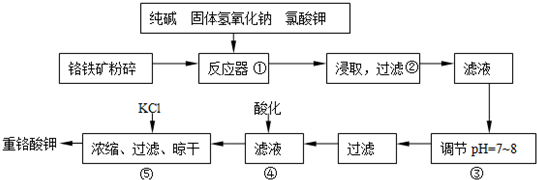
��Ŀ����
 ��֪��X��Y��Z��W�����ֶ�����Ԫ�أ����ǵ�ԭ������������������X��Y��Zͬ���ڣ�X��Wͬ���壻X��Z�γɵij���������֮һ�������ڸ�¯��������X��Zԭ�ӵ�L�㶼��2��δ�ɶԵ��ӣ�Q+����M��3d�������Ϊȫ����״̬����ش��������⣺
��֪��X��Y��Z��W�����ֶ�����Ԫ�أ����ǵ�ԭ������������������X��Y��Zͬ���ڣ�X��Wͬ���壻X��Z�γɵij���������֮һ�������ڸ�¯��������X��Zԭ�ӵ�L�㶼��2��δ�ɶԵ��ӣ�Q+����M��3d�������Ϊȫ����״̬����ش��������⣺��1��X��Y��Z�ĵ�һ��������С�����˳��Ϊ
��2��������X2H4Z2�������г��õ�ζƷ֮һ����ÿ�������к���
��3��X��W�ֱ���Z�γɵĻ�ѧ���ļ��Թ�ϵΪX-Z
��4��������XZ2���۵�Զ����WZ2����������
��5��д����̬Qԭ�ӵĵ����Ų�ʽ��
���㣺λ�ýṹ���ʵ����ϵӦ��
ר�⣺
������X��Z�γɵij���������֮һ�������ڸ�¯��������X��̼��Z������Q+����X��Y��Z�����ֶ�����Ԫ�أ����ǵ�ԭ������������������Y�ǵ�Ԫ�أ�X��Wͬ���壬��W�ǹ裬M��3d�������ȫ������E��ԭ��������29��ΪCuԪ�أ����Ԫ�������ɼ�ԭ�ӽṹ�������
���
�⣺X��Z�γɵij���������֮һ�������ڸ�¯��������X��̼��Z������Q+����X��Y��Z�����ֶ�����Ԫ�أ����ǵ�ԭ������������������Y�ǵ�Ԫ�أ�X��Wͬ���壬��W�ǹ裬M��3d�������ȫ������E��ԭ��������29��ΪCuԪ�أ�
��1����һ������ͬ���ڴ��������ӣ����еڢ�A����A�쳣������X��Y��Z�ĵ�һ��������С�����˳��ΪC��O��N��ͬ����ԭ�Ӱ뾶�����Ҽ�С�����뾶��С�����˳��ΪO��N��C���ʴ�Ϊ��C��O��N��O��N��C��
��2��������C2H4O2�Ľṹ��ʽΪCH3COOH������һ��̼��˫���⣬�������ǵ���������һ����7���Ҽ���1���м����ʴ�Ϊ��7��1��
��3��̼�ķǽ�����ǿ�ڹ裬��̼�����ļ���ǿ�ڹ��������ʴ�Ϊ������
��4��������̼�Ƿ��Ӿ��������������ԭ�Ӿ��壬���Ӽ��������ȹ��ۼ����ö࣬������̼��ֱ���ͷ��ӣ������е�̼��sp�ӻ����ʴ�Ϊ��������̼�Ƿ��Ӿ��������������ԭ�Ӿ��壬���Ӽ��������ȹ��ۼ����öࣻsp�ӻ���
��5��Q��29��Ԫ��ͭ����ԭ�Ӻ�����29�����ӣ����ݹ���ԭ��֪�����̬ԭ�Ӻ�������Ų�ʽΪ��ls22s22p63s23p63d104s1��HԪ����CuԪ�ؿ��γ�һ�ֺ�ɫ������ɾ���ṹ��Ԫ��֪��4��Hԭ��λ���ڲ���6��Hԭ��λ�����ϣ�������Hԭ����Ŀ=4+6��
=6��3��Cuԭ��Ϊ�ڲ���2��λ�����ġ�12��λ�ڶ��㣬�ʾ�����Cuԭ����Ŀ=3+2��
+12��
=6���ʸû�����Ļ�ѧʽΪCuH���û��������������ȼ�գ�����һ���ػ�ɫ�����һ�����壬Ӧ����CuCl2��HCl����Ӧ����ʽΪ��2CuH+3Cl2
2CuCl2+2HCl��
�ʴ�Ϊ��ls22s22p63s23p63d104s1��CuH��2CuH+3Cl2
2CuCl2+2HCl��
��1����һ������ͬ���ڴ��������ӣ����еڢ�A����A�쳣������X��Y��Z�ĵ�һ��������С�����˳��ΪC��O��N��ͬ����ԭ�Ӱ뾶�����Ҽ�С�����뾶��С�����˳��ΪO��N��C���ʴ�Ϊ��C��O��N��O��N��C��
��2��������C2H4O2�Ľṹ��ʽΪCH3COOH������һ��̼��˫���⣬�������ǵ���������һ����7���Ҽ���1���м����ʴ�Ϊ��7��1��
��3��̼�ķǽ�����ǿ�ڹ裬��̼�����ļ���ǿ�ڹ��������ʴ�Ϊ������
��4��������̼�Ƿ��Ӿ��������������ԭ�Ӿ��壬���Ӽ��������ȹ��ۼ����ö࣬������̼��ֱ���ͷ��ӣ������е�̼��sp�ӻ����ʴ�Ϊ��������̼�Ƿ��Ӿ��������������ԭ�Ӿ��壬���Ӽ��������ȹ��ۼ����öࣻsp�ӻ���
��5��Q��29��Ԫ��ͭ����ԭ�Ӻ�����29�����ӣ����ݹ���ԭ��֪�����̬ԭ�Ӻ�������Ų�ʽΪ��ls22s22p63s23p63d104s1��HԪ����CuԪ�ؿ��γ�һ�ֺ�ɫ������ɾ���ṹ��Ԫ��֪��4��Hԭ��λ���ڲ���6��Hԭ��λ�����ϣ�������Hԭ����Ŀ=4+6��
| 1 |
| 3 |
| 1 |
| 2 |
| 1 |
| 6 |
| ||
�ʴ�Ϊ��ls22s22p63s23p63d104s1��CuH��2CuH+3Cl2
| ||
������������Ҫ������̼��Ԫ�ص��й�֪ʶ���ѶȲ�����ʱע��Ҫ�������ó��������ʽṹ��֪ʶ��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
�����йػ�ѧ�����ʾ��ȷ���ǣ�������
| A�������ǵĽṹ��ʽ��C6H12O6 | ||
| B��NaHCO3�ĵ��룺NaHCO3=Na++H++CO32- | ||
C��������Ϊ53��������Ϊ78�ĵ�ԭ�ӣ�
| ||
D��CO2�ĵ���ʽ��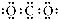 |
��ѧ�����������а�������Ҫ��ɫ������˵����Ӧ����ȷ���ǣ�������
| A�������������һ�κϳɵĵ����� |
| B�����ά�ͺϳ���ά�������л��߷��Ӳ��� |
| C���������͡���ˮ�Ҵ����Ǵ����� |
| D��ú��������Һ��������ѧ�仯���̣��ɱ�Ϊ�����Դ |
����ʵ��ɴﵽʵ��Ŀ���ǣ���������
�ٽ�CH3CH��OH��CH3��NaOH�Ĵ���Һ�����Ʊ�CH3CH=CH2
�� ������NaHCO3��Һ��Ӧ�Ʊ�
������NaHCO3��Һ��Ӧ�Ʊ�
����CH3CH2Br�е���AgNO3��Һ�Լ�����Ԫ��
������ˮ���ɼ���CH2=CH-CHO�е�̼̼˫����
�ٽ�CH3CH��OH��CH3��NaOH�Ĵ���Һ�����Ʊ�CH3CH=CH2
��
 ������NaHCO3��Һ��Ӧ�Ʊ�
������NaHCO3��Һ��Ӧ�Ʊ�
����CH3CH2Br�е���AgNO3��Һ�Լ�����Ԫ��
������ˮ���ɼ���CH2=CH-CHO�е�̼̼˫����
| A��ֻ�Т� | B��ֻ�Т٢� |
| C��ֻ�Т٢ۢ� | D�������� |
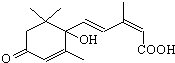 �����ڼ���찲�Ź㳡���������ʻ�ʩ����S-�տ����Ƽ����Ա�֤�ʻ�ʢ����S-�տ��صķ��ӽṹ��ͼ�����й��ڸ����ʵ�˵����ȷ���ǣ�������
�����ڼ���찲�Ź㳡���������ʻ�ʩ����S-�տ����Ƽ����Ա�֤�ʻ�ʢ����S-�տ��صķ��ӽṹ��ͼ�����й��ڸ����ʵ�˵����ȷ���ǣ�������| A�������ʽΪC15H18O4 |
| B��1mol��������NaOH��Һ��Ӧ����������2mol NaOH |
| C�����ܷ����ӳɷ�Ӧ�����ܷ���ȡ����Ӧ����ȥ��Ӧ |
| D��������FeCl3��Һ������ɫ��Ӧ������ʹ����KMnO4��Һ��ɫ |