��Ŀ����
�±������ڱ���һ����,�Իش���������:
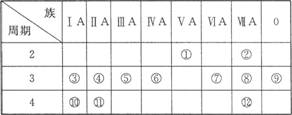
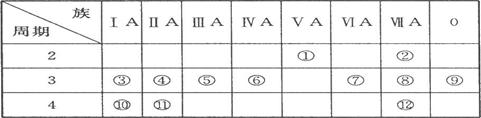
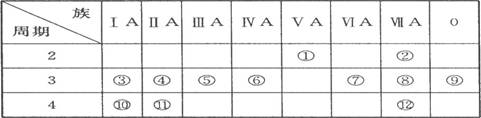
(1)д������Ԫ�صķ��ţ��� ���� ���� ����
(2)��ЩԪ���У���һ��������С��Ԫ���� (����ţ���ͬ)���縺������Ԫ���� ��
(3)����ЩԪ�ص�����������Ӧˮ�����У�������ǿ���� ��������ǿ���� �������Ե����������� ��д������֮�����Ӧ�����ӷ���ʽ�� ;
.
(4)����ЩԪ���У�ԭ�Ӱ뾶��С���� (��Ԫ�ط��ţ���ͬ)��ԭ�Ӱ뾶������ ��
(5)�ڢ�����У���ѧ���ʽϻ��õ��� �������û�ѧʵ��֤��?
(1)N Si S K
(2)�� ��
(3)HClO4 KOH
Al(OH)3 3H++Al(OH)3=A13++3H2O��Al(OH)3+0Hһ=A102-+2H20��H++OHһ=H2O
(4)F K
(5)Na ��ˮ��Ӧ��Na��ˮ��Ӧ�����ҡ�

��ϰ��ϵ�д�
 ���ɿ��õ�Ԫ����AB��ϵ�д�
���ɿ��õ�Ԫ����AB��ϵ�д� С�����ϵ�д�
С�����ϵ�д�
�����Ŀ
�±������ڱ���һ���֣��������е���ĸ�ֱ����ijһ��ѧԪ�ء�
| b | | | |||||||||||||||
| | | | | h | | j | | | |||||||||
| a | c | f | | i | | l | m | ||||||||||
| | | | e | | | | | | | | | | g | | k | | |
| | d | | | | | | | | | | | | | | | | |
(2) ��Ԫ��a��c��d��f��l��b��ԭ�Ӱ뾶���ɴ�С��˳������________________________��
(3) iԪ������_______________���������ǽ�����Ԫ�أ���������ϼ���____________��
��ͻ��ϼ���___________��������������Ӧ������һ����ˮ�Ժ�ǿ�����ʣ�����������ʵ�����ﳣ�����������д��i�ĵ�����������Ӧ�Ļ�ѧ����ʽ��_________________
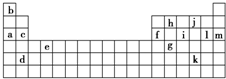 �±������ڱ���һ���֣��������е���ĸ�ֱ����ijһ��ѧԪ�أ�
�±������ڱ���һ���֣��������е���ĸ�ֱ����ijһ��ѧԪ�أ�