��Ŀ����
��8�֣����ڿ���������������ҪӦ�á�ij��ȤС����0��50mol��L-1KI��0��2��������Һ��0��20mol��L-1K2S2O8��0��10mol��L-1Na2S2O3���Լ���̽����Ӧ�����Ի�ѧ��Ӧ���ʵ�Ӱ�졣
��֪��S2O82-+2I-�T2SO42-+I2��������I2+2S2O32-�TS4O62-+2I- ���죩��
��1����KI��Na2S2O3����۵Ļ����Һ�м���һ������K2S2O8��Һ������Һ�е�Na2S2O3�ľ�����Һ�� ɫ����α仯 ��Ϊȷ���ܹ۲쵽������S2O32����S2O82����ʼ�����ʵ���������ķ�ΧΪ��n��S2O32������n��S2O82���� ��
��2��Ϊ̽�ַ�Ӧ��Ũ�ȶԻ�ѧ��Ӧ���ʵ�Ӱ�죬��Ƶ�ʵ�鷽�����±���
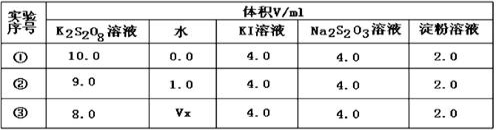
����Vx= mL����Ӧ���������� ������ţ���
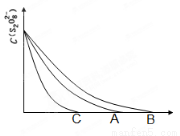
��3����֪ͼ��A����Ϊij�����£�Ũ��c(S2O82��)�� ��Ӧʱ��t�ı仯����ͼ�������������������䣬 ���B����C��������Ϊ���ͷ�Ӧ�¶ȣ� ���B����C��������Ϊ���������
��1������ɫ��Ϊ��ɫ����2�֣�<2 ��2�֣���2��2��0���� ��3��B��C��
��������
�����������1����KI��Na2S2O3����۵Ļ����Һ�м���һ������K2S2O8��Һ������Һ�е�Na2S2O3�ľ���I2�Ĵ����������Һ���ʹ��Һ��ɫ����ɫ��Ϊ��ɫ�����ڻᷢ����ӦS2O82-+2I-�T2SO42-+I2��I2+2S2O32-�TS4O62-+2I-��������Ҫ��֤�ܹ��۲쵽��Ӧ��������n(S2O32��)��n(S2O82-)<2:1����2������ʹ��2��3�Աȸı���й�ֻ��S2O82-��Ũ�ȣ�����Ķ���ͬ�����Կ�����Vx=2��0���ٵķ�Ӧ��Ũ�����������������������ʱ������Ӧ���Ũ�ȿ��Լӿ컯ѧ��Ӧ���ʣ����Է�Ӧ���������Ǣ٣���3����֪ͼ��A����Ϊij�����£�Ũ��c(S2O82��)�� ��Ӧʱ��t�ı仯����ͼ�������������������䣬���ڽ��ͷ�Ӧ���¶ȣ���ѧ��Ӧ���ʼ�С������B����Ϊ���ͷ�Ӧ�¶ȣ������������ѧ��Ӧ���ʼӿ죬��������C�Ǽ��������Ӱ��ġ�
���㣺���黯ѧ��Ӧ�������������ѧ��Ӧ���ʵıȽϡ�Ӱ�����ص�֪ʶ��

 ʱ�����������ҵԭ���ܳ�����ϵ�д�
ʱ�����������ҵԭ���ܳ�����ϵ�д� ����νӽ̲���ĩ���Ԥϰ�人������ϵ�д�
����νӽ̲���ĩ���Ԥϰ�人������ϵ�д� ������ҵ��ٳɳ����½������������ϵ�д�
������ҵ��ٳɳ����½������������ϵ�д�
 cC(g)+dD (g)
cC(g)+dD (g)