��Ŀ����
A��B��C��D��E������NH4Cl��Ba��OH��2��KCl��K2SO4����NH4��2SO4��ɫ��Һ�е�һ�֣��������������ʱ�����������ǣ�
��A��B��Ϻ������ɫ���������Ⱥ�����������
��B��C���Ҳ������ɫ���������Ⱥ����������������ʹʪ��ĺ�ɫʯ����ֽ������
��B��E��Ϻ������������Ⱥ�Ҳ����ʹʪ��ĺ�ɫʯ����ֽ���������壮
��D���κ�һ����Һ��Ϻ������Ա仯�������������ش��������⣺
��1��A�� ��B�� ��C�� ��D�� ��E�ĵ���ʽ ��
��2��д���йط�Ӧ�����ӷ���ʽ��
A+B�� ��
B+C�� ��
B+E�� ��
��A��B��Ϻ������ɫ���������Ⱥ�����������
��B��C���Ҳ������ɫ���������Ⱥ����������������ʹʪ��ĺ�ɫʯ����ֽ������
��B��E��Ϻ������������Ⱥ�Ҳ����ʹʪ��ĺ�ɫʯ����ֽ���������壮
��D���κ�һ����Һ��Ϻ������Ա仯�������������ش��������⣺
��1��A��
��2��д���йط�Ӧ�����ӷ���ʽ��
A+B��
B+C��
B+E��
���㣺���ʵļ���ͼ����ʵ�鷽�����,���ӷ���ʽ����д
ר�⣺���ӷ�Ӧר��
���������������������ʷ�Ӧ���ɰ�ɫ������ֻ��Ba��OH��2����BΪBa��OH��2��
��A��B��ϣ�������ɫ����������ʱ�̼����������ɣ�˵��AΪK2SO4��
��B��C���Ҳ������ɫ������������ʱ�д̼����������ɣ��Ҹ�������ʹʪ��ĺ�ɫʯ����ֽ������������Ϊ��������CΪ��NH4��2SO4��
��B��E��ϣ��������������Ⱥ������ʹʪ��ĺ�ɫʯ����ֽ���������壬EӦΪNH4Cl������DΪKCl��Ȼ�������ʵ����ʼ���ѧ���������
��A��B��ϣ�������ɫ����������ʱ�̼����������ɣ�˵��AΪK2SO4��
��B��C���Ҳ������ɫ������������ʱ�д̼����������ɣ��Ҹ�������ʹʪ��ĺ�ɫʯ����ֽ������������Ϊ��������CΪ��NH4��2SO4��
��B��E��ϣ��������������Ⱥ������ʹʪ��ĺ�ɫʯ����ֽ���������壬EӦΪNH4Cl������DΪKCl��Ȼ�������ʵ����ʼ���ѧ���������
���
�⣺��1�����������������ʷ�Ӧ���ɰ�ɫ������ֻ��Ba��OH��2����BΪBa��OH��2��
��A��B��ϣ�������ɫ����������ʱ�̼����������ɣ�˵��AΪK2SO4��
��B��C���Ҳ������ɫ������������ʱ�д̼����������ɣ��Ҹ�������ʹʪ��ĺ�ɫʯ����ֽ������������Ϊ��������CΪ��NH4��2SO4��
��B��E��ϣ��������������Ⱥ������ʹʪ��ĺ�ɫʯ����ֽ���������壬EӦΪNH4Cl�������ʽΪ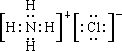 ������DΪKCl��
������DΪKCl��
�ʴ�Ϊ��K2SO4��Ba��OH��2����NH4��2SO4��KCl��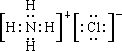 ��
��
��2��AΪK2SO4��BΪBa��OH��2�����߷�Ӧ�Ļ�ѧ����ʽΪK2SO4+Ba��OH��2=BaSO4��+2KOH���������ӷ���ʽΪBa2++SO42-=BaSO4����
BΪBa��OH��2��CΪ��NH4��2SO4�����߷�Ӧ�Ļ�ѧ����ʽΪBa��OH��2+��NH4��2SO4=BaSO4��+2NH3��+2 H2O���������ӷ���ʽΪBa2++2OH-+2NH4++SO42-=BaSO4��+2NH3?H2O��
BΪBa��OH��2��EΪNH4Cl�����߷�Ӧ�Ļ�ѧ����ʽΪBa��OH��2+2NH4Cl
BaCl2+2NH3��+2 H2O�����ӷ���ʽΪOH-+NH4+
NH3��+H2O��
�ʴ�Ϊ��Ba2++SO42-=BaSO4����Ba2++2OH-+2NH4++SO42-=BaSO4��+2NH3?H2O��OH-+NH4+
NH3��+H2O��
��A��B��ϣ�������ɫ����������ʱ�̼����������ɣ�˵��AΪK2SO4��
��B��C���Ҳ������ɫ������������ʱ�д̼����������ɣ��Ҹ�������ʹʪ��ĺ�ɫʯ����ֽ������������Ϊ��������CΪ��NH4��2SO4��
��B��E��ϣ��������������Ⱥ������ʹʪ��ĺ�ɫʯ����ֽ���������壬EӦΪNH4Cl�������ʽΪ
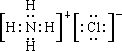 ������DΪKCl��
������DΪKCl���ʴ�Ϊ��K2SO4��Ba��OH��2����NH4��2SO4��KCl��
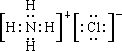 ��
����2��AΪK2SO4��BΪBa��OH��2�����߷�Ӧ�Ļ�ѧ����ʽΪK2SO4+Ba��OH��2=BaSO4��+2KOH���������ӷ���ʽΪBa2++SO42-=BaSO4����
BΪBa��OH��2��CΪ��NH4��2SO4�����߷�Ӧ�Ļ�ѧ����ʽΪBa��OH��2+��NH4��2SO4=BaSO4��+2NH3��+2 H2O���������ӷ���ʽΪBa2++2OH-+2NH4++SO42-=BaSO4��+2NH3?H2O��
BΪBa��OH��2��EΪNH4Cl�����߷�Ӧ�Ļ�ѧ����ʽΪBa��OH��2+2NH4Cl
| ||
| ||
�ʴ�Ϊ��Ba2++SO42-=BaSO4����Ba2++2OH-+2NH4++SO42-=BaSO4��+2NH3?H2O��OH-+NH4+
| ||
���������⿼�����ʵļ���ͼ���ʵ�鷽������ƣ�Ϊ��Ƶ���㣬�ۺϿ���Ԫ�ػ�����֪ʶ�������ڿ���ѧ���ۺ����û�ѧ֪ʶ��������ע����ݷ�Ӧ����������жϣ���Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
�����л��������������ǣ�������
| A���Ҵ� | B������ | C���ױ� | D����ȩ |
����˵����ȷ���ǣ�������
| A��c��H+��=0.1mol/L�ļ�����Һ�У�HCOO-��H+ ��Ŀ֮��Ϊ0.1 NA |
| B��ͨ������£�����Һ̬�����ǹ�̬������I-I����Br-Br�� |
| C����ϡ��ˮ��μ���ϡ�����У�����ҺpH=7ʱ��2C��NH4+��=C��SO42-�� |
D���ϳ�˳����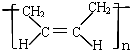 ���ĵ�����CH2=CH-CH=CH2 ���ĵ�����CH2=CH-CH=CH2 |
��ѧ����������������أ������뻯ѧ�йص�˵����ȷ���ǣ�������
| A����ѧҩƷ�Ż𣬶�Ҫ������ˮ����ĭ�������� |
| B���������ƹ�����Դ��ʵ�ֵ�̼�����;��֮һ |
| C��ʳƷ��װ���г�����С������ʯ�ң�Ŀ���Ƿ�ֹʳƷ�������� |
| D����ά���������ڿ�ˮ��Ϊ�����ǣ��ʿ��������Ӫ������ |


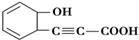 ������HBr����1��1�ӳɵIJ��������
������HBr����1��1�ӳɵIJ��������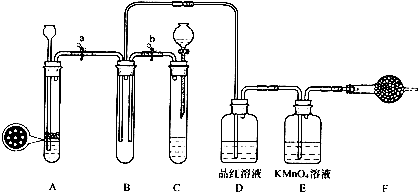
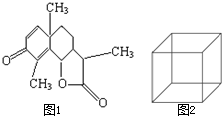 ��1��ͼ1��һ�����׳�ҩ--ɽ����Ľṹ��ʽ����ȷ�������ʽΪ��
��1��ͼ1��һ�����׳�ҩ--ɽ����Ľṹ��ʽ����ȷ�������ʽΪ��