��Ŀ����
��֪A��B��C��D��E��Ϊ������Ԫ�أ�ԭ��������A��E��B��C��D��˳�����ε�����AԪ��ԭ���γɵ����Ӿ���һ�����ӣ�Cԭ�ӵ������������Ǵ�����3����C��D���γ����ֹ��廯�������һ��Ϊ����ɫ���壻B��C���γɶ�����̬�������A��B��C����Ԫ�ؿ��γ����ӻ�����X��X��A��B��CԪ��ԭ�ӵ����ʵ���֮��ΪA��B��C=4��2��3��Eԭ���������������ڲ��������2�����ش��������⣺
��1����д��Ԫ�ط��ţ�B ��C ��D ��E ��
��2��д��C��D�γɵĵ���ɫ���廯����ĵ���ʽ ��
��3��������X�������� ��
��4����B��CԪ�����Ԫ��ԭ��������ΪB��C=7��12�Ļ�����Ļ�ѧʽ ��
��5����A��B��C��E����Ԫ�ذ�ԭ�Ӹ�����Ϊ5��1��3��1��ɵĻ������ˮ��Һ��������NaOH��Һ�ڼ���ʱ������Ӧ�����ӷ���ʽΪ ��
��1����д��Ԫ�ط��ţ�B
��2��д��C��D�γɵĵ���ɫ���廯����ĵ���ʽ
��3��������X��������
��4����B��CԪ�����Ԫ��ԭ��������ΪB��C=7��12�Ļ�����Ļ�ѧʽ
��5����A��B��C��E����Ԫ�ذ�ԭ�Ӹ�����Ϊ5��1��3��1��ɵĻ������ˮ��Һ��������NaOH��Һ�ڼ���ʱ������Ӧ�����ӷ���ʽΪ
���㣺λ�ýṹ���ʵ����ϵӦ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
������A��B��C��D��E��Ϊ������Ԫ�أ�ԭ��������A��E��B��C��D��˳�����ε�����AԪ��ԭ���γɵ����Ӿ���һ�����ӣ���AΪHԪ�أ�Cԭ�ӵ������������Ǵ�����3����Cԭ��ֻ����2�����Ӳ㣬����������Ϊ6����CΪOԪ�أ�C��D���γ����ֹ��廯�������һ��Ϊ����ɫ���壬��DΪNa��B��C���γɶ�����̬�������BΪNԪ�أ���A��B��C����Ԫ�ؿ��γ����ӻ�����X��X��A��B��CԪ��ԭ�ӵ����ʵ���֮��ΪA��B��C=4��2��3����XΪNH4NO3��Eԭ���������������ڲ��������2����Eԭ��ֻ����2�����Ӳ㣬����������Ϊ4����EΪCԪ�أ��ݴ˽��
���
�⣺A��B��C��D��E��Ϊ������Ԫ�أ�ԭ��������A��E��B��C��D��˳�����ε�����AԪ��ԭ���γɵ����Ӿ���һ�����ӣ���AΪHԪ�أ�Cԭ�ӵ������������Ǵ�����3����Cԭ��ֻ����2�����Ӳ㣬����������Ϊ6����CΪOԪ�أ�C��D���γ����ֹ��廯�������һ��Ϊ����ɫ���壬��DΪNa��B��C���γɶ�����̬�������BΪNԪ�أ���A��B��C����Ԫ�ؿ��γ����ӻ�����X��X��A��B��CԪ��ԭ�ӵ����ʵ���֮��ΪA��B��C=4��2��3����XΪNH4NO3��Eԭ���������������ڲ��������2����Eԭ��ֻ����2�����Ӳ㣬����������Ϊ4����EΪCԪ�أ�
��1��������������֪��BΪNԪ�ء�CΪOԪ�ء�DΪNa��EΪCԪ�أ�
�ʴ�Ϊ��N��O��Na��C��
��2��C��D�γɵĵ���ɫ���廯����ΪNa2O2�������ʽΪ��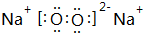 ��
��
�ʴ�Ϊ�� ��
��
��3��XΪNH4NO3������Ϊ������泥�
�ʴ�Ϊ������泥�
��4��N��OԪ�����Ԫ��ԭ��������=7��12����Nԭ����Oԭ����Ŀ֮��=
��
=2��3���û�����Ļ�ѧʽΪ��N2O3��
�ʴ�Ϊ��N2O3��
��5����H��N��O��C����Ԫ�ذ�ԭ�Ӹ�����Ϊ5��1��3��1��ɵĻ�����ΪNH4HCO3������������NaOH��Һ�ڼ���ʱ��Ӧ����̼���ơ�������ˮ����Ӧ�����ӷ���ʽΪ��NH4++HCO3-+2OH-
CO32-+NH3��+2H2O��
�ʴ�Ϊ��NH4++HCO3-+2OH-
CO32-+NH3��+2H2O��
��1��������������֪��BΪNԪ�ء�CΪOԪ�ء�DΪNa��EΪCԪ�أ�
�ʴ�Ϊ��N��O��Na��C��
��2��C��D�γɵĵ���ɫ���廯����ΪNa2O2�������ʽΪ��
 ��
���ʴ�Ϊ��
 ��
����3��XΪNH4NO3������Ϊ������泥�
�ʴ�Ϊ������泥�
��4��N��OԪ�����Ԫ��ԭ��������=7��12����Nԭ����Oԭ����Ŀ֮��=
| 7 |
| 14 |
| 12 |
| 16 |
�ʴ�Ϊ��N2O3��
��5����H��N��O��C����Ԫ�ذ�ԭ�Ӹ�����Ϊ5��1��3��1��ɵĻ�����ΪNH4HCO3������������NaOH��Һ�ڼ���ʱ��Ӧ����̼���ơ�������ˮ����Ӧ�����ӷ���ʽΪ��NH4++HCO3-+2OH-
| ||
�ʴ�Ϊ��NH4++HCO3-+2OH-
| ||
���������⿼��ṹ����λ�ù�ϵӦ�ã��ƶ�Ԫ���ǽ���ؼ������ضԻ�ѧ����Ŀ��飬�ѶȲ���

��ϰ��ϵ�д�
 �����ѧСѧ�꼶�νӽݾ��㽭��ѧ������ϵ�д�
�����ѧСѧ�꼶�νӽݾ��㽭��ѧ������ϵ�д�
�����Ŀ
�������ӷ���ʽ��д��ȷ���ǣ�������
A����ⱥ��ʳ��ˮ2H++2Cl?
| ||||
| B����NH4HSO3��Һ�е�������NaOH��Һ��HSO3-+OH-=SO32-+H2O | ||||
| C���������������м���������ϡ���Fe��OH��2+2H+=Fe2++2H2O | ||||
| D������AlCl3��Һ��С�մ���Һ��ϣ�Al3++3HCO3-�TAl��OH��3��+3CO2�� |
�����ĵ��´�ѩʹ�ҹ����������ġ�ů��������Ϊ������Ʒ��ů�������õ������ġ��������ȡ�ԭ����ʹ�䷢��ԭ��ط�Ӧ��������ԭ����г䵱��������
| A������ | B������ | C������ | D������ |
���з�Ӧ�����ӷ���ʽ������ǣ�������
| A�����������Һ��ͨ�����������̼��Ca2++2ClO-+H2O+CO2=CaCO3��+2HClO |
| B��������NaHSO4��Ba��OH��2��Һ��Ӧ��Ba2++2OH-+2H++SO42-=BaSO4��+2H2O |
| C������������Һ�м������������Һ��Fe2++2H2O2+4H+=Fe3++4H2O |
| D����������Һ�мӹ�����ˮ��Fe3++3NH3?H2O=Fe��OH��3��+3NH4+ |

 ��������HCl ��NaOH ��CH3COOH ��NH3?H2O ��CH3COONa ��NH4Cl
��������HCl ��NaOH ��CH3COOH ��NH3?H2O ��CH3COONa ��NH4Cl