��Ŀ����
2��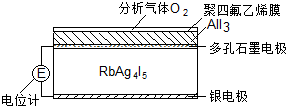 ���������Ǿ�����ǿ�����ˮ��Һ�ĵ������൱��һ�������壮�������ͨ�����е�����Ǩ�ƽ��е�ɴ��ݣ�����ֳ�Ϊ�������ӵ��壮Ŀǰ��������������ȫ��̬��ؼ�
���������Ǿ�����ǿ�����ˮ��Һ�ĵ������൱��һ�������壮�������ͨ�����е�����Ǩ�ƽ��е�ɴ��ݣ�����ֳ�Ϊ�������ӵ��壮Ŀǰ��������������ȫ��̬��ؼ�������������̽�����ȷ����Ӧ������㷺����RbAg4I5���壬����Ǩ�Ƶ�����ȫ��Ag+�����������Ǿ�����ǿ�����ˮ��Һ�ĵ������൱��һ�������壮�������ͨ�����е�����Ǩ�ƽ��е�ɴ��ݣ�����ֳ�Ϊ�������ӵ��壮Ŀǰ��������������ȫ��̬��ؼ�
������������̽�����ȷ����Ӧ������㷺����RbAg4I5���壬����Ǩ�Ƶ�����ȫ��Ag+��
���µ����ʴ�0.27��-1•cm-1������RbAg4I5���壬�����Ƴɵ绯ѧ��������������ͼ��һ�ֲⶨO2���������崫����ʾ��ͼ����������O2���������ķ���ϩ��Ĥ���ɵ�ص綯�Ʊ仯���Ե�֪O2�ĺ����������崫�������������У����б仯�϶�û�з������ǣ�������
| A�� | 4AlI3+3O2=2Al2O3+6I2 | B�� | I2+2Ag++2e-=2AgI | ||
| C�� | Ag-e-=Ag+ | D�� | I2+2Rb++2e-=2RbI |
���� ��ͼ��֪���������з���4AlI3+3O2�T2Al2O3+6I2��ԭ��صĸ�������Ag-e-=Ag+����������I2+2Ag++2e-=2AgI��������Ũ��Խ��EԽ���Դ������
��� �⣺��ͼ��֪���������з���4AlI3+3O2�T2Al2O3+6I2��ԭ��صĸ�������Ag-e-=Ag+����������I2+2Ag++2e-=2AgI��
��Ȼ������D�еķ�Ӧ��
��ѡD��
���� ���⿼��ԭ��ؼ�������ԭ��Ӧ����ȷ�����Ļ�ѧ��Ӧ�ǽ����Ĺؼ���Ҳ�ǽ����ѵ㣬ע������Ũ������Ũ�ȼ����ƵĹ�ϵ���ɽ����Ŀ�ѶȽϴ�

��ϰ��ϵ�д�
�����Ŀ
12��NA���������ӵ�����ֵ������˵����ȷ���ǣ�������
| A�� | 9 g H2O�����еĵ�����Ϊ5NA | |
| B�� | 1 mol MgCl2�к���������ΪNA | |
| C�� | 1 L 0.1 mol•L-1 Na2SO4��Һ����0.1NA ��Na+ | |
| D�� | 1 mol C10H20������ԭ������Ϊ13NA |
10������˵����ȷ���ǣ�������
| A�� | ��ϵͳ���������л��� 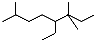 ������Ϊ3��3��7-����-4-�һ����� ������Ϊ3��3��7-����-4-�һ����� | |
| B�� | ʯ������ϩ��ú��Һ���������ʵ�����������ȥ���۾��漰��ѧ�仯 | |
| C�� | ���������Ҵ�����ϩ��ȫȼ�գ�ǰ�����ɸ����ˮ | |
| D�� | ȫ�������� 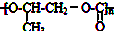 ���ɻ������飨 ���ɻ������飨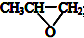 ����CO2��Ӧ�Ƶ� ����CO2��Ӧ�Ƶ� |
7�������£�pH=9��NaOH��Һ��Na2S��Һ����ˮ�������c��H+���ֱ�Ϊx mol/L��y mol/L�����߹�ϵ�ǣ�������
| A�� | x��y | B�� | x��y | C�� | x��y | D�� | ���ж� |
11��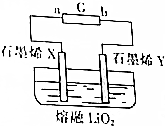 ��Ӣ��������ʱ������վ���������Ӣ����ѧ�����Ƴ�һ�ֳ�������﮵�أ������ع���ԭ����ͼ��ʾ��X��Y����ʯīϩ�Ƴɵ����Լ��õġ����ɡ��缫������ʯīϩX������ﮣ��õ�صĹ���ԭ����Li��s��+O2��g��$?_{���}^{�ŵ�}$LiO2��s��������˵������ȷ���ǣ�������
��Ӣ��������ʱ������վ���������Ӣ����ѧ�����Ƴ�һ�ֳ�������﮵�أ������ع���ԭ����ͼ��ʾ��X��Y����ʯīϩ�Ƴɵ����Լ��õġ����ɡ��缫������ʯīϩX������ﮣ��õ�صĹ���ԭ����Li��s��+O2��g��$?_{���}^{�ŵ�}$LiO2��s��������˵������ȷ���ǣ�������
 ��Ӣ��������ʱ������վ���������Ӣ����ѧ�����Ƴ�һ�ֳ�������﮵�أ������ع���ԭ����ͼ��ʾ��X��Y����ʯīϩ�Ƴɵ����Լ��õġ����ɡ��缫������ʯīϩX������ﮣ��õ�صĹ���ԭ����Li��s��+O2��g��$?_{���}^{�ŵ�}$LiO2��s��������˵������ȷ���ǣ�������
��Ӣ��������ʱ������վ���������Ӣ����ѧ�����Ƴ�һ�ֳ�������﮵�أ������ع���ԭ����ͼ��ʾ��X��Y����ʯīϩ�Ƴɵ����Լ��õġ����ɡ��缫������ʯīϩX������ﮣ��õ�صĹ���ԭ����Li��s��+O2��g��$?_{���}^{�ŵ�}$LiO2��s��������˵������ȷ���ǣ�������| A�� | �����̣���ԴG��b��Ϊ���� | |
| B�� | �ŵ���̣�Y��������ԭ��Ӧ | |
| C�� | �ŵ���̣�X���ĵ缫��ӦʽΪLi-e-+O2-�TLiO2 | |
| D�� | �����̣�ÿת��2 mol���ӣ�Y���ų�44.8L����״����O2 |
5�����100mL��c��H+��=0.30mol•L-1��������Һ������·��ͨ��0.04mol����ʱ�������������������������ǣ�������
| A�� | 0.10 mol•L-1AgNO3 | B�� | 0.20 mol•L-1Zn��NO3��2 | ||
| C�� | 0.20 mol•L-1Cu��NO3��2 | D�� | 0.20 mol•L-1Pb��NO3��2 |

 ��
�� +3NaOH $��_{��ѹ}^{����}$
+3NaOH $��_{��ѹ}^{����}$ +NaCl+2H2O��
+NaCl+2H2O�� ��
�� ����дһ�ֽṹ��ʽ����
����дһ�ֽṹ��ʽ����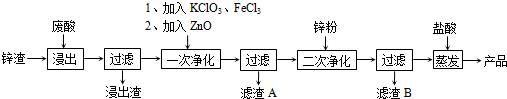
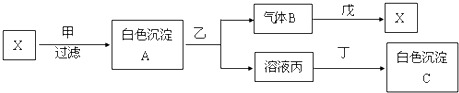
 ��
��