��Ŀ����
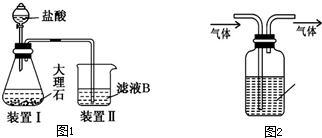
10�������к��зḻ�ĵ⣮Ϊ�˴Ӻ�������ȡ�⣬ij�о���ѧϰС����Ʋ�����������ʵ�飮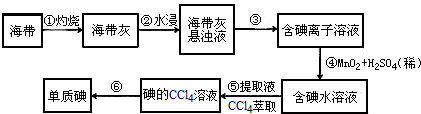
��ش������й����⣺
��1�����պ���ʱ���ǽ������õĺ����þƾ�ʪ����ڸɹ��н��У������оƾ�����ʪ���Ŀ���Ǵٽ���������ȼ�գ�
��2������۵�ʵ����������ǹ��ˣ���ʵ���ҽ��и������ʱ���õ��IJ�����������©��������������ձ���
��3������ܷ�Ӧ�����ӷ���ʽΪ2I-+MnO2+4H+=I2+Mn2++2H2O���ò�����������������������ˮ��H2O2�������
��4����CCl4�ӵ�ˮ����ȡ�Ⲣ�÷�Һ©������������Һ����ʵ������ɷֽ�Ϊ���¼�����
��A����ʢ����Һ�ķ�Һ©����������̨����Ȧ�ϣ�
��B����50mL��ˮ��15mLCCl4�����Һ©���У����Ǻò�������
��C�������Һ©���Ļ������ϿڵIJ������Ƿ�©ˮ��
��D����ת©������������ʱ�����������������رջ������ѷ�Һ©��������
��E���������������ձ�������Һ��
��F���ӷ�Һ©���Ͽڵ����ϲ�ˮ��Һ��
��G����©���ϿڵIJ�������ʹ���ϱ߰��ۻ�С��©�����ϵ�С�ף�
��H�����á��ֲ㣮
����ȷ���������˳���ǣ��������������ı����ĸ��գ���
C��B��D��A��G��H��E��F��
�ڣ�G�����������Ŀ���DZ�֤©����Һ��˳��������
��5����֪3I2+6OH-=5I-+IO3-+3H2O��5I-+IO3-+6H+=3I2��+3H2O���ڽ��еڢ�����ʱ�����ڵ��CCl4�м�������40%NaOH��Һ����CCl4���Ϊ��ɫ����Һ���������IJ���������������ˮ��Һ�м��������ϡ���ᣬ���˵õⵥ�ʣ�
���� �Ӻ�������ȡ�⣺�������պ�õ������ҽ��ݺ�õ������ҵ���Һ��������Һ����Ϊ�����ͺ���������ҺӦѡ����˵ķ��������˵õ��������ӵ���Һ����������̺�ϡ��������������Ϊ�ⵥ�ʣ��õ�����ˮ��Һ�������л��ܼ����Ȼ�̼����ȡ��Һ�õ���������Ȼ�̼��Һ��ͨ������õ��ⵥ�ʣ�
��1���ƾ�Ϊ��ȼ��Ʒ���ƾ�����ʪ�����ܴٽ���������ȼ�գ�
��2��������ǽ�����Һ����ɺ������ӵ���ҹ�Ͳ������ʲ����ǹ��ˣ����ݹ��˲�������ѡ�������������õ����������ձ���©�����������ȣ�
��3��MnO2���н�ǿ�������ԣ������������������¿ɱ�MnO2�������ɵ��ʵ⣬�ò�����������������������ˮ����������������
��4���ٰ���ȡ����Һ�����������ӷ�Һ©�����²�Һ�壬�����Ͽڵ����ϲ���Һ���ڴ�Һ©���Ͽڲ�������ʹ���ϵİ��۶�©���Ͽڵ�С�ף���ʹ��Һ©������ѹ��ƽ�⣬ʹҺ���������£�
��5������NaOH��Һ��Ӧ����NaI��NaIO��ˮ��CCl4���Ϊ��ɫ����Һ����������ˮ��Һ�м��������ϡ���ᣬ�����������£�+1�۵ĵ��-1�۵ĵⷢ��������ԭ��Ӧ���ɵⵥ�ʣ����˵õⵥ�ʣ�
��� �⣺��1���ƾ�Ϊ��ȼ��Ʒ��ȼ�����ɶ�����̼��ˮ���þƾ�����ʪ�����õĺ�����Ȼ��������գ��ܴٽ���������ȼ�գ�
�ʴ�Ϊ���ٽ���������ȼ�գ�
��2����������Һ���ù��ˣ��������պ�õ������ҽ��ݺ�õ������ҵ���Һ��������Һ����Ϊ�����ͺ���������ҺӦѡ����˵ķ���������ʱ��Ҫ������������©�����̶�����������̨�������õIJ��������н���Һ���ձ��������õ��IJ�����������©��������������ձ���
�ʴ�Ϊ�����ˣ��ձ���
��3�������������������¿ɱ�MnO2�������ɵ��ʵ⣺���ӷ���ʽΪ��2I-+MnO2+4H+=Mn2++I2+2H2O��ͨ��������Cl2+2I-=I2+2Cl-�����������Ӻ��������������Ϊ��������Ҳ�ܽ�������ת��Ϊ���ʵ⣬���ӷ���ʽΪ2H++2I-+H2O2�TI2+2H2O��
�ʴ�Ϊ��2I-+MnO2+4H+=I2+Mn2++2H2O����ˮ��H2O2�ȣ�
��4���ټ���Һ©���Ƿ�©Һ����50mL��ˮ��15mLCCl4�����Һ©���У��Ǻò���������ת��Һ©��������ʢ����Һ�ķ�Һ©����������̨����Ȧ�ϡ����ã��ֲ����Һ©���Ͽڲ�������ʹ���ϵİ��۶�©���Ͽڵ�С�ס��������������ձ�ʢ����Һ���ӷ�Һ©���Ͽڵ����ϲ���Һ��������ȷ�IJ���˳���ǣ�CBDAGH��
�ʴ�Ϊ��CBDAGH��
�ڲ�������Һ���ڴ���ѹ��������������Ŀ���DZ��ַ�Һ©������ѹ��ƽ�⣬ʹҺ���������£�
�ʴ�Ϊ����֤©����Һ��˳��������
��5������������Ʒ�Ӧ���ɵ⻯�ơ��ε����ƺ�ˮ����Ӧ�Ļ�ѧ����ʽΪ��I2+2NaOH�TNaI+NaIO+H2O���ⵥ�ʱ����ģ�CCl4���Ϊ��ɫ����Һ����������ˮ��Һ�м��������ϡ���ᣬ�����������£�+1�۵ĵ��-1�۵ĵⷢ��������ԭ��ӦI-+IO-+2H+=I2+H2O�����˵õⵥ�ʣ�
�ʴ�Ϊ���ޣ�������ˮ��Һ�м��������ϡ���ᣬ���˵õⵥ�ʣ�
���� ���⿼���˺�ˮ��Դ���ۺ����ã�Ϊ��Ƶ���㣬�漰֪ʶ��϶࣬����ʵ�����̼������ķ�Ӧ����ȡ����ѡȡ��������ʵ��ԭ����װ�õ�֪ʶ��Ϊ���Ĺؼ������ظ�Ƶ����Ŀ��飬��Ŀ�Ѷ��еȣ�

| A�� | �����۲�����ɫ��ζ�������Һ�У�K+��Na+��Cl-��NO3- | |
| B�� | ��ʹʯ����Һ��������ɫ����Һ�У�Na+��OH-��SO32-��I- | |
| C�� | ij������Һ�У�Fe3+��K+��SCN-��Cl- | |
| D�� | ��ˮϡ��ʱ$\frac{c��{H}^{+}��}{c��O{H}^{-}��}$��ֵ���Լ�С����Һ��Al3+��Cl-��NO3-��Na+ |
 ú̿ȼ�ջ����������̼������������к����壬��Ⱦ������
ú̿ȼ�ջ����������̼������������к����壬��Ⱦ��������1����������������������Լ�����֪��
4NO2��g��+O2��g��?2N2O5��g����H=-56.70kJ•mol-1
3O2��g��?2O3��g����H=+28.80kJ•mol-1
��������Ӧ2NO2��g��+O3��g��?N2O5��g��+O2��g���ġ�H=-42.75kJ•mol-1kJ•mol-1��
��2����������Ӧ�ں����ܱ������н��У�����˵����ȷ����abd��
a�������¶ȣ�ƽ�ⳣ����С
b������O3���������NO2ת����
c�������¶ȣ��������NO2��ת���ʣ����ܼӿ췴Ӧ����
d����ͼ��ʾ��t1ʱʹ���˴���
��3��ijʵ��С��ģ��ϳɼ״��Ĺ��̣���6molCO2��8molH2����һ�ݻ�Ϊ2L���ܱ������У�������ӦCO2��g��+3H2��g��?CH3OH��g��+H20��g����H=-49.0kJ•mol-1�����H2�����ʵ�����ʱ��仯���±���ʾ���ش��������⣺
| ʱ��/min | 0 | 1 | 3 | 8 | 11 |
| n��H2��mol | 8 | 6 | 3 | 2 | 2 |
��4�����ݰ�������˹��ʽ��֪����ѧ��Ӧ���ʳ������¶ȱ仯�Ĺ�ϵΪk=Ae��-Ea/Rr��������EaΪ��ܣ�RΪ������AΪ������ij�����������ͬ�¶��£����EaԽ��ѧ��Ӧ���ʳ���kԽС���Խ��ԽС����
��5��ij�о�С�����۶�Li2CO3������ʣ���ԭCO2��ʯī���������������ĵ缫��Ӧʽ3CO2+4e-=C+CO32-��
| A�� | ��λʱ����ÿ����1molN2��ͬʱ����2molNH3 | |
| B�� | c��N2����c��H2����c��NH3��=1��1��1 | |
| C�� | N2��H2�����ʵ�����NH3�����ʵ������ | |
| D�� | N2��H2��NH3�������������ٸı� |
��C60��C70�����ʯ��ʯī��
��C2H5OH��CH3OCH3
��${\;}_{6}^{12}$C��${\;}_{6}^{13}$C��${\;}_{6}^{14}$
��HOCH2COOH��HOCH2CH2COOH��HOCH2CH2CH2COOH��
��
 ��
��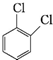
| A�� | �٢ڢۢݢ� | B�� | �ۢݢܢ٢� | C�� | �ܢڢ٢ݢ� | D�� | �ۢܢڢ٢� |
 �������ͷӲ�Ӧ���еĻ�ѧ�����ǣ�������
�������ͷӲ�Ӧ���еĻ�ѧ�����ǣ�������| A�� | ���Է����Ӿ۷�Ӧ | B�� | ������ǿ����Һ��Ӧ | ||
| C�� | ��NaHCO3��Ӧ�ų�CO2 | D�� | ������ˮ�����ӳɷ�Ӧ |
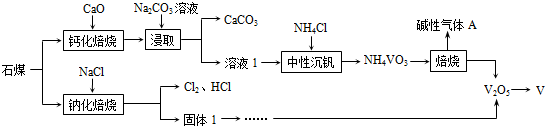
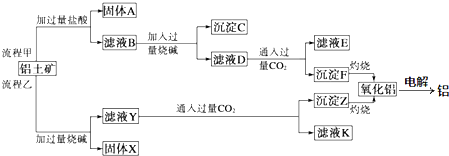
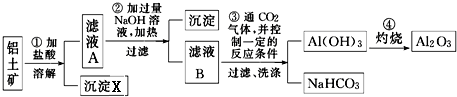
 ��
��