题目内容
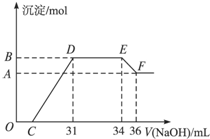 (2012?上海模拟)某同学取一定量的Al和Fe固体混合物,与2.0L极稀的硝酸充分反应,反应过程中无气体放出.在反应结束后的溶液中,逐滴加入4mol/L的氢氧化钠溶液,所加氢氧化钠溶液的体积(mL)与产生的沉淀的物质的量(mol)的关系如图所示.试回答下列问题:
(2012?上海模拟)某同学取一定量的Al和Fe固体混合物,与2.0L极稀的硝酸充分反应,反应过程中无气体放出.在反应结束后的溶液中,逐滴加入4mol/L的氢氧化钠溶液,所加氢氧化钠溶液的体积(mL)与产生的沉淀的物质的量(mol)的关系如图所示.试回答下列问题:(1)图中OC段没有沉淀生成,此阶段发生反应的离子方程式为
H++OH-=H2O
H++OH-=H2O
.在DE段,沉淀的物质的量没有变化,则此阶段发生反应的离子方程式为NH4++OH-=NH3?H2O
NH4++OH-=NH3?H2O
;上述现象说明溶液中Al3+、Fe3+
Al3+、Fe3+
结合OH-的能力比NH4+
NH4+
强(填写离子符号).(2)B与A的差值为
0.008
0.008
mol.(3)B点对应的沉淀的物质的量为
0.032
0.032
mol,C点对应的氢氧化钠溶液的体积为7
7
mL.(4)求原硝酸溶液的物质的量浓度?
分析:一定量的Al和Fe固体混合物,与2.0L极稀的硝酸充分反应,反应过程中无气体放出,说明硝酸被还原性为NH4+,反应后加入NaOH,OC段没有生成沉淀,应发生H++OH-=H2O,CD段反应生成Fe(OH)3、Al(OH)3,DE段反应为NH4++OH-=NH3?H2O,EF段发生Al(OH)3+NaOH=NaAlO2+2H2O,以此解答该题.
解答:解:(1)OC段没有沉淀生成,此阶段发生反应的离子方程式为H++OH-=H2O,CD段反应生成Fe(OH)3、Al(OH)3,在DE段,沉淀的物质的量没有变化,则此阶段发生反应的离子方程式为NH4++OH-=NH3?H2O,说明Al3+、Fe3+结合OH-的能力比NH4+强,
故答案为:H++OH-=H2O;NH4++OH-=NH3?H2O;Al3+、Fe3+;NH4+;
(2)EF段发生Al(OH)3+NaOH=NaAlO2+2H2O,此时n(NaOH)=0.002L×4mol/L=0.008mol,由方程式可知n(EF段发生Al(OH)3+NaOH=NaAlO2+2H2O,)=0.008mol,
故答案为:0.008;
(3)DE段反应为NH4++OH-=NH3?H2O,此时n(NH4+)=n(NaOH)=0.003L×4mol/L=0.012mol,则硝酸得到电子为0.012mol×8=0.096mol,氧化剂和还原剂得失电子数目相等,所以铁和铝的总物质的量为
mol=0.032mol,
生成Fe(OH)3、Al(OH)3,需要0.096molNaOH,体积应为
=0.024L,即24mL,而完全沉淀时需要31mLNaOH溶液,说明有7mLNaOH发生中和反应,即C为7,
故答案为:0.032;7;
(4)E点时溶液溶质为NaNO3,同时生成0.012molNH3?H2O,则由Na守恒可知n(NaNO3)=n(NaOH)=0.034L×4mol/L=0.136mol,
由N守恒可知原硝酸溶液中硝酸的物质的量为0.136mol+0.012mol=0.148mol,
则原硝酸溶液的物质的量浓度为
=0.074mol/L,
答:原硝酸溶液的物质的量浓度为0.074mol/L.
故答案为:H++OH-=H2O;NH4++OH-=NH3?H2O;Al3+、Fe3+;NH4+;
(2)EF段发生Al(OH)3+NaOH=NaAlO2+2H2O,此时n(NaOH)=0.002L×4mol/L=0.008mol,由方程式可知n(EF段发生Al(OH)3+NaOH=NaAlO2+2H2O,)=0.008mol,
故答案为:0.008;
(3)DE段反应为NH4++OH-=NH3?H2O,此时n(NH4+)=n(NaOH)=0.003L×4mol/L=0.012mol,则硝酸得到电子为0.012mol×8=0.096mol,氧化剂和还原剂得失电子数目相等,所以铁和铝的总物质的量为
| 0.096 |
| 3 |
生成Fe(OH)3、Al(OH)3,需要0.096molNaOH,体积应为
| 0.096mol |
| 4mol/L |
故答案为:0.032;7;
(4)E点时溶液溶质为NaNO3,同时生成0.012molNH3?H2O,则由Na守恒可知n(NaNO3)=n(NaOH)=0.034L×4mol/L=0.136mol,
由N守恒可知原硝酸溶液中硝酸的物质的量为0.136mol+0.012mol=0.148mol,
则原硝酸溶液的物质的量浓度为
| 0.148mol |
| 2L |
答:原硝酸溶液的物质的量浓度为0.074mol/L.
点评:本题考查混合物的计算,题目难度较大,注意硝酸的还原产物为解答该题的关键,根据反应的图象判断各段反应.

练习册系列答案
 天天向上一本好卷系列答案
天天向上一本好卷系列答案 小学生10分钟应用题系列答案
小学生10分钟应用题系列答案
相关题目
 (2012?上海模拟)“绿色化学实验”进课堂,某化学教师为“氯气与金属钠反应”设计了如下装置与操作以替代相关的课本实验.实验操作:钠与氯气反应的装置可作如图改进,将一根玻璃管与氯气发生器相连,玻璃管内放一块黄豆粒大的金属钠(已吸净煤油),玻璃管尾部塞一团浸有NaOH溶液的棉花球.先给钠预热,到钠熔融成圆球时,撤火,通入氯气,即可见钠着火燃烧,生成大量白烟.以下叙述错误的是( )
(2012?上海模拟)“绿色化学实验”进课堂,某化学教师为“氯气与金属钠反应”设计了如下装置与操作以替代相关的课本实验.实验操作:钠与氯气反应的装置可作如图改进,将一根玻璃管与氯气发生器相连,玻璃管内放一块黄豆粒大的金属钠(已吸净煤油),玻璃管尾部塞一团浸有NaOH溶液的棉花球.先给钠预热,到钠熔融成圆球时,撤火,通入氯气,即可见钠着火燃烧,生成大量白烟.以下叙述错误的是( )