��Ŀ����
15��5�ֹ�������A��B��C��D��E���±��в�ͬ������������ɣ����Ǿ�������ˮ��| ������ | Na+��Al 3+��Fe3+��Cu2+��Ba2+ |
������ | OH-��Cl-��CO32-��NO3-��SO4- |
��C��E��Һ�Լ��ԣ�A��B��D��Һ�����ԣ�0.1mol/L��E��ҺPH��13��
��B��Һ��E��Һ��Ϻ�������ɫ������ͬʱ�����������壻
������C��Һ��D��Һ��Ϻ������ɫ����������C��Һ��D��Һ��Ϻ�������
�ܽ�38.4g CuƬͶ��װ������D��Һ���Թ��У�Cu���ܽ⣬�ٵμ�1.6mol•L-1ϡH2SO4��Cu���ܽ⣬�ܿڸ����к���ɫ������֣�
��1���ݴ��ƶ�C��D�Ļ�ѧʽΪ��CBa��OH��2��DAl��NO3��3��
��2��д��������з�����Ӧ�����ӷ�Ӧ����ʽ2Fe3++3CO32-+3H2O=2Fe��OH��3��+3CO2����
��3�����������Ҫ��CuƬ��ȫ�ܽ⣬���ټ���ϡH2SO4�������500mL��
��4������ȷ������ҺΪB��A������ĸ��ţ���
���� ��C��E��Һ�Լ��ԣ���Һ����Ϊ����Һ��ǿ�������Σ�A��B��D��Һ�����ԣ�0.1mol/L��E��ҺpH��13����1molE�к���С��1mol�����������ӣ�ֻ��Ϊ���������ˮ�⣬�������ӹ����֪��E����CO32-���ӣ�������ӹ��棬Eֻ��Ϊ̼���ƣ�������ӹ����֪��CΪ����������
��B��Һ��̼������Һ��Ϻ�������ɫ������ͬʱ�����������壬��B�к���Fe3+����̼�������˫ˮ�ⷴӦ���ɶ�����̼�������������ɫ������
����������������Һ��D��Һ��Ϻ������ɫ��������������������Һ��D��Һ��Ϻ�������˵��D�к���Al3+���Ҳ�������������
�ܽ�38.4 g CuƬͶ��װ������D��Һ���Թ��У�CuƬ���ܽ⣬�ٵμ�1.6 mol•L-1ϡH2SO4��Cu���ܽ⣬�ܿڸ����к���ɫ������֣�˵��D�к���NO3-������ôDΪ����������ôAΪ����ͭ�����Ȼ�ͭ����ôB��Ϊ�Ȼ����������������Դ˽����⣮
��� �⣺��C��E��Һ�Լ��ԣ���Һ����Ϊ����Һ��ǿ�������Σ�A��B��D��Һ�����ԣ�0.1mol/L��E��ҺpH��13����1molE�к���С��1mol�����������ӣ�ֻ��Ϊ���������ˮ�⣬�������ӹ����֪��E����CO32-���ӣ�������ӹ��棬Eֻ��Ϊ̼���ƣ�������ӹ����֪��CΪ����������
��B��Һ��̼������Һ��Ϻ�������ɫ������ͬʱ�����������壬��B�к���Fe3+����̼�������˫ˮ�ⷴӦ���ɶ�����̼�������������ɫ������
����������������Һ��D��Һ��Ϻ������ɫ��������������������Һ��D��Һ��Ϻ�������˵��D�к���Al3+���Ҳ�������������
�ܽ�38.4 g CuƬͶ��װ������D��Һ���Թ��У�CuƬ���ܽ⣬�ٵμ�1.6 mol•L-1ϡH2SO4��Cu���ܽ⣬�ܿڸ����к���ɫ������֣�˵��D�к���NO3-������ôDΪ����������ôAΪ����ͭ�����Ȼ�ͭ����ôB��Ϊ�Ȼ���������������
��1�������Ϸ�����֪��CΪBa��OH��2��DΪAl��NO3��3���ʴ�Ϊ��Ba��OH��2��Al��NO3��3��
��2�������Ϊ̼�����������ӵĻ���ˮ�ⷴӦ��������Ӧ�����ӷ�Ӧ����ʽΪ2Fe3++3CO32-+3H2O=2Fe��OH��3��+3CO2����
�ʴ�Ϊ��2Fe3++3CO32-+3H2O=2Fe��OH��3��+3CO2����
��3��38.4 g Cu�����ʵ���Ϊ��$\frac{38.4g}{64g/mol}$=0.6mol��������з��������ӷ�Ӧ����ʽΪ��3Cu+8H++3NO3-=3Cu2++2NO+4H2O����Ҫ��CuƬ��ȫ�ܽ⣬��Ҫ�����ӵ����ʵ���Ϊ1.6mol�������ټ���ϡH2SO4�������ΪV����1.6mol/L��V��2=1.6mol����V=500mL���ʴ�Ϊ��500��
��4���ɷ�����֪AΪ����ͭ�����Ȼ�ͭ����ôB��Ϊ�Ȼ���������������A��B������ȷ�����ʴ�Ϊ��A��
���� ���⿼��������ƶϣ���Ŀ�ۺ��Խ�ǿ���漰�����ƶϡ����Ӽ��顢���ӹ��桢�����ܽ�ȱȽϣ���Ҫѧ���������ջ���֪ʶ�������ڿ���ѧ���������������Ѷ��еȣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | ����Һ�У�2c ��H+��=c��A-��+c��OH-�� | |
| B�� | ��pH=3��HA��Һ��pH=11��NaOH��Һ�������ϣ�������Һ�У�c��Na+����c��A-����c��OH-����c��H+�� | |
| C�� | Ũ�Ⱦ�Ϊ0.1 mol•L-1��HA��NaA��Һ�������ϣ�������Һ�У�c��A-����c��HA����c��Na+����c��OH-����c��H+�� | |
| D�� | 0.1 mol•L-1 HA��Һ��0.05 mol•L-1 NaOH��Һ�������ϣ�������Һ�У�2c��H+��+c��HA���Tc��A-��+2c��OH-�� |
| A�� | Al��Fe2O3�ܷ������ȷ�Ӧ������MnO2Ҳ�ܷ������ȷ�Ӧ | |
| B�� | AlCl3�����NaOH��Һ��Ӧ����AlO2-���������NH3•H2O��ӦҲ����AlO2- | |
| C�� | Fe��Cl2��Ӧ����FeCl3������I2��ӦҲ����FeI3 | |
| D�� | Na2O2��CO2��Ӧ����Na2CO3��O2������SO2��ӦҲ����Na2SO3��O2 |
| A�� | ����Һ�м�KSCN����Һ�Ժ�ɫ��֤��ԭ��Һ����Fe3+����Fe2+ | |
| B�� | ����ͨ����ˮCuSO4����ĩ������֤��ԭ�����к���ˮ���� | |
| C�� | ���հ�ɫ��ĩ������ʻ�ɫ��֤��ԭ��ĩ����Na+����K+ | |
| D�� | �����ó���ʯ��ˮ������Na2CO3��Һ��NaHCO3��Һ |
| A�� | �ŵ�ʱ���������Һ��pH���Ͻ���������������PbSO4 ���� | |
| B�� | �ŵ�ʱ��ÿͨ��1mol���ӣ����ؾ�Ҫ����2mol H2SO4 | |
| C�� | ���ʱ��������Ӧ��PbSO4+2e-=Pb+SO42- | |
| D�� | ���ʱ��Ǧ���صĸ�������ӵ�Դ�ĸ������� |
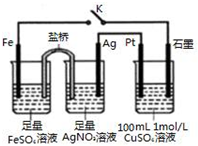
| A�� | �����еĵ���ʲ�������KCl | |
| B�� | ������ͨ��0.5 mol e-����CuSO4��Һ�м���12.4g CuCO3������Һ�ɻָ�ԭ��� | |
| C�� | �պ�K��Pt�缫�Ϸ����ķ�ӦΪ��4OH--4e-=O2��+2H2O | |
| D�� | �պ�K��������·�е���������Ϊ��Ag�缫��Pt�缫��ʯī�缫��Fe�缫��Ag�缫 |
| A�� | ����̼������Һ������ɫ�������ټ����ᣬ��ɫ������ʧ��һ����Ca2+ | |
| B�� | �ò�����պȡ������ˮ������pH��ֽ�����룬�����ɫ���Ƚϣ��ⶨ��ˮ��pH | |
| C�� | ij��Һ��NaOH��Һ���ȣ�����ʹʪ���ɫʯ����ֽ�������壬˵��ԭ��Һ�д���NH4+ | |
| D�� | �����Ȼ�����Һ�а�ɫ�����������ټ����ᣬ��������ʧ��һ����SO42- |