡ����ȫ��Ӧ���䷴Ӧ�ȷֱ�Ϊ��H1����H2����H3�����H1����H2����H3�Ĵ�С��ϵΪ
��5���������������������Һ���ܶȶ���1g?cm-3����֪��H=
| (m��+m��)?c?(t��-tʼ) |
| n |
c=4.18J?g-1?��-1=4.18��10-3 kJ?g-1?��-1��nΪ����H2O�����ʵ�����
Ϊ�˼����к��ȣ�ijѧ��ʵ���¼�������£�
| ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�¶�t2/�� | |
| ���� | ������Һ | ����������Һ | |
| 1 | 20.0 | 20.1 | 23.2 |
| 2 | 20.2 | 20.4 | 23.4 |
| 3 | 20.5 | 20.6 | 23.6 |
���㣺�к��ȵIJⶨ
ר�⣺��ѧ��Ӧ�е������仯
��������1��Ϊ�˱�֤����Ǽ��е�һ����ȫ��Ӧ��������֤һ��������
��2����NaOH��Һ����С�ձ��У��ּ��ε��룬�ᵼ������ɢʧ��Ӱ��ⶨ�����
��3��������������ƻ��ʱ���������¶ȼ��ϵĻ��β������������ؽ�����ʹ������NaOH��Һ��Ͼ��ȣ�
��4��H+��aq��+OH-��aq���TH2O��l������H=-57.3kJ?mol-1���к�����ǿ��ǿ��ϡ��Һ��ȫ��Ӧ����1molˮ�ų���������������ʴ��ڵ���ƽ�⣬������������ȹ��̣�
��5�������к��ȼ��㹫ʽQ=cm��T���漰��δ֪���ݽ����жϣ�
��2����NaOH��Һ����С�ձ��У��ּ��ε��룬�ᵼ������ɢʧ��Ӱ��ⶨ�����
��3��������������ƻ��ʱ���������¶ȼ��ϵĻ��β������������ؽ�����ʹ������NaOH��Һ��Ͼ��ȣ�
��4��H+��aq��+OH-��aq���TH2O��l������H=-57.3kJ?mol-1���к�����ǿ��ǿ��ϡ��Һ��ȫ��Ӧ����1molˮ�ų���������������ʴ��ڵ���ƽ�⣬������������ȹ��̣�
��5�������к��ȼ��㹫ʽQ=cm��T���漰��δ֪���ݽ����жϣ�
���
�⣺��1��Ϊ��ȷ�����ᱻ��ȫ�кͣ�����NaOH��ҺҪ�Թ������ʴ�Ϊ��ȷ�����ᱻ��ȫ�кͣ�
��2����������������Һʱ������һ��Ѹ�ٵĵ��룬Ŀ���Ǽ���������ɢʧ�����ּܷ��ε�������������Һ������ᵼ������ɢʧ��Ӱ��ⶨ�����
��ѡb��
��3��ʹ������NaOH��Һ��Ͼ��ȵ���ȷ���������ǣ��������¶ȼ��ϵĻ��β������������ؽ������¶ȼ��Dz����¶ȵģ�����ʹ���¶ȼƽ��裻Ҳ������������ձ���������ܵ���Һ�彦��������ɢʧ��Ӱ��ⶨ����������ܴ�ӲֽƬ�ò��������裬�����������ɢʧ��
��ѡD��
��4���к�����ǿ��ǿ��ϡ��Һ��ȫ��Ӧ����1molˮ�ų���������һ������ϡ����������Һ��ϡ����������Һ��1L 1mol?L-1��ϡ����ǡ����ȫ��Ӧ����57.3kJ��һˮ�ϰ���������ʣ����ڵ���ƽ�⣬������������ỵ̄�ϡ��ˮ��1L 1mol?L-1��ϡ����ǡ����ȫ��Ӧ����С��57.3kJ����Ӧ�ʱ��Ǹ�ֵ�����ԡ�H1=��H2����H3��
�ʴ�Ϊ����H1=��H2����H3��
��5����1��ʵ�������NaOH��Һ��ʼƽ���¶�Ϊ20.05�棬��Ӧ���¶�Ϊ��23.2�棬��Ӧǰ���¶Ȳ�Ϊ��3.15�棻
��2��ʵ�������NaOH��Һ��ʼƽ���¶�Ϊ20.3�棬��Ӧ���¶�Ϊ��23.4�棬��Ӧǰ���¶Ȳ�Ϊ��3.1�棻
��3��ʵ�������NaOH��Һ��ʼƽ���¶�Ϊ20.55�棬��Ӧ���¶�Ϊ��23.6�棬��Ӧǰ���¶Ȳ�Ϊ��3.05�棻
50mL��0.50mol/L������50mL��0.55mol/L����������Һ��������Ϊm=100mL��1g/cm3=100g��c=4.18J/��g?�棩�����빫ʽQ=cm��T������0.025mol��ˮ�ų�����Q=4.18J/��g?�棩��100g��
=1.036kJ��������0.025mol��ˮ�ų�����Ϊ��1.2959kJ����������1mol��ˮ�ų�����Ϊ1.2959kJ��
=51.8kJ������ʵ���õ��к��ȡ�H=-51.8kJ/mol��
�ʴ�Ϊ��-51.8kJ/mol��
��2����������������Һʱ������һ��Ѹ�ٵĵ��룬Ŀ���Ǽ���������ɢʧ�����ּܷ��ε�������������Һ������ᵼ������ɢʧ��Ӱ��ⶨ�����
��ѡb��
��3��ʹ������NaOH��Һ��Ͼ��ȵ���ȷ���������ǣ��������¶ȼ��ϵĻ��β������������ؽ������¶ȼ��Dz����¶ȵģ�����ʹ���¶ȼƽ��裻Ҳ������������ձ���������ܵ���Һ�彦��������ɢʧ��Ӱ��ⶨ����������ܴ�ӲֽƬ�ò��������裬�����������ɢʧ��
��ѡD��
��4���к�����ǿ��ǿ��ϡ��Һ��ȫ��Ӧ����1molˮ�ų���������һ������ϡ����������Һ��ϡ����������Һ��1L 1mol?L-1��ϡ����ǡ����ȫ��Ӧ����57.3kJ��һˮ�ϰ���������ʣ����ڵ���ƽ�⣬������������ỵ̄�ϡ��ˮ��1L 1mol?L-1��ϡ����ǡ����ȫ��Ӧ����С��57.3kJ����Ӧ�ʱ��Ǹ�ֵ�����ԡ�H1=��H2����H3��
�ʴ�Ϊ����H1=��H2����H3��
��5����1��ʵ�������NaOH��Һ��ʼƽ���¶�Ϊ20.05�棬��Ӧ���¶�Ϊ��23.2�棬��Ӧǰ���¶Ȳ�Ϊ��3.15�棻
��2��ʵ�������NaOH��Һ��ʼƽ���¶�Ϊ20.3�棬��Ӧ���¶�Ϊ��23.4�棬��Ӧǰ���¶Ȳ�Ϊ��3.1�棻
��3��ʵ�������NaOH��Һ��ʼƽ���¶�Ϊ20.55�棬��Ӧ���¶�Ϊ��23.6�棬��Ӧǰ���¶Ȳ�Ϊ��3.05�棻
50mL��0.50mol/L������50mL��0.55mol/L����������Һ��������Ϊm=100mL��1g/cm3=100g��c=4.18J/��g?�棩�����빫ʽQ=cm��T������0.025mol��ˮ�ų�����Q=4.18J/��g?�棩��100g��
| 3.15��+3.1��+3.05�� |
| 3 |
| 1mol |
| 0.025mol |
�ʴ�Ϊ��-51.8kJ/mol��
���������⿼�����к��ȵIJⶨ���������㡢����������Ŀ�ѶȲ���ע�����ղⶨ�к��ȵ���ȷ��������ȷʵ����������йؼ����ھ����ܼ�������ɢʧ��ʹ�ⶨ�������ȷ��

��ϰ��ϵ�д�
�����Ŀ
��֪X��Y��Z��W��T�Ƕ�������ԭ���������������5������Ԫ�أ�����Y��X����Ԫ�ؿ������1��1��1��2��1��3��1��4�ȶ��ֻ���� Zԭ�������������Ǵ�����������3����W��Xͬ���壬W��T�γɵĻ�����WT�dz�������ĵ�ζƷ������˵����ȷ���ǣ�������
| A��ԭ�Ӱ뾶��T��W��Z��Y |
| B��W��T�ĵ��ʿ�ͨ����ⱥ�͵�WTˮ��Һ��� |
| C��Z��W�γɵ����ֻ������л�ѧ��������ȫ��ͬ |
| D��Ԫ��X��Z��T���γɶ����� |
�����ױ���ʴ������Ҫԭ���ǣ�������
| A������һ������� |
| B�����Ľ�����Ա�þ�� |
| C�������нϸ��۵� |
| D�������γ����ܵ������ﱡĤ |
���������в���ȷ���ǣ�������
| A���嵥����ǿ�����ԣ��ж������Կ���������Ѭ������ɱ���� |
| B��Һ��Ӧ������ĥ�ڲ�������ɫϸ���Լ�ƿ�У���������ˮ����ˮ�� |
| C���������ھƾ����������þƾ��ӵ�ˮ�н�����ȡ���� |
| D���ü��ȵķ����ɽ����л��е�����ɳ�ӳ�ȥ |
�����������ڻ�ԭ���ǵ��ǣ�������
| A������ | B�������� |
| C������ | D������ |
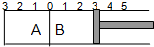 ��ͼ��ʾ���¶Ȳ����ij�����ָ���A��B�����֣�A�����ݻ��̶����䣬B�п��ƶ��Ļ���������A�г���2molSO2��1molO2����B�г���2molSO3��1molN2������ͬ�����·������淴Ӧ��2SO2��g��+O2��g��?2SO3��g����
��ͼ��ʾ���¶Ȳ����ij�����ָ���A��B�����֣�A�����ݻ��̶����䣬B�п��ƶ��Ļ���������A�г���2molSO2��1molO2����B�г���2molSO3��1molN2������ͬ�����·������淴Ӧ��2SO2��g��+O2��g��?2SO3��g����