��Ŀ����
4���ϳɰ���ӦΪ N2��g��+3H2��g��?2NH3��g��+92.4kJ������˵����ȷ���ǣ�������| A�� | һ�������£���N2������������N2�ķֽ��������ʱ���÷�Ӧ����ƽ��״̬ | |
| B�� | ȡ1mol N2��3mol H2�����ܱ������ڷ�Ӧ��ƽ��ʱ������2 mol NH3 | |
| C�� | һ�������·�Ӧ�ﵽƽ�⣬�ٳ���N2��������Ӧ���������淴Ӧ���ʼ�С | |
| D�� | һ�������·�Ӧ�ﵽƽ�⣬�������¶ȣ���Ӧ��������ƽ��������Ӧ�����ƶ� |
���� A����ѧ��Ӧ�ﵽƽ��״̬ʱ�����淴Ӧ������ȣ�
B���÷�ӦΪ���淴Ӧ��������ȫ��Ӧ��
C��Ũ���������淴Ӧ���ʶ�����
D�������¶ȣ���Ӧ��������ƽ�������ȷ�Ӧ�����ƶ���
��� �⣺A��һ�������£���N2������������N2�ķֽ�������ȣ�˵�����淴Ӧ������ȣ��ﵽ��ƽ�⣬��A��ȷ��
B���÷�ӦΪ���淴Ӧ��������ȫ��Ӧ�����ɵ� NH3С��2mol����B����
C������N2��Ũ���������淴Ӧ���ʶ�����������Ӧ���ʴ����淴Ӧ���ʣ���C����
D���÷�Ӧ���ȣ������¶ȣ���Ӧ��������ƽ�������ȷ�Ӧ�����ƶ������淴Ӧ����D����
��ѡA��
���� ���⿼�鷴Ӧ���ʵ�Ӱ�����ء���ѧƽ���Ӱ�����أ��Ƕ�ѧ������֪ʶ�Ŀ��飬�ϼ�

��ϰ��ϵ�д�
 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д�
�����Ŀ
13�����л�ѧ��Ӧ�У����ӷ���ʽ��ʾ��ȷ���ǣ�������
| A�� | ������̼���Ʒ�Ӧ CO32-+2H+=CO2��+H2O | |
| B�� | ��м��ϡ���ᷴӦ �������� 2Fe+6H+�T2Fe3++3H2�� | |
| C�� | Na2CO3��Һ�����ʯ��ˮ��Ӧ ���� CO32-+Ca2+�TCaCO3�� | |
| D�� | H2SO4��Һ��Ba��OH��2��Һ��ϲ������� SO42-+Ba2+�TBaSO4�� |
14����������������ǣ�������
| A�� | ��a L 0.1 mol•L-1��CH3COOH��Һ��b L 0.1 mol•L-1��KOH��Һ��ϣ�������Һ��һ�����ڣ�c��K+��+c��H+��=c��CH3COO-��+c��OH-�� | |
| B�� | ��0.1 mol•L-1��CH3COOH��Һ��0.3 mol•L-1��Ba��OH��2��Һ�������ϣ�������Һ��һ�����ڣ�c��OH-����c��Ba2+����c��CH3COOH����c��H+�� | |
| C�� | ��1 mol•L-1��CH3COOH��Һ�м�������CH3COONa���壬����CH3COONaˮ���Լ��ԣ�������Һ��pH���� | |
| D�� | �����£���pH=3��CH3COOH��Һ��pH=11��NaOH��Һ�У�ˮ�ĵ���̶���ͬ |
11��ij����Է�������Ϊ82�����ж��������ǣ�������
| A�� | ������ | B�� | ������ | ||
| C�� | ����5��̼ԭ�ӵ��� | D�� | ��ϩ�� |
18��Ҫ��֤Mg��Fe��Zn���ֽ����Ļ��˳��ѡ�������������ʽ���ʵ�飨������
| A�� | Zn��FeCl2��Һ��MgCl2��Һ | B�� | Fe��Zn��MgCl2��Һ | ||
| C�� | Mg��ZnCl2��FeSO4��Һ | D�� | Fe��ZnSO4��Һ��MgCl2��Һ |
9�������£���0.1000mol•L-1NaOH��Һ�ζ�20.00mL 0.1000mol•L-1CH3 COOH��Һ���õζ�������ͼ������˵����ȷ���ǣ�������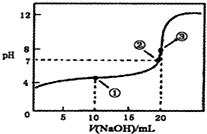

| A�� | �ٴ�ˮ�ĵ���̶ȴ��ڢڴ�ˮ�ĵ���̶� | |
| B�� | �ڴ������NaOH��Һǡ�ý�CH3COOH�к� | |
| C�� | �۴���ʾ��Һ��c��N+����c��CH3COO-�� | |
| D�� | �ζ������п��ܳ��֣�c��CH3COOH����c��CH3COO-����c��H+����c��Na+����c��OH-�� |
16����������Һ��һ���ܴ���������������ǣ�������
| A�� | pH=2����Һ��Fe2+��NH4+��Cl-��NO3- | |
| B�� | ��ʹpH��ֽ������ɫ����Һ��Na+��NO3-��S2-��CO32- | |
| C�� | ���д���Fe3+����Һ��SCN-��I-��K+��Br- | |
| D�� | ����������ɫ��Һ��ClO-��MnO4-��Al3+��S2- |
13����1�����ױ������ϵ�һ�����������ͬ���칹�壬��Щһ��������������Ķ�Ӧ���ױ����۵�ֱ�Ϊ��
�۵�Ϊ234����ӵĽṹ��ʽΪ
��2����ȫȼ���������ٱ��飨C3H8�����ڶ�ϩ��C4H8��������ϩ��C2H4�����ܼ��飨C6H14���������ʵ�������������������������С��Ϊ�ۣ���д��ţ���������ʱ�������������Ϊ�٣���д��ţ���
��3��0.2molij��A����������ȫȼ�պ�����CO2��H2O��1.2mol���Իش�
������A����ʹ��ˮ��ɫ������һ��������������������ȡ����Ӧ����һ��ȡ����ֻ��һ�֣�����A�Ľṹ��ʽΪ ��
��
������A��ʹ��ˮ��ɫ���ڴ��������£���H2�ӳɷ�Ӧ������2��2-�������飬����A��������3��3-����-1-��ϩ��
��4����ɷ���CnH2n-2��ij���������ӽṹ��û��֧�������������ȫȼ��ʱ������O2�������ͬ״���¸������������8.5�����ɴ˷����ش�
��������Ϊ��״��ϩ��������������ʵ�����Br2�ӳɺ�ֻ�ܵõ���һ���������Ľṹ��ʽΪCH2�TCH-CH2-CH2-CH�TCH2
��������ֻ��������ʵ�����Br2�����ӳɷ�Ӧ������ṹ��ʽΪ ������һ�֣���
������һ�֣���
| һ������ױ� | 234�� | 206�� | 213.8�� | 204�� | 214.5�� | 205 |
| ��Ӧ�Ķ��ױ� | -13�� | -54�� | -27�� | -54�� | -27�� | -54�� |

��2����ȫȼ���������ٱ��飨C3H8�����ڶ�ϩ��C4H8��������ϩ��C2H4�����ܼ��飨C6H14���������ʵ�������������������������С��Ϊ�ۣ���д��ţ���������ʱ�������������Ϊ�٣���д��ţ���
��3��0.2molij��A����������ȫȼ�պ�����CO2��H2O��1.2mol���Իش�
������A����ʹ��ˮ��ɫ������һ��������������������ȡ����Ӧ����һ��ȡ����ֻ��һ�֣�����A�Ľṹ��ʽΪ
 ��
��������A��ʹ��ˮ��ɫ���ڴ��������£���H2�ӳɷ�Ӧ������2��2-�������飬����A��������3��3-����-1-��ϩ��
��4����ɷ���CnH2n-2��ij���������ӽṹ��û��֧�������������ȫȼ��ʱ������O2�������ͬ״���¸������������8.5�����ɴ˷����ش�
��������Ϊ��״��ϩ��������������ʵ�����Br2�ӳɺ�ֻ�ܵõ���һ���������Ľṹ��ʽΪCH2�TCH-CH2-CH2-CH�TCH2
��������ֻ��������ʵ�����Br2�����ӳɷ�Ӧ������ṹ��ʽΪ
 ������һ�֣���
������һ�֣���
14��NAΪ�����ӵ�����������˵����ȷ���ǣ�������
| A�� | ��״���£�11.2 L�����������ķ�����Ϊ0.5NA | |
| B�� | 30 g�����������õ��Ӷ���ĿΪ6NA | |
| C�� | ��״���£�11.2 L���ȼ�������������Ϊ0.5NA | |
| D�� | ����C2H4��C3H6��C4H8�Ļ�����干14 g����ԭ����Ϊ3NA |