��Ŀ����
13��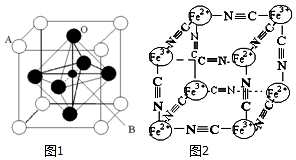 Ti��Fe��Cr��Mn�Ⱦ�Ϊ����Ԫ�أ����������������Ų����������Ҫ���ã����䵥�ʺͻ������Ӧ���о���Ŀǰ��ѧ�о���ǰ��֮һ����ش��������⣺
Ti��Fe��Cr��Mn�Ⱦ�Ϊ����Ԫ�أ����������������Ų����������Ҫ���ã����䵥�ʺͻ������Ӧ���о���Ŀǰ��ѧ�о���ǰ��֮һ����ش��������⣺��1��CrԪ�صĻ�̬ԭ�ӵ����Ų�ʽΪ1s22s22p63s23p63d54s1���Ƚ�Fe�� Mn�ĸ��������ܺ��֣���̬Mn2+��ʧȥһ�����ӱ���̬Fe2+��ʧȥһ�������ѣ���ѡ����ס�����
��2��CuԪ�ش������ڱ�ds������ʢ������ͭ���Թ�����백ˮ�������γ���ɫ�������������백ˮ�������ܽ⣬��ʱ�����ӷ���ʽΪCu��OH��2+4NH3•H2O=[Cu��NH3��4]2++2OH-+4H2O���������Ҵ�����������ɫ�ľ��壬�������ӵ����ӹ���Ϊƽ���ı���
��3��ij���������������ͼ1������Aԭ��Ϊ�����Ķ��㣬Aλ������Ca��Sr��Ba��Pb����Bλ��V��Cr��Mn��Feʱ�����ֻ��������CMRЧӦ���ŵ���ЧӦ������A��B��O��ʾ�������⾧��Ļ�ѧʽ��ABO3��
��4����һ����ɫ����ɱ�ʾΪ��[KxFey��CN��z]���о��������Ľṹ������Fe2+��Fe3+�ֱ�ռ��������Ķ��㣬�����������ڣ���CN-λ������������ϣ�K+λ�������������ģ���K+��ȱ��Ϊ50%��������û��K+��ռ�����ĵİٷֱȣ����侧���е������Ӿ����ṹ��ͼ2��ʾ���þ���Ļ�ѧʽ�ɱ�ʾΪKFe2��CN��6��
���� ��1�������������ԭ�������ع���������д������ϵĵ��Ӵ���ȫ����������ȫ��ʱ���ȶ������Ӳ�����ʧȥ��
��2��Cuλ�����ڱ��������ڢ�B�壬Ϊds��Ԫ�أ���ʢ������ͭˮ��Һ���Թ�����백ˮ�������γ�������ͭ�����������Ӱ�ˮ��������ͭ�ܽ�õ��İ���ͭ�����ӣ���Һ������ɫ����Һ�������뼫�Խ�С���ܼ��Ҵ������ɵ�[Cu��NH3��4]SO4�����õ�����ɫ�ľ��壻
��3�����ݾ�̯�����㾧����A��B��Oԭ����Ŀ������ȷ����ѧʽ��
��4������ͼ2�����ṹ��֪�������к���Fe2+�ĸ���Ϊ4��$\frac{1}{8}$=$\frac{1}{2}$��Fe3+�ĸ���Ϊ4��$\frac{1}{8}$=$\frac{1}{2}$��CN-�ĸ���Ϊ12��$\frac{1}{4}$=3������Fe2+��Fe3+��CN-�ĸ�����Ϊ1��1��6�����ݻ��ϼ۴�����Ϊ���֪��x=1������Ļ�ѧʽ�ɱ�ʾΪKFe2��CN��6��
��� �⣺��1�������������ԭ�������ع�����������֪CrԪ�ػ�̬ԭ�ӵ����Ų�ʽΪ1s22s22p63s23p63d54s1����Ԫ�ص�3d�ܼ���4s�ܼ��ϵĵ��Ӷ��Ǽ۵��ӣ�MnԪ�ػ�̬ԭ�ӵļ۵����Ų�ʽΪ3d54s2����Mn2+ת��ΪMn3+ʱ��3d�ܼ��ɽ��ȶ���3d5�����״̬ת��Ϊ���ȶ���3d4״̬��Ҫ�������϶ࣻ��Fe2+ת��ΪFe3+ʱ��3d�ܼ��ɲ��ȶ���3d6ת��Ϊ�ȶ���3d5��������ȶ�״̬����Ҫ���������Ҫ�٣�
�ʴ�Ϊ��1s22s22p63s23p63d54s1���ѣ�
��2��Cuλ�����ڱ��������ڢ�B�壬Ϊds��Ԫ�أ�������ͭ�백ˮ������Ӧ��Cu��OH��2+4NH3•H2O=[Cu��NH3��4]2++2OH-+4H2O�������ܽ⣬�����Һ�м��Ҵ������ɵ�[Cu��NH3��4]SO4�ڼ��Լ��Ե��Ҵ����ܽ�ȼ�С�������õ�����ɫ�ľ��壬������[Cu��NH3��4]2+�У�Cu�γ�4���Ҽ���d������Ӳ����ӻ�������sp3�ӻ���Ϊƽ���ı��νṹ��
�ʴ�Ϊ��ds��Cu��OH��2+4NH3•H2O=[Cu��NH3��4]2++2OH-+4H2O��������ƽ���ı��Σ�
��3��������Aԭ����Ŀ=8��$\frac{1}{8}$=1��Bԭ����Ŀ=1��Oԭ����Ŀ=6��$\frac{1}{2}$=3���������⾧��Ļ�ѧʽΪ��ABO3��
�ʴ�Ϊ��ABO3��
��4������ͼ2�����ṹ��֪�������к���Fe2+�ĸ���Ϊ4��$\frac{1}{8}$=$\frac{1}{2}$��Fe3+�ĸ���Ϊ4��$\frac{1}{8}$=$\frac{1}{2}$��CN-�ĸ���Ϊ12��$\frac{1}{4}$=3������Fe2+��Fe3+��CN-�ĸ�����Ϊ1��1��6�����ݻ��ϼ۴�����Ϊ���֪��x=1������Ļ�ѧʽ�ɱ�ʾΪKFe2��CN��6��
�ʴ�Ϊ��KFe2��CN��6��
���� ���⿼���Ϊ�ۺϣ������ʽṹ�Ŀ��飬Ϊ�߿��������ͺ�Ƶ���㣬���ؿ���ѧ���ķ��������ͼ�����������Ŀ�漰��������Ų��������ܡ���λ������������ȣ�ּ�ڿ���ѧ���Ի���֪ʶ�����ռ��ۺ�Ӧ�ã��Ѷ��еȣ�

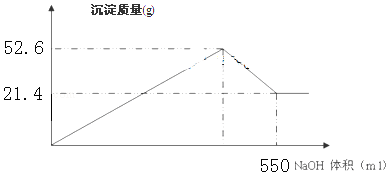
| A�� | ����̿��SO2��Na2O2����ʹƷ����Һ��ɫ����ԭ����ͬ | |
| B�� | �ǽ��������ﲻһ���������������������������Ǽ��������� | |
| C�� | ͬ��ͬѹ�£�������ͬ���������ۣ��ֱ���������ϡ�����ϡ���ᷴӦ����������������ͬ | |
| D�� | ��SO2����ͨ��BaCl2��Һ��������δ���������ɣ�����ͨ��NO2���г������� |
| A�� | ������ʹ���Ը��������Һ��ɫ | B�� | ������ʹ���Ը��������Һ��ɫ | ||
| C�� | ��ʹ�����Ļ�����ǿ | D�� | ����ʹ���Ļ�����ǿ |
| A�� | 4 �� | B�� | 6 �� | C�� | 8 �� | D�� | 10 �� |
| A�� | CH4��SiH4��GeH4��SnH4 | B�� | NH3��PH3��AsH3��SbH3 | ||
| C�� | H2O��H2S��H2Se��H2Te | D�� | HCl��HBr��HI��HF |
 �о�CO2�����öԴٽ���̼���Ĺ���������Ҫ�����壮
�о�CO2�����öԴٽ���̼���Ĺ���������Ҫ�����壮��1����CO2�뽹̿��������CO��CO�����������ȣ�
����֪��Fe2O3��s��+3C��ʯī��=2Fe��s��+3CO��g����H1=+489.0kJ•mol-1
C��ʯī��+CO2��g��=2CO��g����H2=+172.5kJ•mol-1
��CO��ԭFe2O3���Ȼ�ѧ����ʽΪ3CO��g��+Fe2O3��s��=2Fe��s��+3CO��g����H=-28.5kJ•mol-1��
����֪Fe��CO���γ����ʻ���[Fe��CO��5]���û������൱����������H2�����⻯�ʻ������⻯�ʻ���Ϊ��Ԫ���ᣬ����NaOH��Ӧ�������ʻ�������ƣ���д�����ʻ�������H2�ķ�Ӧ����ʽFe��CO��5+H2=H2Fe��CO��4+CO��
������ȼ�շ�Ӧ����Ƴ�CO/O2ȼ�ϵ�أ���KOH��ҺΪ���Һ����д���õ�صĸ�����ӦʽCO-2e-+4OH-=CO32-+2H2O��
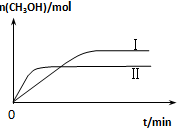
��2��ijʵ�齫CO2��H2����һ��������ܱ������У������ֲ�ͬ�����·�����Ӧ��
CO2��g��+3H2��g��?CH3OH��g��+H2O��g����H=-49.0kJ•mol-1
���CH3OH�����ʵ�����ʱ��仯����ͼ��ʾ���ش����⣺
������I�����Ӧ��ƽ�ⳣ����С��ϵΪK�����K��������ڡ��������ڡ���С�ڡ�����
��һ���¶��£����ݻ���ͬ�ҹ̶��������ܱ������У������·�ʽͶ�뷴Ӧ�һ��ʱ���ﵽƽ�⣮
| �� �� | �� | �� |
| ��Ӧ��Ͷ���� | 1molCO2��3molH2 | a molCO2��b molH2��c molCH3OH��g����c molH2O��g�� |