��Ŀ����
2��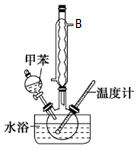 �����ױ����졢�䡢�����֣����ж������ױ���ҽҩ��Ⱦ�ϵȹ�ҵ��һ����Ҫ�л��м��壬������Ũ����Ϊ��������Ũ����Ϊ������ͨ���ױ���������Ӧ�Ʊ���һ���µ��Ʊ��������ױ���ʵ�鷽���ǣ��Է�������Ϊ������������NaHSO4Ϊ��������ѭ��ʹ�ã�����CCl4��Һ�У�����������������ˮ���ã���45�淴Ӧ1h����Ӧ�������ˣ���Һ�ֱ���5%NaHCO3����Һ��ˮϴ�����ԣ��پ������ᴿ�õ��������ױ���
�����ױ����졢�䡢�����֣����ж������ױ���ҽҩ��Ⱦ�ϵȹ�ҵ��һ����Ҫ�л��м��壬������Ũ����Ϊ��������Ũ����Ϊ������ͨ���ױ���������Ӧ�Ʊ���һ���µ��Ʊ��������ױ���ʵ�鷽���ǣ��Է�������Ϊ������������NaHSO4Ϊ��������ѭ��ʹ�ã�����CCl4��Һ�У�����������������ˮ���ã���45�淴Ӧ1h����Ӧ�������ˣ���Һ�ֱ���5%NaHCO3����Һ��ˮϴ�����ԣ��پ������ᴿ�õ��������ױ�����ش��������⣺
��1������B�����������������ܣ���������������������
��2�������ڷ�Һ©����ϴ�Ӿ����л��㴦���²㣨��ϡ����¡�����Һʱ��������Һ�����������������ԭ�����Һ©�����������⣬���з�Һ©���Ͽ�����δ��
��3�������µ����鷽���й��˵�Ŀ���ǻ���NaHSO4����Һ��5%NaHCO3��Һϴ�ӵ�ԭ���dz�ȥ���ᡢ���ᡢ���ᣮ
��4�����и����˴������༰�����Լױ�������ӦӰ���ʵ������
| ���� | n�������� | ���������и����칹������������%�� | �ܲ��� ��%�� | ||
| n���ױ��� | �������ױ� | �������ױ� | �������ױ� | ||
| ŨH2SO4 | 1.0 | 35.6 | 60.2 | 4.2 | 82.0 |
| 1.2 | 36.5 | 59.2 | 4.0 | 83.8 | |
NaHSO4 | 0.25 | 46.3 | 52.8 | 0.9 | 92.8 |
| 0.32 | 48.0 | 51.7 | 0.3 | 95.0 | |
| 0.36 | 45.0 | 54.4 | 0.6 | 93.9 | |
���ɱ������ݿ�֪�ױ�������Ҫ�õ��������ױ����������ױ���ԭ���DZ����ϵļ�ʹ���������Ӽ���̼ԭ�ӵ��ڶ�λ������Ӧ�������У�
����Ũ������ױ�������ȣ�NaHSO4���ױ����������������������ж������ױ�������ߣ����������������ѭ��ʹ�ã����ش�һ����
�ܶ������ױ����������ױ��IJ����������������
| �ܶ�/g•cm-3 | �е�/�� | �ܽ��� | |
| �ױ� | 0.866 | 110.6 | ������ˮ�������������ױ� |
| �������ױ� | 1.286 | 237.7 | ������ˮ��������Һ̬�� |
| �������ױ� | 1.162 | 222 | ������ˮ��������Һ̬�� |

���У�����1������Ϊ��Һ������2����Ҫʹ�����������е�ABC������ţ���
A�������� B���ƾ��� C���¶ȼ� D����Һ©�� E��������
���� ��1������BΪ���������ܣ���Ӧ��ױ��������ӷ��������ܵ�����������������
��2��CCl4���ܶȱ�ˮ���л������²㣻��Һ©�����Һ��Ų����������˷�Һ©�����������п����Ƿ�Һ©���Ͽڻ���δ��
��3������ʱ��Һ�к���NaHSO4����ѭ��ʹ�ã���Һ��NaHCO3��Һϴ�ӳ�ȥ�������ʣ�
��4���ٴ�����ױ���������ʵ���֮��Ϊ���������ߵ�ʱ������13.0g�ױ����ɵ������������ɱ��е����ݼ���������ױ���������
�ڴӼױ��Ľṹ���ǣ�
�۹۲���Ŀ�е����ݣ�����NaHSO4Ϊ����ʱ�Է�Ӧ����Ʊ����н�ǿ��ѡ���Դ����ã���߶������ױ��ı�����
�ܸ���ͼ�в����ϱ�����Ϣ�ɴ�
��� �⣺��1���������ṹ֪����BΪ���������ܣ���Ӧ��ױ��������ӷ��������ܵ�����������������
�ʴ�Ϊ�����������ܣ�����������
��2��CCl4���ܶȱ�ˮ���л������²㣻��Һ©�����Һ��Ų����������˷�Һ©�����������п����Ƿ�Һ©���Ͽڻ���δ��
�ʴ�Ϊ���£� ��Һ©���Ͽ�����δ��
��3��NaHSO4�ڸ÷�Ӧ����Ϊ��������˷�Ӧ����˵�Ŀ����Ϊ�˻���NaHSO4����Һ��NaHCO3��Һϴ�ӳ�ȥ�����������ᡢ���ᡢ���
�ʴ�Ϊ������NaHSO4����ȥ���ᡢ���ᡢ���
��4���ٴ�������ݷ�������������ױ��ı���Ϊ0.32ʱ���ܲ�������Ҷ������ױ��ĺ�����ߣ���ʱ��ʹ��13.0g�ױ������ݷ��̼ױ��������ױ����������ױ����ܲ���Ϊ95%�����ж������ױ�����������Ϊ48%����������ױ�������Ϊ��13g��$\frac{137g/mol}{92g/mol}$��95%��48%��8.8g��
�ʴ�Ϊ��0.32��8.8��
�ڱ����ϵļ�ʹ���������Ӽ���̼ԭ�ӵ��ڶ�λ������Ӧ�������У�
�ʴ�Ϊ�������ϵļ�ʹ���������Ӽ���̼ԭ�ӵ��ڶ�λ������Ӧ�������У�
����NaHSO4 ���������ŵ������������ж������ױ��ı�����ߡ�ͬʱ������ѭ��ʹ�ã�
�ʴ�Ϊ�������������ж������ױ�������ߣ����������������ѭ��ʹ�ã���
�ܾ�������1�õ��л������������������1Ϊ��Һ������ͼ������������ױ����������ױ��ķ���Ϊ������Ҫ�����ܡ��ƾ��ơ��¶ȼƣ�
�ʴ�Ϊ����Һ��ABC��
���� ������Ҫ����������ʵ����ʺ��Ʊ���ͬʱ���������ݵĴ���������������ܹ�Ѹ���ڱ�����ȡ��������Ϣ��������Ϣ����й����⣬ע�ضԻ���֪ʶ���飬��ǿ���������Ŀ��飮

 �óɼ�1��1��ĩ���100��ϵ�д�
�óɼ�1��1��ĩ���100��ϵ�д� ��״Ԫ���źþ�ϵ�д�
��״Ԫ���źþ�ϵ�д�| A�� | 31.6 g���������ȫ�ֽ����O2�����ԼΪ2.24 L | |
| B�� | ��25�桢101KPaʱ��24.5L��������������ԼΪ10NA | |
| C�� | 0.3 mol•L-1��������Һ������Na+��SO42-�����ʵ���Ϊ0.9 mol | |
| D�� | 25�桢101KPa�£�2.24 L������̼���������Ļ��������������ԭ����ԼΪ0.2NA |
| ������ | ������ | |
| A | Ư���ڿ����о��ñ��� | Ư���е�CaCl2������е�CO2��Ӧ����CaCO3 |
| B | �ǽ����ԣ�Cl��S | ���ԣ�HClO3��H2SO4 |
| C | �����£�NaHCO3���ܽ�ȱ�Na2CO3С | ��Na2CO3��Һ��ͨ��CO2�������� |
| D | �����£�SO2���������NaOH��Һ���ɷ�Ӧ | SO2������������ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | lgH2 �� 8gO2 | |
| B�� | 150�桢1.01��105Pa ʱ��18LH2O�� 18LCO2 | |
| C�� | 0.1 molHCl��2.24LHe | |
| D�� | ��״���£�2.24LCCl4��6.02��1022��CO���� |
| A�� | K+��Na+��NO3-��SO42- | B�� | K+��Na+��Cl-��Cr2O42- | ||
| C�� | Al3+��K+��AlO2-��Cl- | D�� | Fe3+��NO3-��SO42-��NH4+ |
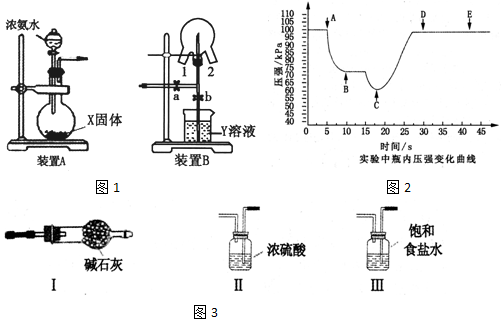
 Ϊ���ѧѧ����ȡ�õijɾ��Լ������������Ĺ��ף����Ϲ���2011�궨Ϊ�����ʻ�ѧ�ꡱ��������Ϊ����ѧ�������ǵ�������ǵ�δ��������ѧ��עʳƷ��ȫ�����ཡ�������Ŀɳ�����չ��Ϊ�����������������������������ˮƽ��������Ҫ���ã���ش��������⣺
Ϊ���ѧѧ����ȡ�õijɾ��Լ������������Ĺ��ף����Ϲ���2011�궨Ϊ�����ʻ�ѧ�ꡱ��������Ϊ����ѧ�������ǵ�������ǵ�δ��������ѧ��עʳƷ��ȫ�����ཡ�������Ŀɳ�����չ��Ϊ�����������������������������ˮƽ��������Ҫ���ã���ش��������⣺