��Ŀ����
 ��ϩ��������������ʯ�ͺ�ú�Ļ�������ԭ�ϣ��Ҵ������������������г������л������
��ϩ��������������ʯ�ͺ�ú�Ļ�������ԭ�ϣ��Ҵ������������������г������л��������1�����𱽡��Ҵ�������������ɫҺ�壬��ѡ����Լ���
��̼������Һ
����ɫʯ����Һ
����ˮ
�����Ը��������Һ
��2����һ�������£���ϩ��ˮ�����ӳɷ�Ӧ�����ò���Ľṹʽ��
��3������ͼʾװ����ȡ������������������գ�
���Թ�a�������Ũ���ᡢ��������Ҵ���2mL����ȷ�ļ���˳������
�ڷ�Ӧ��ѧ����ʽΪ
��Ϊ��ֹA�е�Һ����ʵ��ʱ�������У��ڼ���ǰӦ��ȡ�Ĵ�ʩ��
��ʵ���м����Թܵ�Ŀ���ǣ�
���Թ�b�м��б���Na2CO3��Һ����������
��Ӧ���������Թ�b���ã��۲쵽��������
���㣺������������ȡ,�л���ļ���
ר�⣺ʵ����,�л���Ӧ
��������1�������Ҵ�������ֱ��̼������Һ���֣��ֲ㡢���ܡ�ð���ݣ�
�����Ҵ�������ֱ����ɫʯ����Һ���֣��ֲ㡢���ܡ���죻
�����Ҵ�������ֱ����ˮ���֣��ֲ㡢���ܡ����ܣ�
�����Ҵ�������ֱ�����Ը��������Һ���֣��ֲ㡢��ɫ�����ܣ�
��2����ϩ��ˮ�����ӳ������Ҵ����Ҵ��Ĺ��������ǻ������Ҵ���Ϊͬ���칹���ֻ�м��ѣ�
��3����Ũ�����ܶȴ�Ӧ��Ũ������뵽�Ҵ��У��Է���Һ�ɽ��������ӷ�����ȴ���ټ������
���������Ҵ���Ũ���������¼��ȷ���������Ӧ��������������ˮ��CH3CH2OH+CH3COOH
CH3COOCH2CH3+H2O��
��Һ�����Ҫ�����Ƭ���ʯ����ֹ���У�
��ʵ���м����Թܵ�Ŀ�Ĵӻ�ѧ��Ӧ���ʺͻ�ѧƽ����ƶ������Ƕȷ�����
�ݱ���̼������Һ�����ᷴӦ��ȥ���ᡢͬʱ���������������ܽ�ȣ����ڷֲ㣻
��������������״̬���ܶȽ��з�����
�����Ҵ�������ֱ����ɫʯ����Һ���֣��ֲ㡢���ܡ���죻
�����Ҵ�������ֱ����ˮ���֣��ֲ㡢���ܡ����ܣ�
�����Ҵ�������ֱ�����Ը��������Һ���֣��ֲ㡢��ɫ�����ܣ�
��2����ϩ��ˮ�����ӳ������Ҵ����Ҵ��Ĺ��������ǻ������Ҵ���Ϊͬ���칹���ֻ�м��ѣ�
��3����Ũ�����ܶȴ�Ӧ��Ũ������뵽�Ҵ��У��Է���Һ�ɽ��������ӷ�����ȴ���ټ������
���������Ҵ���Ũ���������¼��ȷ���������Ӧ��������������ˮ��CH3CH2OH+CH3COOH
| ŨH2SO4 |
| �� |
��Һ�����Ҫ�����Ƭ���ʯ����ֹ���У�
��ʵ���м����Թܵ�Ŀ�Ĵӻ�ѧ��Ӧ���ʺͻ�ѧƽ����ƶ������Ƕȷ�����
�ݱ���̼������Һ�����ᷴӦ��ȥ���ᡢͬʱ���������������ܽ�ȣ����ڷֲ㣻
��������������״̬���ܶȽ��з�����
���
�⣺��1�������Ҵ�������ֱ��̼������Һ���֣��ֲ㡢���ܡ�ð���ݣ��ʢٿ�ѡ��
�����Ҵ�������ֱ����ɫʯ����Һ���֣��ֲ㡢���ܡ���죬�ʢڿ�ѡ��
�����Ҵ�������ֱ����ˮ���֣��ֲ㡢���ܡ����ܣ��ʢ۲�ѡ��
�����Ҵ�������ֱ�����Ը��������Һ���֣��ֲ㡢��ɫ�����ܣ��ʢܿ�ѡ��
�ʴ�Ϊ���٢ڢܣ�
��2����һ�������£���ϩ��ˮ�����ӳɷ�Ӧ�����Ҵ����ṹʽΪ�� ���Ҵ��Ĺ��������ǻ������Ҵ���Ϊͬ���칹���ֻ�м��ѣ���ṹ��ʽΪ��CH3OCH3��
���Ҵ��Ĺ��������ǻ������Ҵ���Ϊͬ���칹���ֻ�м��ѣ���ṹ��ʽΪ��CH3OCH3��
�ʴ�Ϊ�� ���ǻ���CH3OCH3��
���ǻ���CH3OCH3��
��3����Ϊ��ֹ��Һ�ɽ���Ӧ���ܶȴ��Һ����뵽�ܶ�С��Һ���У������ӷ�����ȴ���ټ������ᣬ
�ʴ�Ϊ���ȼ����Ҵ���Ȼ���ҡ���Թܱ���������Ũ���ᣬ�ټӱ����
���������Ҵ���Ũ���������¼��ȷ���������Ӧ��������������ˮ��CH3CH2OH+CH3COOH
CH3COOCH2CH3+H2O��
�ʴ�Ϊ��CH3CH2OH+CH3COOH
CH3COOCH2CH3+H2O��
��Һ�������Ҵ��е�ͣ�����Ҫ�����Ƭ����ֹ���У�
�ʴ�Ϊ�����Թ�a�м��뼸����ʯ�������Ƭ����
��ʵ���м����Թܣ��ٷ�Ӧ���¶ȸߣ���Ӧ�ٶȿ죻�ڸ÷�ӦΪ���淴Ӧ���¶ȸߣ����������ķе�ͣ��ӷ������Ի�ѧƽ��������Ӧ�����ƶ���������������������
�ʴ�Ϊ���ӿ췴Ӧ���ʣ���ʱ��������������������������ƽ�����������������ķ����ƶ���
���Ʊ���������ʱ���ñ���̼������Һ��Ŀ�����кͻӷ����������ᣬʹ֮ת��Ϊ����������ˮ�У��ܽ�ӷ��������Ҵ�����������������ˮ�е��ܽ�ȣ����ڷֲ�õ�����
�ʴ�Ϊ���к����ᣬ�ܽ��Ҵ�����������ˮ�е��ܽ������������������
���������������ܶȱ�ˮС��������̼������Һ���Ϸ�����ɫ��״Һ����֣��ʴ�Ϊ��b�е�Һ��ֲ㣬�ϲ���������״Һ�壮
�����Ҵ�������ֱ����ɫʯ����Һ���֣��ֲ㡢���ܡ���죬�ʢڿ�ѡ��
�����Ҵ�������ֱ����ˮ���֣��ֲ㡢���ܡ����ܣ��ʢ۲�ѡ��
�����Ҵ�������ֱ�����Ը��������Һ���֣��ֲ㡢��ɫ�����ܣ��ʢܿ�ѡ��
�ʴ�Ϊ���٢ڢܣ�
��2����һ�������£���ϩ��ˮ�����ӳɷ�Ӧ�����Ҵ����ṹʽΪ��
 ���Ҵ��Ĺ��������ǻ������Ҵ���Ϊͬ���칹���ֻ�м��ѣ���ṹ��ʽΪ��CH3OCH3��
���Ҵ��Ĺ��������ǻ������Ҵ���Ϊͬ���칹���ֻ�м��ѣ���ṹ��ʽΪ��CH3OCH3���ʴ�Ϊ��
 ���ǻ���CH3OCH3��
���ǻ���CH3OCH3����3����Ϊ��ֹ��Һ�ɽ���Ӧ���ܶȴ��Һ����뵽�ܶ�С��Һ���У������ӷ�����ȴ���ټ������ᣬ
�ʴ�Ϊ���ȼ����Ҵ���Ȼ���ҡ���Թܱ���������Ũ���ᣬ�ټӱ����
���������Ҵ���Ũ���������¼��ȷ���������Ӧ��������������ˮ��CH3CH2OH+CH3COOH
| ŨH2SO4 |
| �� |
�ʴ�Ϊ��CH3CH2OH+CH3COOH
| ŨH2SO4 |
| �� |
��Һ�������Ҵ��е�ͣ�����Ҫ�����Ƭ����ֹ���У�
�ʴ�Ϊ�����Թ�a�м��뼸����ʯ�������Ƭ����
��ʵ���м����Թܣ��ٷ�Ӧ���¶ȸߣ���Ӧ�ٶȿ죻�ڸ÷�ӦΪ���淴Ӧ���¶ȸߣ����������ķе�ͣ��ӷ������Ի�ѧƽ��������Ӧ�����ƶ���������������������
�ʴ�Ϊ���ӿ췴Ӧ���ʣ���ʱ��������������������������ƽ�����������������ķ����ƶ���
���Ʊ���������ʱ���ñ���̼������Һ��Ŀ�����кͻӷ����������ᣬʹ֮ת��Ϊ����������ˮ�У��ܽ�ӷ��������Ҵ�����������������ˮ�е��ܽ�ȣ����ڷֲ�õ�����
�ʴ�Ϊ���к����ᣬ�ܽ��Ҵ�����������ˮ�е��ܽ������������������
���������������ܶȱ�ˮС��������̼������Һ���Ϸ�����ɫ��״Һ����֣��ʴ�Ϊ��b�е�Һ��ֲ㣬�ϲ���������״Һ�壮
���������⿼�����л�������ֺ������������Ʊ�����Ŀ�ѶȲ����ʱ��ע��������Ӧ��ԭ���ͱ���̼������Һ�����ã�

��ϰ��ϵ�д�
�����Ŀ
��2mol����A��3mol����B��һ�������·�����Ӧ��A��g��+3B��g��?2C��g����H��0���ﵽƽ��ʱ��B��ת����Ϊ50%����A��ת����Ϊ��������
| A��17% | B��20% |
| C��25% | D��50% |
�ࣨSb������Ȼ��һ�����������ʽ���ڣ��ҹ�����̲���ռ�����һ������������ȡ������һ�������ڸ����½�����ת��Ϊ���������̼��ԭ��2Sb2S3+3O2+6Fe=Sb4O6+6FeS��Sb4O6+6C=4Sb+6CO��
���ڷ�Ӧ�١���Ӧ�ڵ�˵����ȷ���ǣ�������
���ڷ�Ӧ�١���Ӧ�ڵ�˵����ȷ���ǣ�������
| A����Ӧ�ٵ���������Sb2S3����Ӧ�ڵ���������Sb4O6 |
| B����Ӧ����ÿ����3 mol FeSʱ����ת��6 mol���� |
| C����Ӧ��˵��������Sb�Ļ�ԭ�Ա�Cǿ |
| D����Ӧ�ٵ�����������Sb4O6��FeS����Ӧ�ڵ�����������CO |
 ��ͼ�������Լ�ƿ��ǩ�ϵIJ���˵�����֣���
��ͼ�������Լ�ƿ��ǩ�ϵIJ���˵�����֣���
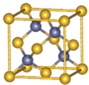 �黯��Ϊ�������뵼����ϣ������ṹ��ͼ��ʾ��
�黯��Ϊ�������뵼����ϣ������ṹ��ͼ��ʾ�� ��139mL�ܶ�Ϊ1.05g/cm3��CaCl2��HCl�����Һ�еμ�Na2CO3��Һ��������ɵij���������y�ᣩ�����Na2CO3��Һ�������x�ᣩ�Ĺ�ϵ��ͼ����֪ԭ��Һ�к�CaCl2Ϊ7.6%��ǡ����ȫ��Ӧʱ������Na2CO3��Һ��������Ϊ191.9g�����ʱ��Һ�ܶ�Ϊ1.02g/cm3����
��139mL�ܶ�Ϊ1.05g/cm3��CaCl2��HCl�����Һ�еμ�Na2CO3��Һ��������ɵij���������y�ᣩ�����Na2CO3��Һ�������x�ᣩ�Ĺ�ϵ��ͼ����֪ԭ��Һ�к�CaCl2Ϊ7.6%��ǡ����ȫ��Ӧʱ������Na2CO3��Һ��������Ϊ191.9g�����ʱ��Һ�ܶ�Ϊ1.02g/cm3����