��Ŀ����
������һ����Ҫ�IJ��ϣ������������������벻����������ͼ��ʾ�˽������˳�����ͭ������������Ԫ�ر�������ģ���������õĴ������ޣ�
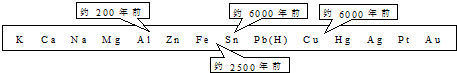
��1������ͼ�����ݺ��йصĻ�ѧ֪ʶ������Ϊ�������ģ���������õ��Ⱥ�˳���������Щ�����й� ��
�ٽ���ұ�������׳̶� �ڽ����ĵ����� �۽����Ļ�� �ܵؿ��н���Ԫ�صĺ���
��2��������������ʹ����������������������һ����Ҫ��־������д��һ����̼�ڸ��������°����ӳ������ﻹԭ�����Ļ�ѧ����ʽ
��3��ÿ�������ϸ����IJ����ܸߣ�����������ʴҲ����������˾����ʧ�����ڿ�������ʴ��ʵ���������� �� ����������õĽ����
��4�����Ļ�Ա���ǿ�������������ȴ���ֳ����õĿ�ʴ�ԣ���ԭ�� ��
��5������������֯�ѽ�������ΪʳƷ��ȾԴ֮һ��ָ��ÿ��ÿ�����İ�ȫ������Ӧ������4�������£�ij��ʳ����1000����ۡ�4������[��ѧʽΪKAl��SO4��2?12H2O]��С�մ�ʳ�εȸ��ϼ�����ˮ����ը������24����ƽ��ÿ��50�ˣ��ʣ�
����������Է�������Ϊ ��
�������м�Ԫ�ء���Ԫ�ء���Ԫ�ء���Ԫ�ء���Ԫ�ص�������Ϊ ��
����֪��������Ԫ�ص���������Ϊ5.7%����4����������Ԫ�ص�����Ϊ ��

��1������ͼ�����ݺ��йصĻ�ѧ֪ʶ������Ϊ�������ģ���������õ��Ⱥ�˳���������Щ�����й�
�ٽ���ұ�������׳̶� �ڽ����ĵ����� �۽����Ļ�� �ܵؿ��н���Ԫ�صĺ���
��2��������������ʹ����������������������һ����Ҫ��־������д��һ����̼�ڸ��������°����ӳ������ﻹԭ�����Ļ�ѧ����ʽ
��3��ÿ�������ϸ����IJ����ܸߣ�����������ʴҲ����������˾����ʧ�����ڿ�������ʴ��ʵ����������
��4�����Ļ�Ա���ǿ�������������ȴ���ֳ����õĿ�ʴ�ԣ���ԭ��
��5������������֯�ѽ�������ΪʳƷ��ȾԴ֮һ��ָ��ÿ��ÿ�����İ�ȫ������Ӧ������4�������£�ij��ʳ����1000����ۡ�4������[��ѧʽΪKAl��SO4��2?12H2O]��С�մ�ʳ�εȸ��ϼ�����ˮ����ը������24����ƽ��ÿ��50�ˣ��ʣ�
����������Է�������Ϊ
�������м�Ԫ�ء���Ԫ�ء���Ԫ�ء���Ԫ�ء���Ԫ�ص�������Ϊ
����֪��������Ԫ�ص���������Ϊ5.7%����4����������Ԫ�ص�����Ϊ
���㣺���������Ļ��˳����Ӧ��,�����г����Ͻ�����
ר�⣺�����������Ԫ��
��������1�������Ļ�����Խ����������ұ��Խ���ף�����ʹ��Խ�磻
��2�������£�CO������������������ԭ��Ӧ����Fe�Ͷ�����̼��
��3�����ڳ�ʪ�Ļ����з���������ʴ��
��4�����ı����������ܵ���������Ĥ��������Ĥ������ֹ����һ����������
��5����Է����������ڸ�Ԫ�ص����ԭ��������ԭ�Ӹ���֮���ĺͣ��������и�Ԫ�ص�����֮�ȵ��ڸ�Ԫ�ص����ԭ��������ԭ�Ӹ���֮���ıȣ���Ԫ������=������������������������
��2�������£�CO������������������ԭ��Ӧ����Fe�Ͷ�����̼��
��3�����ڳ�ʪ�Ļ����з���������ʴ��
��4�����ı����������ܵ���������Ĥ��������Ĥ������ֹ����һ����������
��5����Է����������ڸ�Ԫ�ص����ԭ��������ԭ�Ӹ���֮���ĺͣ��������и�Ԫ�ص�����֮�ȵ��ڸ�Ԫ�ص����ԭ��������ԭ�Ӹ���֮���ıȣ���Ԫ������=������������������������
���
�⣺��1�������Ļ�����Խ����������ұ��Խ���ף�����ʹ��Խ�磬������ĵ�����ǿ���������ĺ����أ���ѡ�٢ۣ�
��2�������£�CO������������������ԭ��Ӧ����Fe�Ͷ�����̼����Ӧ����ʽΪ3CO+Fe2O3
2Fe+3CO2���ʴ�Ϊ��3CO+Fe2O3
2Fe+3CO2��
��3�����ڳ�ʪ����������������ʴ��˵����������ʴˮ��Ҫˮ�Ϳ���/�������ʴ�Ϊ��ˮ������/��������4������������Ԫ�أ����ױ����������������������ﱡĤ����������ֹ��һ�����������ʴ�Ϊ�����ı����������ܵ���������Ĥ��������Ĥ������������������ĽӴ���
��5����KAl��SO4��2?12H2O��Է�������=39+27+32��2+16��20+1��24=474���ʴ�Ϊ��474��
�������м�Ԫ�ء���Ԫ�ء���Ԫ�ء���Ԫ�ء���Ԫ�ص�������Ϊ=��39��1������27��1������32��2������16��20������1��24��=39��27��64��320��24���ʴ�Ϊ��39��27��64��320��24��
����Ԫ������=����������������������=4g��4.7%=0.228g���ʴ�Ϊ��0.228g��
��2�������£�CO������������������ԭ��Ӧ����Fe�Ͷ�����̼����Ӧ����ʽΪ3CO+Fe2O3
| ||
| ||
��3�����ڳ�ʪ����������������ʴ��˵����������ʴˮ��Ҫˮ�Ϳ���/�������ʴ�Ϊ��ˮ������/��������4������������Ԫ�أ����ױ����������������������ﱡĤ����������ֹ��һ�����������ʴ�Ϊ�����ı����������ܵ���������Ĥ��������Ĥ������������������ĽӴ���
��5����KAl��SO4��2?12H2O��Է�������=39+27+32��2+16��20+1��24=474���ʴ�Ϊ��474��
�������м�Ԫ�ء���Ԫ�ء���Ԫ�ء���Ԫ�ء���Ԫ�ص�������Ϊ=��39��1������27��1������32��2������16��20������1��24��=39��27��64��320��24���ʴ�Ϊ��39��27��64��320��24��
����Ԫ������=����������������������=4g��4.7%=0.228g���ʴ�Ϊ��0.228g��
���������⿼���˽������й�֪ʶ����ȷ�����������ǽⱾ��ؼ���֪�����Ļ����Դ�����������Fe����ʴ��ǿ��ԭ��Ϊ�״��㣬

��ϰ��ϵ�д�
 �����Ծ���ĩ���100��ϵ�д�
�����Ծ���ĩ���100��ϵ�д� ˫��ͬ������ѵ��ϵ�д�
˫��ͬ������ѵ��ϵ�д�
�����Ŀ
����������ȷ���ǣ�������
| A�����ԣ�H2CO3��C6H5OH��H3COOH |
| B���е㣺PH3��NH3��H2O |
| C���۵㣺MgBr2��CCl4��BN |
| D�����ԣ�Ba��OH��2��Ca��OH��2��KOH |
1�����й�Ħ��������������Ӧ���У���ȷ���ǣ�������
| A��1 mol OH-������Ϊ17 g?mol-1 | ||
| B��������̼��Ħ������Ϊ44 g | ||
| C����ԭ�ӵ�Ħ�����������������ԭ������ | ||
D��һ����ԭ�ӵ���������
|

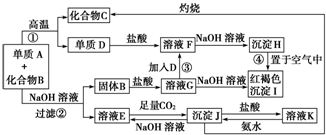
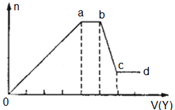 ij��ɫϡ��ҺX�У����ܺ����±����������е�ij���֣�
ij��ɫϡ��ҺX�У����ܺ����±����������е�ij���֣�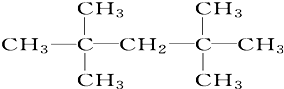 ����Ϊ
����Ϊ