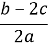��Ŀ����
ij��ɫ��Һ��ֻ��������8�������е�ij���֣�Mg2+��H+��Ag+��Na+��Cl-��HC ��OH-��N
��OH-��N ����֪����Һ��������Ӧ�ų��������Իش��������⣺
����֪����Һ��������Ӧ�ų��������Իش��������⣺
��1������Ӧ������Al3+������Һ�п��ܴ��ڵ������С�������������������һ�����ڵ������С�����������������������һ�������ڵ������С���������������Ӧ�����ӷ���ʽΪ����������������������������
��2������Ӧ������Al ������Һ�п��ܴ��ڵ������С���������������һ�����ڵ������С� ��һ�������ڵ������С�����������������������Ӧ�����ӷ���ʽΪ�� ��
������Һ�п��ܴ��ڵ������С���������������һ�����ڵ������С� ��һ�������ڵ������С�����������������������Ӧ�����ӷ���ʽΪ�� ��
��1��Mg2+��Na+��H+��Cl-��Ag+��HC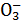 ��N
��N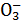 ��OH-��2Al+6H+====2Al3++3H2��
��OH-��2Al+6H+====2Al3++3H2��
��2��N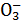 ��Cl-��OH-��Na+��H+��Mg2+��Ag+��HC
��Cl-��OH-��Na+��H+��Mg2+��Ag+��HC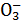
2Al+2OH-+2H2O====2Al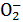 +3H2��
+3H2��
��������
�����������1������Ӧ������Al3+����˵������Һ�����ԣ���HC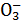 ��OH-�����ڣ�����HNO3��������H2������Ҳ������N
��OH-�����ڣ�����HNO3��������H2������Ҳ������N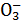 ������ֻ��ΪHCl����Ҳ������Ag+��һ������H+��Cl-��Mg2+��Na+���ܴ��ڡ���2����Ӧ������Al
������ֻ��ΪHCl����Ҳ������Ag+��һ������H+��Cl-��Mg2+��Na+���ܴ��ڡ���2����Ӧ������Al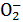 ��˵����Һ�ʼ��ԣ���H+��Mg2+��Ag+��HC
��˵����Һ�ʼ��ԣ���H+��Mg2+��Ag+��HC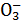 һ�������ڣ�һ������OH-��Na+��N
һ�������ڣ�һ������OH-��Na+��N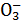 ��Cl-���ܴ��ڡ�
��Cl-���ܴ��ڡ�
���㣺�������Ͻ�

 ����ͬ�����Ծ�ϵ�д�
����ͬ�����Ծ�ϵ�д������й�ʵ�����������ͽ��ͻ���۶���ȷ����
ѡ�� | ���� | ���� | ���ͻ���� |
A | ������Fe���м���ϡHNO3����ַ�Ӧ�μ�KSCN��Һ | ��Һ�ʺ�ɫ | ϡHNO3��Fe����ΪFe3�� |
B | Al������ϡHNO3�� | ������ | Al�����汻ϡHNO3������ �γ����ܵ�����Ĥ |
C | ��ij��Һ�еμ�ϡNaOH��Һ����ʪ�� ��ɫʯ����ֽ�����Թܿ� | ��ֽδ���� | ����ȷ������Һ���Ƿ� ��NH4+ |
D | ����ɫʯ����Һ��ͨ��SO2 | ��Һ��ɫ | SO2��Ư���� |
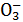 ��Cl-B��K+��Na+��Br-��Cl-
��Cl-B��K+��Na+��Br-��Cl- ��HC
��HC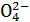
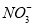 ��Cl-��
��Cl-��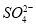 B��Na+��K+��Ba2+��
B��Na+��K+��Ba2+��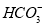
 �����ʵ���Ũ��(��λmol��L-1)Ϊ��(����)
�����ʵ���Ũ��(��λmol��L-1)Ϊ��(����) B��
B��
 D��
D��